By Vince Giuliano
There have been several important research findings in recent years relating to prostate cancer, ones that are transforming our views of the disease process and offering hopes for powerful new preventative and curative therapies. This blog entry covers recent research on epigenetic factors, cancer stem cells, and the role of Nrf2 as related to prostate cancer. Consistent to what I have reported in earlier blog entries, there is hope that plant based substances (phytosubstances) might play important roles in preventing and even possibly curing prostate cancer.
About prostate cancer
From the A.D.A.M. Medical Encyclopedia: “Prostate cancer is cancer that starts in the prostate gland. The prostate is a small, walnut-sized structure that makes up part of a man’s reproductive system. It wraps around the urethra, the tube that carries urine out of the body. — “Prostate cancer is the most common cause of death from cancer in men over age 75. Prostate cancer is rarely found in men younger than 40. — people who are at higher risk include: African-American men, who are also likely to develop cancer at every age, Men who are older than 60, Men who have a father or brother with prostate cancer. — Prostate cancer is less common in people who do not eat meat (vegetarians).”
From Wikipedia: “ Most prostate cancers are slow growing; however, there are cases of aggressive prostate cancers.[1] The cancer cells may metastasize (spread) from the prostate to other parts of the body, particularly the bones and lymph nodes. Prostate cancer may cause pain, difficulty in urinating, problems during sexual intercourse, or erectile dysfunction. Other symptoms can potentially develop during later stages of the disease. — Rates of detection of prostate cancers vary widely across the world, with South and East Asia detecting less frequently than in Europe, and especially the United States.[2] Prostate cancer tends to develop in men over the age of fifty and although it is one of the most prevalent types of cancer in men, many never have symptoms, undergo no therapy, and eventually die of other causes. This is because cancer of the prostate is, in most cases, slow-growing, symptom-free, and since men with the condition are older they often die of causes unrelated to the prostate cancer, such as heart/circulatory disease, pneumonia, other unconnected cancers, or old age. On the other hand, the more aggressive prostate cancers account for more cancer-related deaths among men in the United States than any other cancer except lung cancer.[3] About two-thirds of cases are slow growing, the other third more aggressive and fast developing.[4] == Many factors, including genetics and diet, have been implicated in the development of prostate cancer. The presence of prostate cancer may be indicated by symptoms, physical examination, prostate-specific antigen (PSA), or biopsy. The PSA test increases cancer detection but does not decrease mortality.[5] Moreover, prostate test screening is controversial at the moment and may lead to unnecessary, even harmful, consequences in some patients.[6] Nonetheless, suspected prostate cancer is typically confirmed by taking a biopsy of the prostate and examining it under a microscope. Further tests, such as CT scans and bone scans, may be performed to determine whether prostate cancer has spread.”
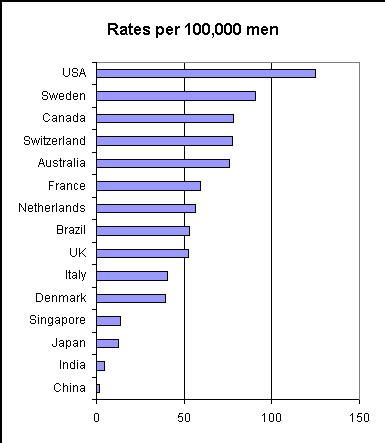
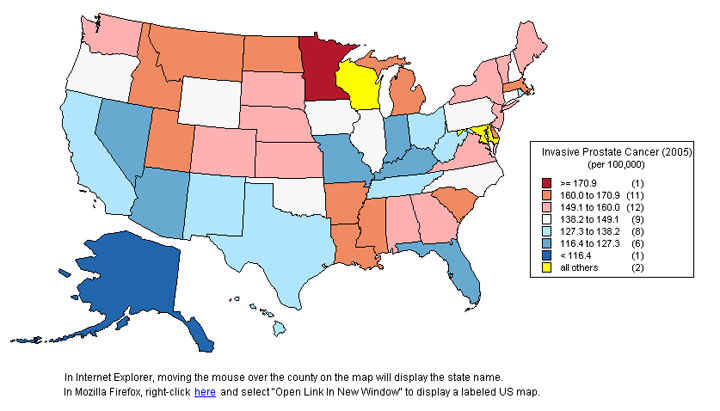
Prostate cancer incidence per 100,000 population vary extremely widely for different countries in the world and even within the US.
