This blog entry focuses on a specific aspect of telomere biology, the epigenetic regulation of telomeres as mediated by TERRA, a recently-discovered RNA. Most popular discussions related to telomeres and telomerase are still based on simplistic earlier models of telomerase biology which do not take TERRA into account. So are the increasingly-popular commercial approaches to telomere lengthening via “telomerase activator” substances. The new TERRA-related research may explain how diet, certain supplements and lifestyle factors like exercise so profoundly affect telomere lengths and can inhibit cell senescence.
First, as a preamble or appetizer for what follows, regarding the relationship of telomere length and aging I invite you to view this 3-minute video.
Background on telomeres and telomerase.
Although this blog entry focuses on epigenetic regulation as a new and very important aspect of telomere biology, I continue to stand behind what I have written related to telomeres and telomerase reflecting a shift over a three-year period. The most-recent relevant blog entries were written in October 2010: Telomere lengths, Part 3: Selected current research on telomere-related signaling, telomere lengths, cancers and disease processes, Part2: lifestyle, dietary, and other factors associated with telomere shortening and lengthening, and Part1: telomere lengths, cancers and disease processes. These entries contain a great deal of information as well as links to multiple earlier blog entries on telomeres and telomerase. And, of course, the 12th theory of aging discussed in my treatise is Telomere Shortening and Damage. Three years ago, I thought that taking astragaloside IV or cycloastragenol supplements to extend telomeres was possibly a good anti-aging intervention. I no longer see that as the case.
In the abovementioned Part 3 blog entry I discussed how damaged or too-shortened telomeres can lead to epigenetic changes. “Another important contribution to the picture of how cell senescence affects aging is provided by the October 2010 publication Reduced histone biosynthesis and chromatin changes arising from a damage signal at telomeres.
“During replicative aging of primary cells morphological transformations occur, the expression pattern is altered and chromatin changes globally. Here we show that chronic damage signals, probably caused by telomere processing, affect expression of histones and lead to their depletion. We investigated the abundance and cell cycle expression of histones and histone chaperones and found defects in histone biosynthesis during replicative aging. Simultaneously, epigenetic marks were redistributed across the phases of the cell cycle and the DNA damage response (DDR) machinery was activated. The age-dependent reprogramming affected telomeric chromatin itself, which was progressively destabilized, leading to a boost of the telomere-associated DDR with each successive cell cycle.” — This same publication puts forward an important new concept: “We propose a mechanism in which changes in the structural and epigenetic integrity of telomeres affect core histones and their chaperones, enforcing a self-perpetuating pathway of global epigenetic changes that ultimately leads to senescence.” Histones, we recall, are the spindles around which DNA is wrapped, and chaperones are “are proteins that assist the non-covalent folding or unfolding and the assembly or disassembly of other macromolecular structures.” in this case the chaperones are ones that assist folding of DNA in histone structures, HSP90 being a main one discussed below.”
Rather than discussing how shortened or damaged telomeres lead to epigenetic changes, the present blog entry is focused on how epigenetic changes arising from other causes (dietary, lifestyle, aging, etc.) can affect telomere health, regulate telomere lengths, and impinge on cell senescence.
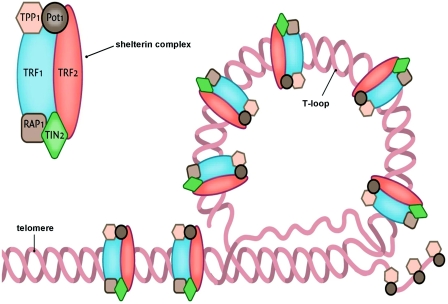
About telomere structures, shelterin and telomere binding proteins
Telomeres are commonly described as simple repeats of inert DNA at the ends of chromosomes, like the tips of shoelaces. In fact, they are quite complex structures. The 2011 publication Healthy aging and disease: role for telomere biology? Contains the following description and diagrams: “Telomeres are specialized DNA structures located at the terminal ends of the chromosomes [5]. Their primary function is to maintain genome stability. Telomeres consist of non-coding double-stranded repeats of guanine-rich tandem DNA sequences (TTAGGG) that are extended 9–15 kb in humans and end in a 50–300 nucleotide 3′ single guanine strand overhang [6]. This overhang can fold back and invade the double-stranded telomere helix, forming a large ‘T-loop’. The T-loop facilitates the formation of a high-order structure mediating the end-capping [7] (Figure 1) (ref).”
