By Vince Giuliano
This blog entry looks at recent research on Andrographis – a herbal substance that is a staple of traditional Chinese, Indian and South Asian medicine. It has been used for centuries in countries like China, India, Thailand and Malaysia for the treatment of sore throat, flu and upper respiratory tract infections and snake bites. Like certain other phytosubstances I have reported on earlier, andrographis has been studied almost only exclusively by Asian scientists and is little known in the Western world except for a few practitioners of herbal medicine and a highly select group of takers of such medicines. As is the case for dashen root (salvia miltiorrhiza bunge) and gambogenic and gambogic acid, other important traditional Chinese medicines, the documented curative powers of andrographis seem to have escaped the attention of Western medical researchers. Asian researchers, on the other hand, are seeking to base new therapies for cancer, asthma, stroke and arthritis on compounds in andrographis and their chemical analogs.
Traditional uses of Andrographis
According to WebMD: “Andrographis is a plant that is native to South Asian countries such as India and Sri Lanka. The leaf and underground stem are used to make medicine. — Andrographis is frequently used for preventing and treating the common cold and flu (influenza). Some people claim andrographis stopped the 1919 flu epidemic in India, although this has not been proven. — Andrographis is also used for a wide assortment of other conditions. It is used for digestive complaints including diarrhea, constipation, intestinal gas, colic, and stomach pain; for liver conditions including an enlarged liver, jaundice, and liver damage due to medications; for infections including leprosy, pneumonia, tuberculosis, gonorrhea, syphilis, malaria, cholera, leptospirosis, rabies, sinusitis, and HIV/AIDS; and for skin conditions including wounds, ulcers and itchiness. — Some people use andrographis for sore throat, coughs, swollen tonsils, bronchitis, and allergies. It is also used for “hardening of the arteries” (atherosclerosis), and prevention of heart disease and diabetes.– Other uses include treatment of snake and insect bites, loss of appetite, kidney problems (pyelonephritis), hemorrhoids, and an inherited condition called familial Mediterranean fever. — Andrographis is also used as an astringent, bacteria killing agent, painkiller, fever reducer, and treatment for worms. — Some Internet vendors offer andrographis products that contain extra amounts of an active ingredient called andrographolide. Some of these products are almost 30% andrographolide. However, be careful; the safety and effectiveness of andrographis preparations with high andrographolide content are unknown.”
Medicine Buddha Dharma Website for image
Other names for Andrographis
Other names depending on culture and location include: “Andrographis Paniculata, Andrographolide, Bhunimba, Bidara, Carmantina, Carmantine, Chiretta, Chirette Verte, Chirreta, Chuan Xin Lian, Chuanxinlian, Chuan Xin Lin, Creat, Échinacée d’Inde, Fa-Tha-Lai-Jone, Fa-Tha-Lai-Jone, Gubak, Herba Andrographitis, Indian Echinacea, Justicia paniculata, Justicie, Kalamegha, Kalmegh, Kalmegha, Kariyat, King of Bitters, Kirta, Mahalita, Nabin Chanvandi, Poogiphalam, Roi des Amers, Sadilata, Sambilata, Shivaphala, Supari, Takila, Vizra Ufar, Yavatikta.(ref)”
Andrographolide
“Andrographolide is a labdanediterpenoid that is the main bioactive component of the medicinal plant Andrographis paniculata.[1] Andrographolide is an extremely bitter substance extracted from the stem and leaves of the andrographis paniculata, which is grown for medicinal purposes in China and India. — Because of its antiinflammatory, immunosuppressant and neuroprotective effects, andrographolide is used experimentally in different areas of research, including Cell signaling, immunomodulation and stroke.[2] (ref)
Andrographis like most other plant-based bioactive substances is quite chemically complex. It contains many other bioactive components in addition to andrographoilide.
The 2011 publication [Chemical constituents from roots of Andrographis paniculata] reports “To investigate the chemical constituents of the roots of Andrographis paniculata, 28 compounds were isolated and identified from the 80% ethanol extract. There are 20 flavonoids: 5, 5′-dihydroxy-7, 8, 2′-trimetroxyflavone (1), 5-hydroxy-7, 8, 2′, 6′-tetramethoxyflavone (2), 5, 3′-dihydroxy-7, 8, 4′-trimethoxyflavone (3), 2′-hydroxy-5, 7, 8-trimethoxyflavone (4), 5-hydroxy-7, 8, 2′, 3′, 4′-pentamethoxyflavone (6), wightin (7), 5, 2′, 6′-trihydroxy-7-methoxyflavone 2′-O-beta-D-glucopyranoside (8), 5, 7, 8, 2′-tetramethoxyflavone (10), 5-hydroxy-7, 8-dimethoxyflavanone (11), 5-hydroxy-7, 8-dimethoxyflavone (12), 5, 2′-dihydroxy-7, 8-dimethoxyflavone (13), 5-hydroxy-7, 8, 2′, 5′-tetramethoxyflavone (14), 5-hydroxy-7, 8, 2′, 3′-tetramethoxyflavone (15), 5-hydroxy-7, 8, 2′-trimethoxyflavone (16), 5, 4′-dihydroxy-7, 8, 2′, 3′-tetramethoxyflavone (17), dihydroneobaicalein (18), andrographidine A (19), andrographidine B (20), andrographidine C (21) and 5, 2′-dihydroxy-7, 8-dimethoxyflavone 2′-O-beta-D-glucopyranoside (22); three diterpenoids: andrograpanin (23), neoandrographolide (24) and andrographolide (25); two phenylpropanoids: trans-cinnamic acid (26) and 4-hydroxy-2-methoxycinnamaldehyde (5); and oleanolic acid (9), beta-sitosterol (27) and beta-daucosterol (28). Compound 1 is a new flavone, compound 4 is a new natural product, compounds 2, 3 and 5 were isolated from the Andrographis genus for the first time and compounds 6-9 were isolated from this plant for the first time.”
Andrographis paniculata contains a number of additional bioactive compounds under study, as reported in the May 2012 publication: Rare noriridoids from the roots of Andrographis paniculata: “The rare noriridoids, Andrographidoids A-E (1-5), along with a known iridoid curvifloruside F (6), were isolated from roots of Andrographis paniculata. All noriridoids were aglycones and 1-4 had (semi-) acetal structures located at C-3 but not at C-1. Their structures were established by a series of 1D and 2D NMR analyses. The antibacterial activity of these iridoids was also assessed using the microtitre plate broth dilution method.”
Andrographolide is a powerful inhibitor of inflammation originated by tumornecrosis factor-α (TNF-α), blocks translocation of NF-kappaB into the nucleus and downregulates the P13K/Akt signaling pathways. These are important themes relevant to many of the actions of andrographis including its anti-cancer activities and therapeutic value for inflammatory diseases..