Image source (Countries)
Image source (US States)
The origins of prostate cancer appear to be age-related epigenetic changes in prostate cells. Epigenetic changes precede and likely cause the genetic changes that define the cancer.
The wide range of PCa incidence varying by country, State and even locality suggests that environmental and cultural/lifestyle factors that affect epigenetic expression drive PC rates. Research bears out this hypothesis.
The 2009 publication Review Epigenetic alterations in human prostate cancers introduces the general situation: “Human prostate cancer cells carry a myriad of genome defects, including both genetic and epigenetic alterations. These changes, which can be maintained through mitosis, generate malignant phenotypes capable of selective growth, survival, invasion, and metastasis. During prostatic carcinogenesis, epigenetic changes arise earlier than genetic defects, linking the appearance of epigenetic alterations in some way to disease etiology. The most common genetic defect thus far described, leading to fusion transcripts between the androgen-regulated gene TMPRSS2 and genes from the ETS family of transcription factors, likely endows prostate cancer cells with the ability to co-opt androgen signaling, the major prostate differentiation pathway, to support the malignant phenotype. Whether epigenetic changes promote the appearance of TMPRSS2-ETS family fusion transcripts or collaborate with fusion transcript expression in the pathogenesis of prostate cancer has not been established. However, a growing list of epigenetic alterations has provided new opportunities for clinical tests that might aid in prostate cancer screening, detection, diagnosis, staging, and risk stratification. The epigenetic changes appear to be more attractive than genetic changes as prostate cancer biomarkers because epigenetic alterations are present in a greater fraction of prostate cancer cases than any of the known genetic defects. In addition, an emerging generation of assay strategies for detection of specific DNA sequences carrying (5-me)C, the major epigenetic genome mark, has pushed somatic epigenetic alterations to the forefront of molecular biomarker assay development for cancer. Finally, a growing portfolio of epigenetic drugs, capable of reversing the phenotypic consequences of somatic epigenetic defects, has entered clinical trials for prostate cancer in the search for a new rational therapy for the disease.”
The 2010 publication Review [Epigenetics of prostate cancer] continues: “Prostate cancer is one of the most common malignant tumors in males, and its etiology and pathogenesis remain unclear. Epigenesis is involved in prostate cancer at all stages of the process, and closely related with its growth and metastasis. DNA methylation and histone modification are the most important manifestations of epigenetics in prostate cancer. The mechanisms of carcinogenesis of DNA methylation include whole-genome hypomethylation, aberrant local hypermethylation of promoters and genomic instability. DNA methylation is closely related to the process of prostate cancer, as in DNA damage repair, hormone response, tumor cell invasion/metastasis, cell cycle regulation, and so on. Histone modification causes corresponding changes in chromosome structure and the level of gene transcription, and it may affect the cycle, differentiation and apoptosis of cells, resulting in prostate cancer. Some therapies have been developed targeting the epigenetic changes in prostate cancer, including DNA methyltransferases and histone deacetylase inhibitors, and have achieved certain desirable results.”
One if the important kinds of epigenetic shifts in prostate cancer cells is CpG island methylation (silencing) of key genes.