“The stability of the T-loop is largely dependent on the integrity of the associated telomere-specific proteins, also called the shelterin complex [8] (Figure 2). The proteins involved in the shelterin complex include TRF (telomeric repeat-binding factor) 1 and TRF2, which bind to the double-stranded segment of telomeric DNA [9,10] (ref).”
“POT1 (protein protection of telomeres 1) binds directly to the single-stranded telomeric DNA and interacts directly with TPP1 (tripeptidyl peptidase 1) [11]. Rap1 (repressor activator protein 1) binds TRF2 [12], and TIN2 (TRF1-interacting nuclear factor 2) is a central component of the complex interacting with TRF1, TRF2 and TPP1 [13,14]. The telomere complex plays a critical role by protecting the chromosomes from recognition by the DNA damage-repair system as DNA ‘breaks’ and activation of the p53 or p16INK4a pathway, eventually leading to cellular senescence or apoptosis [15](ref).”
Since these diagrams don’t show TERRA, the actual situation is even more complex. TERRA accompanies the telomeric DNA binding to TRF2.
The 2009 publication TERRA: telomeric repeat-containing RNA provides additional detail: “Telomeres, the physical ends of eukaryotic chromosomes, consist of tandem arrays of short DNA repeats and a large set of specialized proteins. A recent analysis has identified telomeric repeat-containing RNA (TERRA), a large non-coding RNA in animals and fungi, which forms an integral component of telomeric heterochromatin. — TERRA transcription occurs at most or all chromosome ends and it is regulated by RNA surveillance factors and in response to changes in telomere length. TERRA functions that are emerging suggest important roles in the regulation of telomerase and in orchestrating chromatin remodelling throughout development and cellular differentiation. — The accumulation of TERRA at telomeres can also interfere with telomere replication, leading to a sudden loss of telomere tracts. Such a phenotype can be observed upon impairment of the RNA surveillance machinery or in cells from ICF (Immunodeficiency, Centromeric region instability, Facial anomalies) patients, in which TERRA is upregulated because of DNA methylation defects in the subtelomeric region. Thus, TERRA may mediate several crucial functions at the telomeres, a region of the genome that had been considered to be transcriptionally silent.”
While the role of TERRA in regulation of telomeres is central to this blog entry, I have referred to it in the Part 3 post mentioned above. “The 2009 publication TERRA RNA Binding to TRF2 Facilitates Heterochromatin Formation and ORC Recruitment at Telomeres is one of several recent publications illustrating the wheels-within-wheels complexity of telomere formation and maintenance. “Telomere-repeat-encoding RNA (referred to as TERRA) has been identified as a potential component of yeast and mammalian telomeres. We show here that TERRA RNA interacts with several telomere-associated proteins, including telomere repeat factors 1 (TRF1) and 2 (TRF2), subunits of the origin recognition complex (ORC), heterochromatin protein 1 (HP1), histone H3 trimethyl K9 (H3 K9me3), and members of the DNA-damage-sensing pathway. siRNA depletion of TERRA caused an increase in telomere dysfunction-induced foci, aberrations in metaphase telomeres, and a loss of histone H3 K9me3 and ORC at telomere repeat DNA. Previous studies found that TRF2 amino-terminal GAR domain recruited ORC to telomeres. We now show that TERRA RNA can interact directly with the TRF2 GAR and ORC1 to form a stable ternary complex. We conclude that TERRA facilitates TRF2 interaction with ORC and plays a central role in telomere structural maintenance and heterochromatin formation.”