For example, the November 2011 publicationInhibition of TNF-α-Induced Inflammation by andrographolide via down-regulation of the PI3K/Akt signaling pathwayreports: “Andrographolide (1), an active constituent of Andrographis paniculata, decreased tumor necrosis factor-α (TNF-α)-induced intercellular adhesion molecule-1 (ICAM-1) expression and adhesion of HL-60 cells onto human umbilical vein endothelial cells (HUVEC), which are associated with inflammatory diseases. Moreover, 1 abolished TNF-α-induced Akt phosphorylation. Transfection of an activated Akt1 cDNA vector increased Akt phosphorylation and ICAM-1 expression like TNF-α. In addition, 1 and LY294002 blocked TNF-α-induced IκB-α degradation and nuclear p65 protein accumulation, as well as the DNA-binding activity of NF-κB. Compound 1 exhibits anti-inflammatory properties through the inhibition of TNF-α-induced ICAM-1 expression. The anti-inflammatory activity of 1 may be associated with the inhibition of the PI3K/Akt pathway and downstream target NF-κB activation in HUVEC cells.”
There is significant interest in the anticancer activities of key compounds in andrographis and in basing new anticancer therapies in them. For example, andrographolide inhibits the proliferation of human gliablastoma cells, at least in vitro.
The June 2012 publication Inactivation of PI3K/Akt signaling mediates proliferation inhibition and G2/M phase arrest induced by andrographolide in human glioblastoma cells relates: “AIMS: Andrographolide, a principal diterpenoid lactone isolated from the traditional herbal medicine Andrographis paniculata, has been reported to show anti-tumor activity. Since the high lipid solubility of andrographolide permits it to penetrate the blood-brain barrier and concentrate in the brain, we hypothesized that andrographolide may be a potential chemotherapeutic agent for the treatment of glioblastomas. To clarify this point, we investigated the growth inhibitory effect and mechanisms of actions of andrographolide on human glioblastoma U251 and U87 cells. MAIN METHODS: MTT assay and trypan blue exclusion assay were used to investigate the proliferation inhibitory and cytotoxic effects of andrographolide, respectively. Cell cycle distribution was analyzed using flow cytometry. Apoptosis analysis proceeded by detecting the cleavage of caspase-3. The levels of proteins were probed by Western blotting. KEY FINDINGS: The results showed that non-toxic concentrations of andrographolide inhibited the proliferation of human glioblastoma cells through induction of G2/M arrest, which was accompanied by down-regulating Cdk1 and Cdc25C proteins. Additionally, andrographolide decreased the activity of PI3K/Akt signaling, as demonstrated by down-regulation of the expression of phos-PI3K, phos-Akt, phos-mTOR and phos-p70s6k in U251 and U87 cells. Furthermore, additive effects on the proliferation inhibition, G2/M arrest and down-regulation of G2/M phase-related proteins were observed, when a combined treatment of andrographolide with PI3K inhibitor LY294002 was used in U251 and U87 cells. SIGNIFICANCE: We prove that andrographolide inhibits the proliferation of human glioblastoma cells via inducing G2/M arrest, which is mediated by inhibiting the activity of PI3K/Akt signaling.”
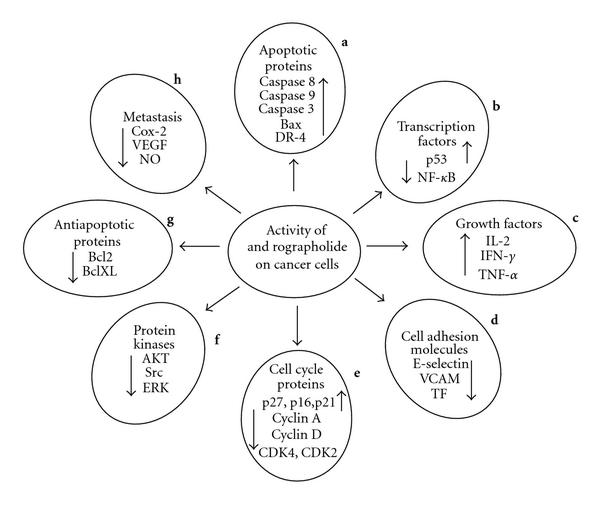
“Figure 2: Effect of andrographolide treatment on cancer cells. Cancer is a multifaceted disease with complex processes and requires a multi-target therapeutic approach to battle it. A similar kind of action is displayed by andrographolide as it modulates various biochemical pathways of cancer cells thereby inhibiting the tumor growth. The compound exerts cytotoxic effect on various cancer cell types in a time and dose dependent manner. Factors required for tumor progression, nourishment and metastasis are down regulated, that is, cyclins A, D, Cdk2, Cdk4, NF-κB, VEGF, E-selectin, VCAM, Akt, TNF, Bcl2, and so forth. On the other hand tumor suppressor elements like p53, caspases, inhibitory proteins p21, p16, p27, and so forth are up regulated as observed in various studies to investigate anti-cancer potential of andrographolide. Up regulation of death receptor 4 to facilitate TRAIL induced apoptosis is of significant interest. The cumulative effect of all these factorial events leads to inhibition of growth in cancer cells.(ref)”
Yet another anti-cancer property of andrographolide is its capability to inhibit angiogenesis in tumors.
The July 2012 publication Andrographolide Inhibits Osteopontin Expression and Breast Tumor Growth Through Down Regulation of PI3 kinase/Akt Signaling Pathwayis one of several recent cell-level and small-animal-level experimental studies on the anticancer capabilities of andrographolide. “Breast cancer is one of the most common cancers among women in India and around the world. Despite recent advancement in the treatment of breast cancer, the results of chemotherapy to date remain unsatisfactory, prompting a need to identify natural agents that could target cancer efficiently with least side effects. Andrographolide (Andro) is one such molecule which has been shown to possess inhibitory effect on cancer cell growth. In this study, Andro, a natural diterpenoid lactone isolated from Andrographis Paniculata has been shown to inhibit breast cancer cell proliferation, migration and arrest cell cycle at G2/M phase and induces apoptosis through caspase independent pathway. Our experimental evidences suggest that Andro attenuates endothelial cell motility and tumor-endothelial cell interaction. Moreover, Andro suppresses breast tumor growth in orthotopic NOD/SCID mice model. The anti-tumor activity of Andro in both in vitro and in vivo model was correlated with down regulation of PI3 kinase/Akt activation and inhibition of pro-angiogenic molecules such as OPN and VEGF expressions. Collectively, these results demonstrate that Andro may act as an effective anti-tumor and anti-angiogenic agent for the treatment of breast cancer.”
Andrographolide can potentiate the effectiveness of the cancer chemotherapy drug cisplatin in inducing apoptosis in cancer cells via suppression of autophagy.