Going back to 2006, the publication CpG island promoter methylation and silencing of 14-3-3sigma gene expression in LNCaP and Tramp-C1 prostate cancer cell lines is associated with methyl-CpG-binding protein MBD2 reported: “14-3-3sigma proteins regulate numerous cellular processes that are important to cancer development. One of its biological roles involves G2 cell-cycle arrest following DNA damage. It has also been reported that the loss of 14-3-3sigma expression via CpG methylation may contribute to malignant transformation by impairing the G2 cell-cycle checkpoint function, thereby allowing an accumulation of genetic defects. However, how the CpG methylation-dependent silencing mechanism works in relation to promoter methylation associated with methyl-CpG-binding proteins (MeCPs) is still unclear. To better understand the mechanism, we first examined the methylation status of the 14-3-3sigma promoter-associated CpG islands and 14-3-3sigma gene expression in a subset of prostate cancer cell lines using methylation-specific PCR (MSP), an HhaI-based DNA methylation assay, and reverse transcription-PCR (RT-PCR). We found that the 14-3-3sigma expression is lost in LNCaP and Tramp-C1 prostate cancer cell lines and that this expression is restored after treatment with epigenetic silencing modifiers 5-aza-2′-deoxycytidine (5-aza) and trichostatin A (TSA). These results imply transcriptional silencing via promoter-associated CpG methylation. Chromatin immunoprecipitation analysis revealed that methyl-CpG-binding protein 2 (MBD2) is associated preferentially to the methylated CpG island in the 14-3-3sigma promoter in LNCaP and Tramp-C1 cells but not in 14-3-3sigma-expressing PC3 and DU145 cells, which contain an unmethylated CpG island in the 14-3-3sigma promoter region. The 14-3-3sigma gene silencing because of CpG methylation correlates with binding of MBD2. In addition, the activation of 14-3-3sigma gene expression by a combination of 5-aza and TSA also involves the release of the MBD2 from the 14-3-3sigma promoter-methylated CpG island in LNCaP and Tramp-C1 cells. Furthermore, MBD2 knockdown by siRNA stimulated 14-3-3sigma expression in LNCaP cells. We also investigated whether the loss of 14-3-3sigma expression in LNCaP and Tramp-C1 cells affects cell proliferation by MTT assays. Interestingly, we observed that 14-3-3sigma-inactivated LNCaP and Tramp-C1 cells had markedly decreased cell proliferation and protein expression of proliferation cell nuclear antigen (PCNA) after restoration of 14-3-3sigma expression with 5-aza and TSA treatment. On the other hand, the same treatment did not significantly affect 14-3-3sigma-active PC3 and DU145 cells, which normally express 14-3-3sigma. Finally, 14-3-3sigma knockdown by siRNA resulted in increased proliferation in PC3 and DU145 cells. These findings suggest that the transcriptional silencing of the 14-3-3sigma gene is caused by promoter CpG island methylation associated with MBD2, and that this may play an important role in prostate cancer progression during the invasive and metastatic stages of the disease.”
Another relatively early publication relating to CpG island promoter methylation in prostate cancer is the 2007 report DNA methylation paradigm shift: 15-lipoxygenase-1 upregulation in prostatic intraepithelial neoplasia and prostate cancer by atypical promoter hypermethylation. “Fifteen (15)-lipoxygenase type 1 (15-LO-1, ALOX15), a highly regulated, tissue- and cell-type-specific lipid-peroxidating enzyme has several functions ranging from physiological membrane remodeling, pathogenesis of atherosclerosis, inflammation and carcinogenesis. Several of our findings support a possible role for 15-LO-1 in prostate cancer (PCa) tumorigenesis. In the present study, we identified a CpG island in the 15-LO-1 promoter and demonstrate that the methylation status of a specific CpG within this island region is associated with transcriptional activation or repression of the 15-LO-1 gene. High levels of 15-LO-1 expression was exclusively correlated with one of the CpG dinucleotides within the 15-LO-1 promoter in all examined PCa cell-lines expressing 15-LO-1 mRNA. We examined the methylation status of this specific CpG in microdissected high grade prostatic intraepithelial neoplasia (HGPIN), PCa, metastatic human prostate tissues, normal prostate cell lines and human donor (normal) prostates. Methylation of this CpG correlated with HGPIN, PCa and metastatic human prostate tissues, while this CpG was unmethylated in all of the normal prostate cell lines and human donor (normal) prostates that either did not display or had minimal basal 15-LO-1 expression. Immunohistochemistry for 15-LO-1 was performed in prostates from PCa patients with Gleason scores 6, 7 [(4+3) and (3+4)], >7 with metastasis, (8-10) and 5 normal (donor) individual males. Quantitative reverse transcription-polymerase chain reaction (qRT-PCR) was used to detect 15-LO-1 in PrEC, RWPE-1, BPH-1, DU-145, LAPC-4, LNCaP, MDAPCa2b and PC-3 cell lines. The specific methylated CpG dinucleotide within the CpG island of the 15-LO-1 promoter was identified by bisulfite sequencing from these cell lines. The methylation status was determined by COBRA analyses of one specific CpG dinucleotide within the 15-LO-1 promoter in these cell lines and in prostates from patients and normal individuals. Fifteen-LO-1, GSTPi and beta-actin mRNA expression in BPH-1, LNCaP and MDAPCa2b cell lines with or without 5-aza-2′-deoxycytidine (5-aza-dC) and trichostatin-A (TSA) treatment were investigated by qRT-PCR. Complete or partial methylation of 15-LO-1 promoter was observed in all PCa patients but the normal donor prostates showed significantly less or no methylation. Exposure of LNCAP and MDAPCa2b cell lines to 5-aza-dC and TSA resulted in the downregulation of 15-LO-1 gene expression. Our results demonstrate that 15-LO-1 promoter methylation is frequently present in PCa patients and identify a new role for epigenetic phenomenon in PCa wherein hypermethylation of the 15-LO-1 promoter leads to the upregulation of 15-LO-1 expression and enzyme activity contributes to PCa initiation and progression.”