TERRA plays a role in mammalian telomere length regulation
The 2010 publication Chromatin regulation and non-coding RNAs at mammalian telomeres reports “In eukaryotes, terminal chromosome repeats are bound by a specialized nucleoprotein complex that controls telomere length and protects chromosome ends from DNA repair and degradation. In mammals the “shelterin” complex mediates these central functions at telomeres. In the recent years it has become evident that also the heterochromatic structure of mammalian telomeres is implicated in telomere length regulation. — Impaired telomeric chromatin compaction results in a loss of telomere length control. Progressive telomere shortening affects chromatin compaction at telomeric and subtelomeric repeats and activates alternative telomere maintenance mechanisms. Dynamics of chromatin structure of telomeres during early mammalian development and nuclear reprogramming further indicates a central role of telomeric heterochromatin in organismal development. In addition, the recent discovery that telomeres are transcribed, giving rise to UUAGGG-repeat containing TelRNAs/TERRA, opens a new level of chromatin regulation at telomeres. Understanding the links between the epigenetic status of telomeres, TERRA/TelRNA and telomere homeostasis will open new avenues for our understanding of organismal development, cancer and ageing.”
TERRA regulates and can inhibit telomerase expression
The 2010 publication The non-coding RNA TERRA is a natural ligand and direct inhibitor of human telomerase reports “Telomeres, the physical ends of eukaryotes chromosomes are transcribed into telomeric repeat containing RNA (TERRA), a large non-coding RNA of unknown function, which forms an integral part of telomeric heterochromatin. — TERRA molecules resemble in sequence the telomeric DNA substrate as they contain 5′-UUAGGG-3′ repeats near their 3′-end which are complementary to the template sequence of telomerase RNA. Here we demonstrate that endogenous TERRA is bound to human telomerase in cell extracts. Using in vitro reconstituted telomerase and synthetic TERRA molecules we demonstrate that the 5′-UUAGGG-3′ repeats of TERRA base pair with the RNA template of the telomerase RNA moiety (TR). — In addition TERRA contacts the telomerase reverse transcriptase (TERT) protein subunit independently of hTR. In vitro studies further demonstrate that TERRA is not used as a telomerase substrate. Instead, TERRA acts as a potent competitive inhibitor for telomeric DNA in addition to exerting an uncompetitive mode of inhibition. Our data identify TERRA as a telomerase ligand and natural direct inhibitor of human telomerase. Telomerase regulation by the telomere substrate may be mediated via its transcription.”
In practical terms: There may be plenty of telomerase present on telomeres. That telomerase will do no good to lengthen the telomeres, however, as long as it is bound up by TERRA
Epigenetic factors are key in determining telomerase activation, e.g. transcription of the telomerase gene hTERT
As pointed out in the Barzilai video, genetics plays a role in telomere length determination. The centenarians tend to have certain mutations associated with the hTERT gene. It has also long been known that many environmental, lifestyle and personal historical factors are also strongly determinative of telomere lengths. See for example the blog entry Part2: lifestyle, dietary, and other factors associated with telomere shortening and lengthening. But the mechanisms through which such factors affect telomere lengths were not understood until recently. There is increasing evidence that the mechanisms affecting telomerase transcription are epigenetic and that TERRA is centrally involved.
The role of epigenetics in regulating telomeres has been suspected for some time and was laid out in the 2007 publication The epigenetic regulation of mammalian telomeres. “Increasing evidence indicates that chromatin modifications are important regulators of mammalian telomeres. Telomeres provide well studied paradigms of heterochromatin formation in yeast and flies, and recent studies have shown that mammalian telomeres and subtelomeric regions are also enriched in epigenetic marks that are characteristic of heterochromatin. Furthermore, the abrogation of master epigenetic regulators, such as histone methyltransferases and DNA methyltransferases, correlates with loss of telomere-length control, and telomere shortening to a critical length affects the epigenetic status of telomeres and subtelomeres. These links between epigenetic status and telomere-length regulation provide important new avenues for understanding processes such as cancer development and ageing, which are characterized by telomere-length defects.”