The March 2012 publication Andrographolide sensitizes cisplatin-induced apoptosis via suppression of autophagosome-lysosome fusion in human cancer cells reports: “Suppression of autophagy has been increasingly recognized as a novel cancer therapeutic approach. Andrographolide (Andro), a diterpenoid lactone isolated from an herbal plant Andrographis paniculata, is known to possess antiinflammatory and anticancer activity. In this study, we sought to examine the effect of Andro on autophagy, and to evaluate whether such effect is relevant to the sensitization effect of Andro on apoptosis induced by DNA damage agents in cancer cells. First, we found that Andro is able to significantly enhance autophagic markers in various cancer cell lines, including GFP-LC3 puncta and LC3-II level. Interestingly, Andro treatment also led to marked increase of p62 protein level and addition of chloroquine (CQ) failed to further enhance either LC3-II or p62 level, indicating that Andro is likely to suppress autophagic flux at the maturation and degradation stage. Next, we provided evidence that Andro inhibits autophagosome maturation not by affecting the lysosomal function, but by impairing autophagosome-lysosome fusion. Lastly, we demonstrated that treatment with cisplatin, a DNA damage agent, induces autophagy in cancer cells. Importantly, Andro is capable of sensitizing cisplatin-induced cell killing determined with both short-term apoptosis assays and long-term clonogenic test, via suppression of autophagy, a process independent of p53. In summary, these observations collectively suggest that Andro could be a promising anti-cancer agent in combination therapy via its potent inhibitory effect on autophagy by disrupting autophagosome-lysosome fusion.”
Studies are being conducted relating key compounds in andrographis to the gene-activation pathways they affect in types of cancer cells. Some compounds works to kill cancer cells by inhibiting the cell cycle or promoting cell cycle arrest.
The February 2012 publicationIdentification of genes involved in the regulation of 14-deoxy-11,12-didehydroandrographolide-induced toxicity in T-47D mammary cells reports: “14-Deoxy-11,12-didehydroandrographolide is one of the principle compounds of the medicinal plant, Andrographis paniculata Nees. This study explored the mechanisms of 14-deoxy-11,12-didehydroandrographolide-induced toxicity and non-apoptotic cell death in T-47D breast carcinoma cells. Gene expression analysis revealed that 14-deoxy-11,12-didehydroandrographolide exerted its cytotoxic effects by regulating genes that inhibit the cell cycle or promote cell cycle arrest. This compound regulated genes that are known to reduce/inhibit cell proliferation, induce growth arrest and suppress cell growth. The growth suppression activities of this compound were demonstrated by a downregulation of several genes normally found to be over-expressed in cancers. Microscopic analysis revealed positive monodansylcadaverine (MDC) staining at 8h, indicating possible autophagosomes. TEM analysis revealed that the treated cells were highly vacuolated, thereby suggesting that 14-deoxy-11,12-didehydroandrographolide may cause autophagic morphology in these cells. This morphology may be correlated with the concurrent expression of genes known to affect lysosomal activity, ion transport, protein degradation and vesicle transport. Interestingly, some apoptotic-like bodies were found, and these bodies contained multiple large vacuoles, suggesting that this compound is capable of eliciting a combination of apoptotic and autophagic-like morphological characteristics.”
Andrographolide inhibits release of NF-kappaB from its binding factor IκB-α and its translocation into the nucleus and activation, and consequentially inhibits release of matrix metalloproteinase-9. It may thus provide the basis for new therapies for inflammatory processes and certain cancers including leukemia.
The February 2012 e-publication Suppression of matrix metalloproteinase-9 expression by andrographolide in human monocytic THP-1 cells via inhibition of NF-κB activation reports: “There is much evidence indicating that human leukemic cells and monocytes/macrophages synthesize, and secrete, several matrix metalloproteinases (MMPs), and participate in the degradation of extracellular matrix components in tissue lesions. In this study, we investigated the effects and mechanisms of andrographolide, extracted from the herb Andrographis paniculata, on human monocytic MMPs expression and activation. Andrographolide (1-50 μM) exhibited concentration-dependent inhibition of MMP-9 activation, induced by either tumor necrosis factor-α (TNF-α), or lipopolysaccharide (LPS), in THP-1cells. In addition, andrographolide did not present an inhibitory effect on MMP-9 enzymatic activity at a concentration of 50 μM. By contrast, enzyme-linked immunosorbent assay (ELISA) showed that andrographolide partially affect TIMP-1 levels. Western blot analysis showed that both TNF-α, and LPS stimulators attenuated MMP-9 protein expression in a concentration-dependent manner. Using reverse transcription polymerase chain reaction (RT-PCR), we found that andrographolide suppressed expression of MMP-9 messenger RNA. Furthermore, we also found that andrographolide could significantly inhibit the degradation of inhibitor-κB-α (IκB-α) induced by TNF-α. We used electrophoretic mobility shift assay and reporter gene detection to show that andrographolide also markedly inhibited NF-κB signaling, anti-translocation and anti-activation. In conclusion, we demonstrate that andrographolide attenuates MMP-9 expression, and its main mechanism might involve the NF-κB signal pathway. These results provide new opportunities for the development of new anti-inflammatory and leukemic therapies.”
The October 2011 publication Andrographolide inhibits oral squamous cell carcinogenesis through NF-κB inactivation again highlights how inhibition of NF-kappaB is important for andrographolide’s capability to combat cancer. It reports: “The NF-κB family of transcription factors is essential for promoting cell proliferation and preventing cell apoptosis. We have previously shown that Andrographolide (Andro) isolated from an herbal plant, Andrographis paniculata, covalently modifies reduced cysteine(62) in the oligonucleotide binding pocket of p50 for inhibition of NF-κB activation. Here we report that Andro, but not its inactive structural analog 4H-Andro, potently suppressed squamous cell carcinogenesis induced by 7,12-dimethyl-1,2-benzanthracene (DMBA) in the hamster model of cheek buccal pouch. Compared with 4H-Andro, Andro reduced phosphorylation of p65 (Ser536) and IκBα (Ser32/36) for inhibiting aberrant NF-κB activation, suppressed c-Myc and cyclin D1 expression and attenuated neoplastic cell proliferation, promoted cancerous cell apoptosis, and mitigated tumor-induced angiogenesis. Consistently, Andro retarded growth, decreased proliferation, and promoted apoptosis of Tb cells, a human tongue squamous cell carcinoma cell line, in time- and dose-dependent manners, with concomitant reduction of the expression of NF-κB targeting molecules in vitro. Our results thus demonstrate that NF-κB activation plays important roles in the pathogenesis of chemically induced squamous cell carcinoma. By inhibition of aberrant NF-κB activation, Andro treats chemically induced oral squamous cell carcinogenesis.”
Andrographolide suppresses activation of Nf-kappaB, inhibits the P38/MAPK pathway, activates Nrf2 and upregulates expression of heme oxygenase. Implications include therapeutic potential for treating coronary heart diseases as well as cancers and multiple other diseases involving runaway inflammatory processes.