The October 2011 review publication Epigenetics in prostate cancer: biologic and clinical relevance looked at the growing literature on the topic at that time. “Context: Prostate cancer (PCa) is one of the most common human malignancies and arises through genetic and epigenetic alterations. Epigenetic modifications include DNA methylation, histone modifications, and microRNAs (miRNA) and produce heritable changes in gene expression without altering the DNA coding sequence. Objective: To review progress in the understanding of PCa epigenetics and to focus upon translational applications of this knowledge. Evidence Acquisition: PubMed was searched for publications regarding PCa and DNA methylation, histone modifications, and miRNAs. Reports were selected based on the detail of analysis, mechanistic support of data, novelty, and potential clinical applications. Evidence Synthesis: Aberrant DNA methylation (hypo- and hypermethylation) is the best-characterized alteration in PCa and leads to genomic instability and inappropriate gene expression. Global and locus-specific changes in chromatin remodeling are implicated in PCa, with evidence suggesting a causative dysfunction of histone-modifying enzymes. MicroRNA deregulation also contributes to prostate carcinogenesis, including interference with androgen receptor signaling and apoptosis. There are important connections between common genetic alterations (eg, E twenty-six fusion genes) and the altered epigenetic landscape. Owing to the ubiquitous nature of epigenetic alterations, they provide potential biomarkers for PCa detection, diagnosis, assessment of prognosis, and post-treatment surveillance. Conclusions: Altered epigenetic gene regulation is involved in the genesis and progression of PCa. Epigenetic alterations may provide valuable tools for the management of PCa patients and be targeted by pharmacologic compounds that reverse their nature. The potential for epigenetic changes in PCa requires further exploration and validation to enable translation to the clinic.”
As I point out later I believe these age-related epigenetic changes related to prostate cancer simply don’t have to happen; they likely can be averted. Further, epigenetic changes are largely reversible in nature.
Dietary factors which impact on epigenetic expression may significantly affect propensity for development of prostate cancer.
A publication which just appeared online this morning (April 2012) is A high-fat diet enhances proliferation of prostate cancer cells and activates MCP-1/CCR2 signaling. “Background: Dietary patterns including high-fat diet (HFD) and high-carbohydrate diet (HCD) play an important role in prostate cancer progression. However, which of these diets have the greatest effect on tumor progression and its underlying mechanisms remains unclear. Methods: We investigated the effects of different diets on prostate cancer cell growth and the relevant circulating factors including serum insulin, growth factors, and inflammatory cytokines using the in vivo and ex vivo model. Results: The tumor growth of prostate cancer LNCaP xenograft was significantly higher in the HFD group than in the HCD and control diet (CD) groups (P = 0.01; HFD vs. HCD, P = 0.025; HFD vs. CD, P = 0.003). The mean level of the serum monocyte chemoattractant protein-1 (MCP-1) in the HFD group was significantly higher than that in the HCD and CD groups (P = 0.024; HFD vs. HCD, P = 0.033; HFD vs. CD, P = 0.001). The mRNA levels of CC chemokine receptor 2 (CCR2), which is an MCP-1 receptor, and the expression of activated Akt were the highest in the HFD group. Furthermore, serum from HFD-fed mice enhanced the proliferation of two PCa cells and CCR2 knockdown inhibited HFD-induced proliferation of LNCaP cells. Conclusions: An HFD enhanced prostate cancer cell growth more strongly than an HCD or CD. MCP-1/CCR2 signaling may be involved in an HFD-induced prostate cancer progression.”
The role of Nrf2 in prostate cancer
As readers, you may or may not be familiar with the triad of blog posts related to Nrf2:
The pivotal role of Nrf2. Part 2 – foods, phyto-substances and other substances that turn on Nrf2, and
The pivotal role of Nrf2. Part 3– Is promotion of Nrf2 expression a viable strategy for human human healthspan and lifespan extension? – and the earlier blog post
Nrf2 and cancer chemoprevention by phytochemicals.