“It is becoming increasingly apparent that epigenetic modifications including CpG methylation, histone methylation and acetylation is important for TERT transcriptional regulation(ref).” This in part has become clear through studies focusing on cancers. “The chromatin structure via the DNA methylation or modulation of nucleosome histones has recently been suggested to be important for regulation of the hTERT promoter. DNA unmethylation or histone methylation around the transcription start site of the hTERT promoter triggers the recruitment of histone acetyltransferase (HAT) activity, allowing hTERT transcription(ref). For example, for hepatocellular carcinoma (HCC) the fifth most common cancer worldwide, “Thus, we propose that hTERT is regulated by a combination of epigenetic mechanisms (DNA methylation and histone modifications) and by the transcription factor c-myc in HCC(ref).”
The 2011 publication Telomeres in cancer and ageing reports “Furthermore, telomere length and integrity are regulated by a number of epigenetic modifications, thus pointing to higher order control of telomere function. In this regard, we have recently discovered that telomeres are transcribed generating long, non-coding RNAs, which remain associated with the telomeric chromatin and are likely to have important roles in telomere regulation.” The authors are talking about TERRA. In the same publication “Telomeric chromatin is also enriched in epigenetic marks that are characteristic of constitutive heterochromatin, such as histone tri-methylation and DNA hypermethylation, which act as negative regulators of telomere length and telomere recombination [2].” Also, “We also demonstrated that the telomeres of iPS cells acquire the epigenetic marks of the telomeres of embryonic stem cells, among them a low density of trimethylated histones H3K9 and H4K20, and that in the iPS cells there is a loss of telomeric silencing and an increase in the abundance of TERRA (telomeric transcripts) levels [29].”
Normal cells and cancer cells have different epigenetic telomere maintenance mechanisms via silencing of TERRA or avoiding TERRA and using ALT
The 2008 publication Telomerase activity is associated with an increase in DNA methylation at the proximal subtelomere and a reduction in telomeric transcription reports: “Tumours and immortalized cells avoid telomere attrition by using either the ribonucleoprotein enzyme telomerase or a recombination-based alternative lengthening of telomeres (ALT) mechanism. Available evidence from mice suggests that the epigenetic state of the telomere may influence the mechanism of telomere maintenance, but this has not been directly tested in human cancer. Here we investigated cytosine methylation directly adjacent to the telomere as a marker of the telomere’s epigenetic state in a panel of human cell lines. We find that while ALT cells show highly heterogeneous patterns of subtelomeric methylation, subtelomeric regions in telomerase-positive cells invariably show denser methylation than normal cells, being almost completely methylated. When compared to matched normal and ALT cells, telomerase-positive cells also exhibit reduced levels of the telomeric repeat-containing-RNA (TERRA), whose transcription originates in the subtelomere. Our results are consistent with the notion that TERRA may inhibit telomerase: the heavy cytosine methylation we observe in telomerase-positive cells may reflect selection for TERRA silencing in order to facilitate telomerase activity at the telomere. These data suggest that the epigenetic differences between telomerase-positive and ALT cells may underlie the mechanism of telomere maintenance in human tumorigenesis and highlight the broad reaching consequences of epigenetic dysregulation in cancer.”