The July 2012 publicationSuppression of NF-κB signaling by andrographolide with a novel mechanism in human platelets: regulatory roles of the p38 MAPK-hydroxyl radical-ERK2 cascadereports: “Andrographolide, a novel nuclear factor-κB (NF-κB) inhibitor, is isolated from leaves of Andrographis paniculata. Platelet activation is relevant to a variety of coronary heart diseases. Our recent studies revealed that andrographolide possesses potent antiplatelet activity by activating the endothelial nitric oxide synthase (eNOS)-NO-cyclic GMP pathway. Although platelets are anucleated cells, they also express the transcription factor, NF-κB, that may exert non-genomic functions in platelet activation. Therefore, we further investigated the inhibitory roles of andrographolide in NF-κB-mediated events in platelets. In this study, NF-κB signaling events, including IKKβ phosphorylation, IκBα degradation, and p65 phosphorylation, were time-dependently activated by collagen in human platelets, and these signaling events were attenuated by andrographolide (35 and 75μM). ODQ and KT5823, respective inhibitors of guanylate cyclase and cyclic GMP-dependent kinase (PKG), strongly reversed andrographolide-mediated inhibition of platelet aggregation, relative [Ca(2+)]i mobilization, and IKKβ, and p65 phosphorylation. In addition, SB203580 (an inhibitor of p38 MAPK), but not PD98059 (an inhibitor of ERKs), markedly abolished IKKβ and p65 phosphorylation. SB203580, NAC (a free-radical scavenger), and BAY11-7082 (an inhibitor of NF-κB) all diminished ERK2 phosphorylation, whereas PD98059, BAY11-7082, and NAC had no effects on p38 MAPK phosphorylation. Furthermore, SB203580, but not BAY11-7082 or PD98059, reduced collagen-induced hydroxyl radical ((●)HO) formation. KT5823 also markedly reversed andrographolide-mediated inhibition of p38 MAPK and ERK2 phosphorylation, and hydroxyl radical formation in platelets. In conclusion, this study demonstrated that andrographolide may involve an increase in cyclic GMP/PKG, followed by inhibition of the p38 MAPK/(●)HO-NF-κB-ERK2 cascade in activated platelets. Therefore, andrographolide may have a high therapeutic potential to treat thromboembolic disorders and may also be considered for treating various inflammatory diseases.”
Based on its ability to inhibit the impact of tumor necrosis factor α on activation of NF-kappaB and upgrade expression of Nrf2 and heme oxygenase, andrographolide is protective against liver tissue injuries created by cytotoxins such as carbon tetrachloride or paracetamol.
The 2011 publication Protective mechanism of andrographolide against carbon tetrachloride-induced acute liver injury in mice reports: “The aim of this study was to investigate the protective effects of andrographolide (AP), a bioactive component isolated from Andrographis paniculata, on carbon tetrachloride (CCl(4))-induced liver injury as well as the possible mechanisms involved in this protection in mice. Acute liver injury was induced by CCl(4) intoxication in mice. Serum biological analysis, lipid peroxides and antioxidant estimation, histopathological studies, reverse transcription polymerase chain reaction (RT-PCR) and Western blot assay were carried out. CCl(4) treatment resulted in severe hepatic injury, as evidenced by significant elevation of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels and typical histopathological changes, such as hepatocyte necrosis. Additionally, CCl(4) administration led to oxidative stress in mice, as indicated by a remarkable increase in the hepatic malondialdehyde (MDA) level, together with a significant decrease in liver reduced glutathione (GSH) content. However, CCl(4)-induced hepatotoxicity was significantly attenuated by pretreatment with AP, as demonstrated by significant reduction of serum ALT, AST levels and hepatic MDA activity, along with a remarkable increase in hepatic GSH content. Histopathological changes induced by CCl(4) were also ameliorated by AP pretreatment. The marked increase of – tumor necrosis factor α (TNF-α) induced by CCl(4) was attenuated by AP, and the dramatic elevation of heme oxygenase-1 (HO-1) at transcriptional and protein levels was augmented following AP pretreatment. AP can effectively prevent liver injury induced by CCl(4), which may be due to inhibition of oxidative stress and inflammatory responses.”
The December 2011 publication Hepatoprotective activity of Andrographis paniculata and Swertia chirayita reported: “ — Oral administration of A. paniculata or S. chirayita extract (100-200mg/kg) offered a significant dose dependent protection against paracetamol induced hepatotoxicity as assessed in terms of biochemical and histopathological parameters. The paracetamol induced elevated levels of serum marker enzymes such as serum glutamate pyruvate transaminase (GPT), serum glutamate oxaloacetate transaminase (GOT), alkaline phosphatase (ALP), and bilirubin in peripheral blood serum and distorted hepatic tissue architecture along with increased levels of lipid peroxides (LPO) and reduction of superoxide dismutase (SOD), catalase, reduced glutathione (GSH) and glutathione peroxidase (GPx) in liver tissue. Administration of the plant extracts after paracetamol insult restored the levels of these parameters to control (untreated) levels. Thus the present study revealed that the extracts of A. paniculata or S. chirayita offered protection against hepatotoxicity induced by paracetamol.”
Andrographolide appears to be gastroprotective; it is protective against certain duodenal ulcers in rats.
The September 2011 publication Effect of andrographolide on cysteamine-induced duodenal ulcer in rats reports: “The aim of this study was to evaluate the gastroprotective efficacy of andrographolide isolated from Andrographis paniculata in rats induced with duodenal ulcers. Duodenal ulcers were induced by cysteamine administration in rats pretreated with 3 mg kg⁻¹ BW day⁻¹ of andrographolide for 30 days. Ulcer score, myeloperoxidase activity, TBARS level, GSH/GSSG ratio and enzyme antioxidants were measured in the duodenal tissue. Brush border and basolateral membranes were isolated to assay sucrase, maltase, alkaline phosphatase and total ATPases. Ulcer score was significantly minimised in rats pretreated with andrographolide. Elevation in myeloperoxidase and TBARS levels were found to be minimised significantly due to andrographolide treatment. Membrane-bound enzyme activities and the thiol redox status of glutathione were significantly maintained in duodenal mucosa of rats that received andrographolide. This study reveals that the major component of A. paniculata, andrographolide, has potent antiulcer properties that are most likely caused by minimising inflammatory changes, counteracting free radical formation and maintaining the thiol redox status in the duodenum.”
The July 2012 publication A biochemical study on the gastroprotective effect of hydroalcoholic extract of Andrographis paniculata in rats speaks to the same point: “Aim: The aim of the present study is to evaluate the gastroprotective effect of hydroalcoholic extract of Andrographis paniculata (HAEAP) in male albino wistar rats. Materials and Methods: Rats were pretreated with HAEAP (100,200,500mg/kg b. wt for 30 days) and then gastric ulcers were induced by ethanol, aspirin, pylorus ligation and cold restraint stress models. Ulcer score was determined in all the ulcer models. pH, gastric volume, titrable acidity, pepsin, mucin, myeloperoxidase, H+K+ATPase, thiobarbituric acid reacting substances (TBARS) and antioxidant enzyme activities were assayed in ethanol-administered rats. Results: The ulcer score was found to be low in HAEAP-pretreated rats. Among the doses studied, 200 mg/kg b.wt was found to be optimum for significant ulcer reduction. The test drug significantly reduced the acidity, pepsin concentration, myeloperoxidase and H+K+ATPase activities in ethanol-administered rats. The elevated TBARS and decreased glutathione (GSH) and mucin levels observed during ulcerogenesis were found to be altered in HAEAP-received animals. Conclusions: The ulcer preventing effect of HAEAP may partly be due to its regulating effect on H+K+ATPase activity and /or mucin preserving effects. The flavonoids present in the HAEAP might be responsible for the gastroprotective action probably by maintaining the antioxidants and thiol status in the gastrointestinal tract.”