In a nutshell, activation of Nrf2 turns on hundreds of protective antioxidant and other protective genes, including many that combat cancers. Further, Nrf2 expression can be promoted by many everyday substances including familiar phytochemicals found in foods and supplements including curcumin, ginger, broccoli, sprouts and resveratrol. Read the Part 2 blog entry mentioned above.
Nrf2 expression is epigenetically silenced in PCa.
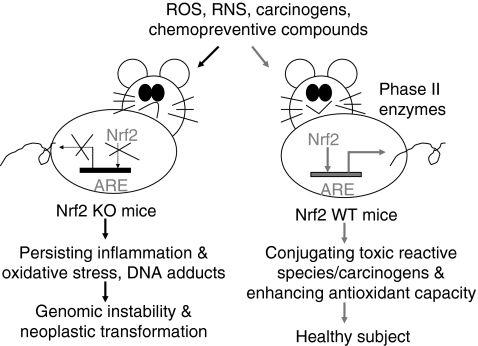
The publication (date unknown) Role of the Nrf2-ARE signaling pathway in prostate tumorigenesis states: “Introduction: Prostate cancer, one of the most frequent cancers in males in Western industrialized countries, is characterized by increased intracellular oxidative stress. Chronic oxidative stress and its associated pathological conditions including inflammation and metabolic disorders have been postulated to drive somatic mutations and neoplastic transformation, thus could play an important role in the development and progression of prostate cancer. To maintain redox homeostasis, mammalian cells have evolved a hierarchy of sophisticated sensing and signaling mechanisms to turn on or off endogenous antioxidant responses accordingly. One the major strategies for preventing cancer and other diseases in the human population is the ability of the chemopreventive agents to induce the expression of cytoprotective enzymes through the activation of the Keap1/Nrf2/ARE pathway (see fig. 1) (3,4) The activity of Nrf2 is normally suppressed in the cytosol by specific binding to the chaperone Keap1. However, when cells are exposed to chemopreventive agents (e.g. dithiolethiones, flavonoids, ITCs) and oxidative stress, Keap1-mediated degradation of Nrf2 is abrogated, releasing Nrf2 to translocate into the nucleus and transactivate the antioxidant response elements (AREs)/electrophile response elements (EpREs) of many cytoprotective genes.(3,4) This leads to the synthesis of a distinct set of antioxidant proteins that efficiently protect mammalian cells from various forms of stress, and consequently, reduce the propensity of tissues and organisms to develop disease or malignancy.(3,4) Upon recovery of cellular redox homeostasis, Keap1 travels into the nucleus to dissociate Nrf2 from the ARE. Subsequently, the Nrf2-Keap1 complex is exported out of the nucleus by the nuclear export sequence (NES) in Keap1. Once in the cytoplasm, the Nrf2-Keap1 complex associates with the Cul3-Rbx1 core ubiquitin machinary, resulting in degradation of Nrf2 and termination of the Nrf2/ARE signaling pathway (see Fig. 2) (5).. Interestingly, recent studies also suggest that overexpression of an importin α7 protein, KPNA6 not only promotes nuclear import of Keap1 but also accelerates the clearance of Nrf2 protein from the nucleus during postinduction phase, therefore, promoting restoration of Nrf2 protein to basal levels.(6). It seems that Keap1may interact with KPNA6 via a mechanism other the classical nuclear localization signals (7). In this study, we provided compelling evidence that the expression of Nrf2 is epigenetically suppressed by its promoter methylation associated with Methyl-CpG-Binding Domain 2 (MBD2) and histone modifications in the prostate cancer tissues of TRAMP mice.(8) — In addition, we reported that both loss of Nrf2 and subsequent induction of the E-cadherin transcriptional repressor Slug can also enhance cellular plasticity and motility in prostate tumor cells, in part by using TGF-β/SMAD malignant signalling (see fig. 3).(9) Figure.1. Proposed pathway for the induction of cytoprotective genes by enzyme inducers. Inducers promote the release of Nrf2 from a cytoplasmic inhibitor Keap1 by altering the structural conformation of Keap1. Protein kinase C (PKC) phosphorylates Nrf2 which can alter the binding of Nrf2 to Keap1. Other signal transduction pathways such as the MAPK cascade and phosphatidylinositol 3-kinase (PI3K) also affect the activation process of Nrf2. Nrf2 then accumulates in the nucleus and transactivates the AREs of many cytoprotective genes as well as Nrf2 itself. The gene families regulated by the Nrf2 pathway include phase 2 enzymes, antioxidants and their modulating enzymes, and the 26S proteasome. Collectively these inducible genes may facilitate the detoxification of carcinogens, enhance the reducing potential against electrophiles and free radicals, and elevate cellular capacity for repair/removal of oxidatively damaged proteins.”