Grainyhead-like 2 is a telomerase gene transcription factor that epigenetically regulates cell lifespans
I continue to be amazed by the continuing discovery of new substances relevant to human longevity. In this case it is Grainyhead-like 2 (GRHL2). The December 2010 publication Grainyhead-like 2 enhances the human telomerase reverse transcriptase gene expression by inhibiting DNA methylation at the 5′-CpG island in normal human keratinocytes reports: “We recently identified Grainyhead-like 2 (GRHL2) as a novel transcription factor that binds to and regulates the activity of the human telomerase reverse transcriptase (hTERT) gene promoter. – In this study, we investigated the biological functions of GRHL2 and the molecular mechanism underlying hTERT gene regulation by GRHL2. Retroviral transduction of GRHL2 in normal human keratinocytes (NHK) led to a significant extension of replicative life span, whereas GRHL2 knockdown notably repressed telomerase activity and cell proliferation. — Using promoter magnetic precipitation coupled with Western blotting, we confirmed the binding of GRHL2 to the hTERT promoter and mapped the minimal binding region at -53 to -13 of the promoter. Furthermore, mutation analysis revealed the three nucleotides from -21 to -19 to be critical for GRHL2 binding. — Because hTERT expression is regulated in part by DNA methylation, we determined the effects of GRHL2 on the methylation status of the hTERT promoter. Senescent NHK exhibited hypermethylation of the CpG island, which occurred with the loss of hTERT expression. On the contrary, the promoter remained hypomethylated in GRHL2-transduced NHK, irrespective of cell proliferation status. — Also, knockdown of endogenous GRHL2 led to hypermethylation of the promoter. These results indicate that GRHL2 regulates the hTERT expression through an epigenetic mechanism and controls the cellular life span.”
The epigenetic reprogramming required to create induced pluripotent stem cells affects TERRA
As usual there is more related research going on out there beyond what I can review here, including that reported in these selected publications:
Promoting transcription of chromosome ends. (Nov. 2010)
Wrapping it up
Key take-away observations from this richness of materials are:
· Telomere lengths tend to get shorter with age but not necessarily uniformly either in time or within a population cohort. As Dr. Barzilai points out in the video, some centenarians have longer telomeres than other people 30 years their junior. Telomere lengths are regulated dynamically with complex epigenetic factors playing central roles.
· The epigenetic factors regulate telomere lengths via several pathways and a newly-discovered RNA called TERRA is a central actor in that regulation. One of the key mechanisms involved in GPC island methylation of the hTERT telomerase gene.
· This epigenetic regulation is how lifestyle and dietary factors affect telomere lengths, a matter I have discussed extensively in previous blog entries. Chronic stress leads to shorter telomeres; a stable and happy lifestyle leads to longer ones, etc.
· Since epigenetic factors affect telomere lengths and shortened telomere lengths themselves generate epigenetic changes that have multiple impacts on cell biology, it appears that both of the following statements are true “Aging drives telomere lengths” and “Telomere lengths drive aging.” It is a chicken-and-egg situation.
· This does not mean that interventions aimed at extending telomere lengths are necessarily anti-aging interventions. Such interventions can normalize lifespans of mice whose lives are shorter because of induced telomerase deficiency, but have not been shown to extend the lives of normal mice. See the blog entry Mouse age reversal – very interesting but misrepresented research.
· Personally, I think we are already past the time to give up the simplistic notion that normal people can take a telomerase-activator supplement and therefore assure having longer telomeres and live longer. Neither our basic understanding nor experimental studies bear that conjecture out. On the other hand there is much research pointing to simple and practical lifestyle and dietary interventions that are likely to keep telomeres longer. See my blog entry Part2: lifestyle, dietary, and other factors associated with telomere shortening and lengthening.
· We still have only an initial and incomplete understanding of how the epigenetic programming of telomeres related to TERRA works. That whole area of research is barely 3 years old though other aspects of telomere biology have been studied for decades. As we learn more about it, we may pick up additional practical tips on how to live longer. Avanti!



GREAT BLOG! You are one of the best writers I’ve seen in a long long time. I hope you keep writing because people like you inspire me!
Pingback: Revisions to my dietary supplement firewall regimen | AGING SCIENCES – Anti-Aging Firewalls
Pingback: Protect Telomeres, Telomeres and Aging | Anti Aging Nutrition News
Pingback: History and future directions for this blog – invitation for associate researchers/writers | AGING SCIENCES – Anti-Aging Firewalls
Pingback: Telomerase update –arguments for and against using telomere extender supplements | AGING SCIENCES – Anti-Aging Firewalls