Again relating to the antiulcer capabilities of andrographis is the July 2011 publication Antiulcer activity of Andrographis paniculata (Burm.f.) wall. against cysteamine-induced duodenal ulcer in rats.
Components of andrographis interact with at least one member of the cytochrome P450 family implying that consumption of andrographis may generate a risk of interactions with certain drugs or an enhanced risk of lung cancer in the case of smokers.
The December 2011 publication Different AhR binding sites of diterpenoid ligands from Andrographis paniculata caused differential CYP1A1 induction in primary culture in mouse hepatocytes reports “Andrographis paniculata has been employed as a folklore remedy. Andrographolide (Andro), 14-deoxy-11,12-didehydroandrographolide (DHA), andrographiside (AS), and neoandrographolide (Neo), are major diterpenoids isolated from this plant. In the present study, influence of the four diterpenoids on CYP1A1 mRNA expression was investigated in primary cultured mouse hepatocytes. Additionally, binding of these compounds to aryl hydrocarbon receptor (AhR) was examined using molecular docking analysis to clarify mechanism of CYP1A1 induction. Andro and DHA induced CYP1A1 expression by itself, and co-treatment with a CYP1A1 inducer (BNF, beta-naphthoflavone) showed a synergistic increase of CYP1A1 expression. Andro demonstrated higher enhancing activity than DHA at every similar concentration. On the other hand, Neo suppressed BNF-induced CYP1A1 expression, but AS did not modify the induction. Results from molecular docking analysis of BNF and four diterpenoids on ligand binding domain of AhR were consistent with levels of CYP1A1 mRNA expressions. Furthermore, difference of binding sites of BNF in the presence of diterpenoids might affect the synergism or inhibition of CYP1A1 expression. These results suggest that use of A. paniculata as a health supplement should be concerned in term of herb-drugs interactions or risk of carcinogenesis, according to its ability to influence CYP1A1 expression.”
Regarding CPY1A1: “This gene, “CYP1A1, encodes a member of the cytochrome P450 superfamily of enzymes. The cytochrome P450 proteins are monooxygenases which catalyze many reactions involved in drug metabolism and synthesis of cholesterol, steroids and other lipids. This protein localizes to the endoplasmic reticulum and its expression is induced by some polycyclic aromatic hydrocarbons (PAHs), some of which are found in cigarette smoke. The enzyme’s endogenous substrate is unknown; however, it is able to metabolize some PAHs to carcinogenic intermediates. The gene has been associated with lung cancer risk(ref).”
Effectiveness of andrographolide as a treatment is compromised by its poor oral bioavailability.
The November 2011 publication Poor oral bioavailability of a promising anticancer agent andrographolide is due to extensive metabolism and efflux by P-glycoprotein reports: “Andrographolide (AP), isolated from Andrographis paniculata (Burm. F.) Nees, is an anticancer agent with significant clinical potential. This study determined its oral bioavailability and how intestinal disposition affects its bioavailability. Pharmacokinetics was evaluated in rats. Intestinal disposition was determined using a single-pass rat intestinal perfusion model and the cultured Caco-2 cells and Madin-Darby canine kidney II cells over expressing human P-gp (MDR1-MDCKII). Absolute bioavailability of AP was 2.67%. In the duodenum and jejunum, AP was rapidly metabolized to a sulfonate, identified as 14-deoxy-12-sulfo- andrographolide. AP was also rapidly metabolized by liver S9 fraction and in blank perfusates collected from duodenum and jejunum. The apparent permeability (P(app) ) of AP from basolateral (B) to apical (A) (4.94 × 10 cm/s) in the Caco-2 model was four times higher than the P(app) from A to B (1.14 × 10(-5) cm/s). Moreover, AP was significantly more permeable in the B to A direction than the opposite direction in MDR1-MDCKII cells. In the perfusion model, the effective permeability (P*(eff) ) for AP was highest in the duodenum, followed by jejunum, and then ileum and colon. In the ileum and colon, the P*(eff) for AP was significantly increased by verapamil, a P-glycoprotein (P-gp) inhibitor. AP has poor oral bioavailability because of its rapid biotransformation and efflux by P-gp.”
A less-cytotoxic analog of Andrographolide may provide the basis for a new therapy for asthma.
The June 2011 publication Protective role of 14-deoxy-11,12-didehydroandrographolide, a noncytotoxic analogue of andrographolide, in allergic airway inflammationreports: “Our group recently reported novel anti-inflammatory effects of andrographolide (2), a bioactive molecule isolated from Andrographis paniculata, in a mouse asthma model. However, 2 has been shown to possess cytotoxic activity. 14-Deoxy-11,12-didehydroandrographolide (1) is an analogue of 2 that can be isolated from A. paniculata. We hypothesized that 1 retains the anti-inflammatory effects for asthma but is devoid of cytotoxicity. In contrast to 2, 1 did not elicit any cytotoxic activity in A549 and BEAS-2B human lung epithelial cells and rat basophilic leukemia (RBL)-2H3 cells using a MTS assay. Compound 1 dose-dependently inhibited ovalbumin (OVA)-induced increases in total and eosinophil counts, IL-4, IL-5, and IL-13 levels in lavage fluid, and serum OVA-specific IgE level in a mouse asthma model. Compound 1 attenuated OVA-induced airway eosinophilia, mucus production, mast cell degranulation, pro-inflammatory biomarker expression in lung tissues, and airway hyper-responsiveness. This substance also blocked p65 nuclear translocation and DNA-binding activity in the OVA-challenged lung and in TNF-α-stimulated human lung epithelial cells. The present findings reveal for the first time that 1 retains the anti-inflammatory activities of 2 for asthma probably through the inhibition of NF-κB. 14-Deoxy-11,12-didehydroandrographolide (1) may be considered as a safer analogue of 2 for the potential treatment of asthma.”
Topical application of andrographis appears to accelerate wound healing, at least in rats.