As mentioned in the first of the above posts, the 2010 publication Nrf2 Expression Is Regulated by Epigenetic Mechanisms in Prostate Cancer of TRAMP Mice reported “Nuclear factor-erythroid 2 p45-related factor 2 (Nrf2) is a transcription factor which regulates the expression of many cytoprotective genes. In the present study, we found that the expression of Nrf2 was suppressed in prostate tumor of the Transgenic Adenocarcinoma of Mouse Prostate (TRAMP) mice. Similarly, the expression of Nrf2 and the induction of NQO1 were also substantially suppressed in tumorigenic TRAMP C1 cells but not in non-tumorigenic TRAMP C3 cells. Examination of the promoter region of the mouse Nrf2 gene identified a CpG island, which was methylated at specific CpG sites in prostate TRAMP tumor and in TRAMP C1 cells but not in normal prostate or TRAMP C3 cells, as shown by bisulfite genomic sequencing. Reporter assays indicated that methylation of these CpG sites dramatically inhibited the transcriptional activity of the Nrf2 promoter. — Taken together, these results indicate that the expression of Nrf2 is suppressed epigenetically by promoter methylation associated with MBD2 and histone modifications in the prostate tumor of TRAMP mice. Our present findings reveal a novel mechanism by which Nrf2 expression is suppressed in TRAMP prostate tumor, shed new light on the role of Nrf2 in carcinogenesis and provide potential new directions for the detection and prevention of prostate cancer.”
“The TRAMP mouse is an autochthonous transgenic animal model of PCa that recapitulates the whole spectrum of human prostate tumorigenesis from the earliest PIN lesions to androgen-independent disease (71). Without chemical or hormonal treatment, 100% of male TRAMP develops PCa and progress from PIN to histological cancer to carcinoma metastasis to lymph nodes, lungs, and occasionally bones sequentially over 12–28 weeks (39, 40)(ref).”
At least in mice, disruption of Nrf2 increases susceptibility to carcinogenesis, and this applies to prostate cancer.
The point is argued in the 2010 review publication Regulation of NF-E2-related factor 2 signaling for cancer chemoprevention: antioxidant coupled with antiinflammatory.
“Schematic presentation showing that Nrf2-disrupted mice (Nrf2 KO) have a higher susceptibility to carcinogenesis. The critical role of Nrf2 in protecting mice from neoplastic transformation when subject to oxidative stress and carcinogens is intact in Nrf2 WT mice having functional Nrf2-ARE signaling, by enhancing expression of detoxifying metabolizing enzymes and maintaining oxidative stress homeostasis by producing antioxidative stress enzymes. Application of chemopreventive compounds in Nrf2 WT mice can further enhance expression of phase II detoxifying and antioxidant enzymes by regulating the Nrf2-ARE signaling. ARE, antioxidant responsive element; Nrf2, NF-E2-related factor 2; Nrf2 KO, Nrf2 knockout; Nrf2 WT mice, wild-type mice with intact Nrf2 function.”
So here we observe at least one smoking gun when it comes to prostate cancers: Nrf2 is epigenetically silenced, neutralizing its multiple anti-cancer effects.
Mutations in the Keap1 protein which binds Nrf2 in in prostate cancer cells causes chemoresistance and radioresistance and promotes tumor growth due to increased Nrf2 activity in the cancer cells.