The June 2012 publication Effect of Andrographis paniculata leaf extract on wound healing in rats reports: “This work was carried out to study the effect of topical application of Andrographis paniculata on the rate of wound enclosure and its histological features. A wound was created in four groups of rat in posterior neck region. Blank placebo was applied topically to the wounds of Group 1. Groups 2 and 3 were dressed with placebo containing 5% and 10% extracts of A. paniculata, respectively. Intrasite gel was applied topically to the wounds of Group 4. Macroscopical examination revealed that the rate of wound healing was significantly accelerated in the wound dressed with A. paniculata extract compared to the blank placebo. The wounds dressed with 10% extract or Intrasite gel healed earlier compared to the wounds dressed with placebo containing 5% A. paniculata extract. Histologically, wounds dressed with A. paniculata extracts showed markedly less scar width and contained large amounts of fibroblast proliferation. More collagen and less angiogenesis with absence of inflammatory cells were seen for wounds dressed with 10% A. paniculata compared to the blank placebo. Conclusion, A. paniculata extracts significantly enhanced rate of wound healing in rats.”
A combined extract of andrographis and danshen together with swimming in a rat model appears to be effective for prevention and treatment of atherosclerosis.
In December 2011 I wrote a comprehensive blog entry on danshen, another important traditional Chinese herbal medicine Focus on phytosubstances – Danshen root – amazing properties of salvia miltiorrhiza Bunge.
The 2011 publicationJoint preventive effects of swimming and Shenlian extract on rat atherosclerosis reports: “This work is to investigate the joint effect of extract from Shenlian (SL, the Chinese abbreviation for Radix Salviae miltiorrhizae and Andrographis paniculata) and swimming on atherosclerosis prevention and treatment. Atherosclerotic rat model was established by combining low shear stress by partial ligation of common carotid artery with afterwards feeding of a hyperlipotic diet. Sprague-Dawley rats after partial ligation of common carotid artery were allotted to a 3 × 3 factorial design with three levels of swimming (0, 1, and 2 hr per day) and three levels of SL extract (0, 2.56, and 5.12 g/kg once daily p.o.) for a total of 9 treatment groups. Then, the feeding of the hyperlipotic diet and the intervention of swimming and SL started at the same time, and lasted for 8 weeks. By the end, blood samples were collected to determine blood viscosity, hematocrit, blood lipids, MCP-1, NF-κB and NO levels. The common carotid arteries of the rats were harvested to investigate pathological changes. The animal model showed early sign of atherosclerosis according to the pathological findings. Joint effects of SL extract and swimming on preventing atherosclerosis appeared significantly: The combination of 1 hour swimming with 2.56 g/kg SL extract showed to be effective for lowering hematocrit, blood viscosity (at 10 s(-1) and 200 s(-1)) and low-density-lipoprotein (p < 0.001). Combined treatment of 2.56 g/kg SL extract with 2 hr swimming led to a synergistic decrease in serum level of MCP-1. As a single factor, SL extract (2.56 g/kg) alone could decrease serum levels of NF-κB significantly (p = 0.003). Swimming alone could decrease cholesterol, triacylglycerols level and increase high-density-lipoprotein. The study demonstrates the combined therapy of oral SL extract with swimming on inhibiting inflammatory factors, improving hemorheological parameters and lipoproteins in rat model of atherosclerosis. It highlights the promising effects of the drugs and shear stress of blood flow, the biomechenopharmacological means, for prevention of atherosclerosis.”
There is considerable activity in developing specialized analogs of Andrographolide that are either more potent in killing cancer cells, less cytotoxic to normal tissues, or have enhanced bioavailability.
The January 2012 publication New substituted C-19-andrographolide analogues with potent cytotoxic activities reports: “Andrographolide, the major diterpenoid lactone from Andrographis paniculata, is toxic against cancer cells. In the present study, we investigated the structure-activity relationships (SARs) of 19 andrographolide analogues which were synthesized by modification at the three hydroxyl groups. A number of the andrographolide analogues showed much higher cytotoxic activities than that of the parent compound on cancer cells including P-388, KB, COL-2, MCF-7, LU-1 and ASK cells. SAR studies of the synthetic analogues indicated that the introduction of silyl ether or triphenylmethyl ether group into C-19 of the parent compound led to increase in toxicity against the cancer cells. The 19-O-triphenylmethyl ether analogue 18 showed higher cytotoxic activity than the potent anticancer drug ellipticine, and this analogue may serve as a potential structure lead for the development of new anticancer drugs.”
The March 2012 publication Andrographolide and its analogues: versatile bioactive molecules for combating inflammation and cancer reports: “1. Andrographis paniculata (Burm. f) Nees, commonly known as ‘king of bitters’, is a herbaceous plant belonging to the Family Acanthaceae. It has been widely used for centuries in Asian countries like China, India, Thailand and Malaysia for the treatment of sore throat, flu and upper respiratory tract infections. 2. Andrographolide, 14-deoxy-11,12-didehydroandrographolide and neoandrographolide are examples of the major labdane diterpenoids isolated from A. paniculata. These bioactive molecules have exhibited varying degrees of anti-inflammatory and anticancer activities in both in vitro and in vivo experimental models of inflammation and cancer. 3. Extensive libraries of andrographolide analogues have been synthesised mainly by modifying the α,β-unsaturated γ-butyrolactone moiety, the two double bonds Δ(8,(17)) and Δ(12,(13)) and the three hydroxyls at C-3 (secondary), C-14 (allylic) and C-19 (primary). Many of these synthetic analogues exhibit superior anticancer activity over the naturally occurring andrographolides. 4. Andrographolide and its derivatives have been shown to have anti-inflammatory effects in experimental models of asthma, stroke and arthritis, as well as in patients with upper respiratory tract infections. Andrographolide reduces the production of cytokines, chemokines, adhesion molecules, nitric oxide and lipid mediators, probably via inhibition of the nuclear factor (NF)-κB signalling pathway. 5. The anticancer mechanisms for andrographolide include inhibition of Janus tyrosine kinases-signal transducers and activators of transcription, phosphatidylinositol 3-kinase and NF-κB signalling pathways, suppression of heat shock protein 90, cyclins and cyclin-dependent kinases, metalloproteinases and growth factors, and the induction of tumour suppressor proteins p53 and p21, leading to inhibition of cancer cell proliferation, survival, metastasis and angiogenesis. 6. Andrographolide drug discovery is a promising strategy for the development of a novel class of anti-inflammatory and anticancer drugs.”
Andrographis can provide significant protection against gastric ulcers.
The November 2011 publication Antioxidant and gastroprotective activities of Andrographis paniculata (Hempedu Bumi) in Sprague Dawley rats reports: “Antioxidant and gastroprotective activities of aqueous and ethanolic extract of Andrographis paniculata leaves in rats have been reported. Sprague Dawley rats, 6 per group were used and rats in groups 1 to 6 were pretreated with (0.25% w/v) carboxymethyl cellulose (negative control, 5 ml/kg), 20 mg/kg omeprazole (positive control), (250 mg/kg and 500 mg/kg) of aqueous leaf extracts (APLAE) and (250 and 500 mg/kg) of ethanol leaf extracts (APLEE) respectively. Animals were orally administered with 95% ethanol (5 ml/kg) 60 min after their pretreatments. Rats were sacrificed 1 h after treatment and gastric contents were collected to measure pH and mucous weight. Stomach was analyzed for gross and histological changes. Ulcer control group showed extensive lesions of gastric mucosal layer, whereas rats pretreated with omeprazole, 250 and 500 mg/kg of APLAE showed significant and dose dependent reduction in gastric lesions with increased pH and mucus content of stomach. Rats pretreated with 250 or 500 mg/kg of APLEE showed significantly better inhibition of gastric mucosal lesions. Further, the in vitro antioxidant studies using 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay showed that ethanol extracts have superior free radical scavenging activity with IC50 value = 10.9 than aqueous extracts with IC50 value = 24.65. Results of this study showed that pretreatment with ethonolic extract of A. paniculata ethanolic provided significant protection against gastric ulcer by regulating of pH, mucous production and antioxidant property.”
Andrographolide may be useful for the treatment of rheumatoid arthritis.
The February 2012 publication Andrographolide induces cell cycle arrest and apoptosis in human rheumatoid arthritis fibroblast-like synoviocytes reports: “The pseudo-tumoral expansion of fibroblast-like synoviocytes is a hallmark of rheumatoid arthritis (RA), and targeting rheumatoid arthritis fibroblast-like synoviocytes (RAFLSs) may have therapeutic potentials in this disease. Andrographolide, a diterpenoid compound isolated from the herb Andrographis paniculata, has been reported to have potent anti-inflammatory activity. In the present study, we aimed to investigate the effects of andrographolide on human RAFLSs and the underlying molecular mechanism(s). RAFLSs were isolated from patients with RA and treated with or without various concentrations (i.e., 10, 20, and 30 μM) of andrographolide for 48 h. 3-[4,5-Dimethyl-2-yl]-2,5-diphenyl tetrazolium bromide assay revealed that andrographolide treatment decreased the proliferation of RAFLSs in a dose-dependent manner. Cell cycle analysis using propidium iodide (PI) staining showed a G0/G1 cell cycle arrest in andrographolide-treated RAFLSs. Immunoblotting analysis of key cell cycle regulators demonstrated that andrographolide treatment caused a dose-dependent increase in the expression of cell-cycle inhibitors p21 and p27 and a concomitant reduction of cyclin-dependent kinase 4. Exposure to andrographolide-induced apoptosis of RAFLSs measured by annexin V/PI double staining, which was coupled with promotion of cytochrome C release from mitochondria and activation of caspase-3. Moreover, andrographolide-treated RAFLSs displayed a significant decrease in the Bcl-2/Bax ratio compared to untreated cells. In conclusion, our data demonstrate that andrographolide exerts anti-growth and pro-apoptotic effects on RAFLSs, thus may have therapeutic potential for the treatment of RA.”
Andrographolide may be the basis for new generations of mosquito killing and repellant agents, possibly providing an important new defense against malaria.
I know it may seem ridiculous to add this claim to the many others related to andrographis. However the May 2912 publication Adulticidal and repellent properties of indigenous plant extracts against Culex quinquefasciatus and Aedes aegypti (Diptera: Culicidae) reports: “Several diseases are associated to the mosquito-human interaction. Mosquitoes are the carriers of severe and well-known illnesses such as malaria, arboviral encephalitis, dengue fever, chikunguniya fever, West Nile virus, and yellow fever. These diseases produce significant morbidity and mortality in humans and livestock around the world. The adulticidal and repellent activities of crude hexane, ethyl acetate, benzene, chloroform, and methanol extracts of leaf of Eclipta alba and Andrographis paniculata were assayed for their toxicity against two important vector mosquitoes, viz., Culex quinquefasciatus and Aedes aegypti (Diptera: Culicidae). The adult mortality was observed after 24 h of exposure. All extracts showed moderate adulticide effects; however, the highest adult mortality was found in methanol extract of A. paniculata against the adults of C. quinquefasciatus and A. aegypti with the LC(50) and LC(90) values were 149.81, 172.37 ppm and 288.12, 321.01 ppm, respectively. The results of the repellent activity of hexane, ethyl acetate, benzene, chloroform, and methanol extract of E. alba and A. paniculata plants at three different concentrations of 1.0, 2.5, and 5.0 mg/cm(2) were applied on skin of forearm in man and exposed against adult female mosquitoes. In this observation, these two plant crude extracts gave protection against mosquito bites without any allergic reaction to the test person, and also, the repellent activity is dependent on the strength of the plant extracts. These results suggest that the leaf solvent plant extracts have the potential to be used as an ideal ecofriendly approach for the control of mosquitoes. This is the first report on the mosquito adulticidal and repellent activities of the reported E. alba and A. paniculata plants.”
The September 2011 report Efficacy of indigenous plant extracts on the malaria vector Anopheles subpictus Grassi (Diptera: Culicidae)concludes similarly: “BACKGROUND & OBJECTIVES: Mosquito control is facing a threat due to the emergence of resistance to synthetic insecticides. Insecticides of plant origin may serve as suitable alternative biocontrol techniques in the future. The purpose of the present study was to assess the ethyl acetate, acetone and methanol extracts of Andrographis paniculata, Eclipta prostrata and Tagetes erecta leaves tested for oviposition-deterrent, ovicidal and repellent activities against malaria vector, Anopheles subpictus Grassi (Diptera: Culicidae). — CONCLUSIONS: The acetone extract of A. paniculata, methanol extract of E. prostrata and T. erecta showed good oviposition-deterrent, ovicidal and repellent activities respectively. These results suggest that the leaf extracts of A. paniculata, E. prostrata and T. erecta may have the potential to be used as an ideal eco-friendly approach for the control of the An. subpictus.”
The March 2011 publication Evaluation of Andrographis paniculata Burm.f. (Family:Acanthaceae) extracts against Culex quinquefasciatus (Say.) and Aedes aegypti (Linn.) (Diptera:Culicidae) speaks again to the same point regarding mosquitos: “CONCLUSIONS: From the results it can be concluded the crude extract of A. paniculata was a potential for controlling Cx. quinquefasciatus and Ae. aegypti mosquitoes.”
Besides possibly providing a powerful agent against malaria-bearing mosquitoes, Andrographolide combined with curcumin might also provide the basis for a powerful anti-malaria therapy for those infected with Plasmodium malaria parasites.
The June 2011 publication Andrographolide: A Novel Antimalarial Diterpene Lactone Compound from Andrographis paniculata and Its Interaction with Curcumin and Artesunate reports: “Andrographolide (AND), the diterpene lactone compound, was purified by HPLC from the methanolic fraction of the plant Andrographis paniculata. The compound was found to have potent antiplasmodial activity when tested in isolation and in combination with curcumin and artesunate against the erythrocytic stages of Plasmodium falciparum in vitro and Plasmodium berghei ANKA in vivo. IC50s for artesunate (AS), andrographolide (AND), and curcumin (CUR) were found to be 0.05, 9.1 and 17.4μM, respectively. The compound (AND) was found synergistic with curcumin (CUR) and addictively interactive with artesunate (AS). In vivo, andrographolide-curcumin exhibited better antimalarial activity, not only by reducing parasitemia (29%), compared to the control (81%), but also by extending the life span by 2-3 folds. Being nontoxic to the in vivo system this agent can be used as template molecule for designing new derivatives with improved antimalarial properties.”