There appears to be another side to the role of Nrf2 in prostate cancer. Nrf2 can also promote the survival of cancer cells confronting attacks by chemotherapy agents or radiation, once they have turned cancerous. The 2010 publication Loss of Kelch-like ECH-associated protein 1 function in prostate cancer cells causes chemoresistance and radioresistance and promotes tumor growthreports: “Loss-of-function mutations in the nuclear factor erythroid-2-related factor 2 (Nrf2) inhibitor Kelch-like ECH-associated protein 1 (Keap1) result in increased Nrf2 activity in non-small cell lung cancer and confer therapeutic resistance. We detected point mutations in Keap1 gene, leading to nonconservative amino acid substitutions in prostate cancer cells. We found novel transcriptional and posttranscriptional mechanisms of Keap1 inactivation, such as promoter CpG island hypermethylation and aberrant splicing of Keap1, in DU-145 cells. Very low levels of Keap1 mRNA were detected in DU-145 cells, which significantly increased by treatment with DNA methyltransferase inhibitor 5-aza-deoxycytidine. The loss of Keap1 function led to an enhanced activity of Nrf2 and its downstream electrophile/drug detoxification pathway. Inhibition of Nrf2 expression in DU-145 cells by RNA interference attenuated the expression of glutathione, thioredoxin, and the drug efflux pathways involved in counteracting electrophiles, oxidative stress, and detoxification of a broad spectrum of drugs. DU-145 cells constitutively expressing Nrf2 short hairpin RNA had lower levels of total glutathione and higher levels of intracellular reactive oxygen species. Attenuation of Nrf2 function in DU-145 cells enhanced sensitivity to chemotherapeutic drugs and radiation-induced cell death. In addition, inhibition of Nrf2 greatly suppressed in vitro and in vivo tumor growth of DU-145 prostate cancer cells. Thus, targeting the Nrf2 pathway in prostate cancer cells may provide a novel strategy to enhance chemotherapy and radiotherapy responsiveness and ameliorate the growth and tumorigenicity, leading to improved clinical outcomes.”
This publication appears to be at odds with the other cited publications that assert that Nrf2 is not expressed in PCa cells. According to this publication, Instead of being epigenetically silenced, Nrf2 is overexpressed due to dysfunctionality of Keap1. I do not know how to reconcile the two viewpoints except to suggest that Nrf2 is protective of normal cells against carcinogenesis, but once they have turned cancerous then Nrf2 may be protective of the cancer cells.
Certain plant polyphenols can help prevent or inhibit proliferation of prostate cancers. The underlying mechanisms no doubt involve Nrf2, either direct activation of Nrf2 or activation via reversal of Nrf2 promoter methylation.
The March 2012 publicationPolyphenols in brewed green tea inhibit prostate tumor xenograft growth by localizing to the tumor and decreasing oxidative stress and angiogenesis reports: “It has been demonstrated in various animal models that the oral administration of green tea (GT) extracts in drinking water can inhibit tumor growth, but the effects of brewed GT on factors promoting tumor growth, including oxidant damage of DNA and protein, angiogenesis and DNA methylation, have not been tested in an animal model. To explore these potential mechanisms, brewed GT was administered instead of drinking water to male severe combined immunodeficiency (SCID) mice with androgen-dependent human LAPC4 prostate cancer cell subcutaneous xenografts. Tumor volume was decreased significantly in mice consuming GT, and tumor size was significantly correlated with GT polyphenol (GTP) content in tumor tissue. There was a significant reduction in hypoxia-inducible factor 1-alpha and vascular endothelial growth factor protein expression. GT consumption significantly reduced oxidative DNA and protein damage in tumor tissue as determined by 8-hydroxydeoxyguanosine/deoxyguanosine ratio and protein carbonyl assay, respectively. Methylation is known to inhibit antioxidative enzymes such as glutathione S-transferase pi to permit reactive oxygen species promotion of tumor growth. GT inhibited tumor 5-cytosine DNA methyltransferase 1 mRNA and protein expression significantly, which may contribute to the inhibition of tumor growth by reactivation of antioxidative enzymes. This study advances our understanding of tumor growth inhibition by brewed GT in an animal model by demonstrating tissue localization of GTPs in correlation with inhibition of tumor growth. Our results suggest that the inhibition of tumor growth is due to GTP-mediated inhibition of oxidative stress and angiogenesis in the LAPC4 xenograft prostate tumor in SCID mice.” The clear implication is that inhibition of oxidative stress was due to activation of Nrf2 and consequent release of endogenous antioxidant enzymes.
Curcumin exercises its anti-cancer effects on prostate cancer at least in part by demethylating the CpG sites in the promoter region of the Neurog1 gene, unsilencing that gene so it restores the expression of Nrf2 which in turn stimulates the ARE elements which result in changes in gene expression in multiple genes which combat the cancer.