Andrographis can partially neutralize poisonous snake and scorpion venom.
See the 2011 publications Anti-cobra venom activity of plant Andrographis paniculata and its comparison with polyvalent anti-snake venom,Partial in vitro and in vivo red scorpion venom neutralization activity of Andrographis paniculataand Antihaemolytic and snake venom neutralizing effect of some Indian medicinal plants.
Comments
This blog could cite only a representative sample of the current literature coming out of Asia on andrographis and its constituent compounds. A Pubmed search on andrographis reveals 423 research publications, a significant portion of which written in the last 3-4 years.
Andrographis appears to be a complex substance with a remarkably large number of both traditional and potential new medical and health applications ranging from treatments of cancers to treatments of toxic liver conditions, rheumatoid arthritis, malaria, and gastric ulcers. It wards off and kills mosquitos and is helpful in cases of venomous snakebites and scorpion stings.
The themes of inhibition of NF-kappaB and activation of Nrf2 emphasized in this blog entry as health-producing strategies are not at all new to my writings. In my online treatise ANTI-AGING FIREWALLS – THE SCIENCE AND TECHNOLOGY OF LONGEVITYand in previous blog entries I have discussed how inhibition of NF-kappaB and activation of Nrf2 seem to go hand-in-hand in providing a variety of health and possibly anti-aging effects. Further I have discussed how a variety of phytochemicals that are traditional medicines, like curcumin and ginger, work to do this. Andrographis seems to be another good example of such a phytochemical. See the blog entry The pivotal role of Nrf2. Part 2 – foods, phyto-substances and other substances that turn on Nrf2.
I am left with a number of disquieting questions. With particular reference to andrographis:
- Given that:
- it appears the Asian scientists who have investigated andrographis and its compounds have drawn on the current research, laboratory tools, theories and thought processes of Western biological and medical sciences and have arrived responsibly at their findings. It appears they are highly educated in current Western biological sciences and know what they are doing, and given
- Asian scientists have repeatedly mentioned the great potential for developing medical therapies based on andrographis and its constituent compounds for cancers and a number of other serious disease conditions of aging,
- Why then:
- Does it appear that Western scientists and especially those in drug companies have paid no attention to andrographis and its compounds? These substances just don’t appear to show on the radar screens of Western biological and medical research institutions or in any of the publications emanating from them.
- Why have big Western pharma companies not seized on the possibility of developing proprietary analogs of andrographis constituent compounds for particular medical conditions – at this time when traditional drug discovery approaches are running dry. (Only one clinical trial has been listed for andrographis or andrographolide, and that was completed in 2008.)
The identical questions could be asked about two other important traditional Asian medical substances I have reported on previously dashen root and gambogenic and gambogic acid. And the same questions probably can be asked about a number of other traditional folk medicines that have been recently subject to Asian scientific scrutiny.
Am I missing something or is there a significant gap in East-West medical scientific research? Information written in English about Asian herbal research is readily available to us in the West. Why do Western researchers appear to pay no attention to it? What is it that leads us in the West to keep ignoring Asian herbal research while such ignorance does not exist in Asia regarding what we are doing in our labs here in the US? Does the problem exist because of how our research and how Asian research are funded, because of the research review process where more of the same is the best way to get funded, or because of the nature on academic research institutions?
Further, why are we here in the West so preoccupied with largely unsuccessful attempts to find magic-bullet kinase inhibitors to cure diseases, simple substances that work on single well-defined pathways [See Victor’s blog entry Kinase Inhibition – A Magic Bullet?], while many Asian researchers are working with impossibly complex natural substances like andrographis long-known to be effective, substances that exercise multiple actions on multiple pathways? Is the issue simply a philosophical one of Western reductionism vs. Asian holism?



Two quick thoughts on why “simple” single target drugs are preferred in Western science and medicine.
With respect to science, we have a paradigm of systems and subsystems — and we are trying to understand the primitive steps or elements that constitute each subsystem. An agent that changes one specific thing and nothing else is thus highly desirable in this type of “analysis”
With respect to medicine, the cost of developing a new drug is enormous (estimates of $1B) due in part to regulatory and testing costs. Also, the litigation costs are enormous (probably many multiples of $1B) for drugs that are marketed and then produce unacceptable side effects. So, the drug companies go for drugs that are “simple” and have as limited a range of action as possible — to reduce the possibility of side effects. I think that the drug developers would endorse a paraphrase of Einstein. The drug (theory) should be as simple as possible but no simpler.
Keep up the great work. Jim Rose
Jim Rose
Astute comments on the preferences of Western medicine for cures that are as simple as possible. On the other hand, body systems are extremely complex and typically involve interactions among multiple pathways. So, if health is an objective, complex substances that impact on multiple pathways may well carry us beyond what is possible using simple one-pathway substances. Big pharma companies which are in a crises because their traditional drug discovery methods are insufficiently productive may become increasingly willing to turn to traditional cures as the basis for creating proprietary products.
Vince
The firewall (Vincent) has discussed and posted on Caloric Restriction, Intermittent Fasting, and Protein Restriction as ways to increase the quality and quantity of our lives so that we might enjoy a very long or indefinite future. Several researches mentioned are a part of this BBC show.
Below is a link via You Tube to a 6 August BBC show
[Horizon] on CR and IF. It is worthy of watching.
The potential of some form of modified fast 2 days a week or every other day to dramatically reduce health care costs and increase healthy life span seems very promising. Also IMO and experiance very do able.
I thought the point on how soon IGF-1 levels dropped by 50% and the association of IGF-1 and so many leading causes or morbidity and mortality would be of specific interest.
My option is every other day with 15% on modified fast days in the form of V8, Coffee, and C3H8O3 or Trehalose to keep up electrolytes.
I try to keep an open mind and look forward to more research and maybe a black swan like Carbon 60 increasing healthy lifespan two fold.
BBC Horizon 2012: Eat, Fast and Live Longer
http://www.youtube.com/watch?v=Pfna7nV7WaM
Pingback: Adaptogens Part 2: – focus on rhodiola – video blog | AGING SCIENCES – Anti-Aging Firewalls
Pingback: PART 1: Slaying Two Dragons with One Stone – How to Prevent Cancer and Aging with the Same Strategy | AGING SCIENCES – Anti-Aging Firewalls
Pingback: Health through stressing fruits and vegetables – the Xenohormetic Live Food Hypothesis | AGING SCIENCES – Anti-Aging Firewalls