The 2011 publication Epigenetic CpG Demethylation of the Promoter and Reactivation of the Expression of Neurog1 by Curcumin in Prostate LNCaP Cells reports “Curcumin (CUR), a major bioactive polyphenolic component from turmeric curry, Curcuma longa, has been shown to be a potent anti-cancer phytochemical with well-established anti-inflammatory and anti-oxidative stress effects. Chromatin remodeling-related epigenetic regulation has emerged as an important mechanism of carcinogenesis, chemoprevention, and chemotherapy. CUR has been found to inhibit histone acetyltransferase activity, and it was also postulated to be a potential DNA methyltransferase (DNMT) and histone deacetylase (HDAC) inhibitor. In this study, we show that when human prostate LNCaP cells were treated with CUR, it led to demethylation of the first 14 CpG sites of the CpG island of the Neurog1 gene and restored the expression of this cancer-related CpG-methylation epigenome marker gene. At the protein level, CUR treatment had limited effects on the expression of epigenetic modifying proteins MBD2, MeCP2, DNMT1, and DNMT3a. Using ChIP assay, CUR decreased MeCP2 binding to the promoter of Neurog1 dramatically. CUR treatment showed different effects on the protein expression of HDACs, increasing the expression of HDAC1, 4, 5, and 8 but decreasing HDAC3. However, the total HDAC activity was decreased upon CUR treatment. Further analysis of the tri-methylation of histone 3 at lysine 27 (H3K27me3) showed that CUR decreased the enrichment of H3K27me3 at the Neurog1 promoter region as well as at the global level. Taken together, our present study provides evidence on the CpG demethylation ability of CUR on Neurog1 while activating its expression, suggesting a potential epigenetic modifying role for this phytochemical compound in human prostate cancer cells.”
The November 2011 publication Pharmacodynamics of curcumin as DNA hypomethylation agent in restoring the expression of Nrf2 via promoter CpGs demethylation reports: “Prostate cancer (PCa) is one of the most deadly malignancies among men in the United States. Although localized prostate cancer can be effectively treated via surgery or radiation, metastatic disease is usually lethal. Recent evidence suggests that the development and progression of human prostate cancer involves complex interplay between epigenetic alterations and genetic defects. We have recently demonstrated that Nrf2, a master regulator of cellular antioxidant defense systems, was epigenetically silenced during the progression of prostate tumorigenesis in TRAMP mice. The aim of this study is to investigate the potential of curcumin (CUR), a dietary compound that we have reported to be able to prevent the development of prostate cancer in TRAMP mice, as a DNA hypomethylation agent. Using bisulfite genomic sequencing (BGS), treatment of TRAMP C1 cells we showed that CUR reversed the methylation status of the first 5 CpGs in the promoter region of the Nrf2 gene. Methylation DNA immunoprecipitation (MeDIP) analysis revealed that CUR significantly reduced the anti-mecyt antibody binding to the first 5 CpGs of the Nrf2 promoter, corroborated the BGS results. Demethylation of Nrf2 was found to be associated with the re-expression of Nrf2 and one of its downstream target gene, NQO-1, one of the major anti-oxidative stress enzymes, both at the mRNA and protein levels. Taken together, our current study suggests that CUR can elicit its prostate cancer chemopreventive effect, potentially at least in part, through epigenetic modification of the Nrf2 gene with its subsequent induction of the Nrf2-mediated anti-oxidative stress cellular defense pathway.”
The new knowledge of epigenetics and Nrf2 gives much new creditability to a very old idea – that the best way to deal with prostate cancer is to avoid getting it in the first place. And that, that most likely can be accomplished by attention to diet and taking selected polyphenol supplements.
The November 2011 publication Dietary Factors and Epigenetic Regulation for Prostate Cancer Prevention reports: “The role of epigenetic alterations in various human chronic diseases has gained increasing attention and has resulted in a paradigm shift in our understanding of disease susceptibility. In the field of cancer research, e.g., genetic abnormalities/mutations historically were viewed as primary underlying causes; however, epigenetic mechanisms that alter gene expression without affecting DNA sequence are now recognized as being of equal or greater importance for oncogenesis. Methylation of DNA, modification of histones, and interfering microRNA (miRNA) collectively represent a cadre of epigenetic elements dysregulated in cancer. Targeting the epigenome with compounds that modulate DNA methylation, histone marks, and miRNA profiles represents an evolving strategy for cancer chemoprevention, and these approaches are starting to show promise in human clinical trials. Essential micronutrients such as folate, vitamin B-12, selenium, and zinc as well as the dietary phytochemicals sulforaphane, tea polyphenols, curcumin, and allyl sulfur compounds are among a growing list of agents that affect epigenetic events as novel mechanisms of chemoprevention. To illustrate these concepts, the current review highlights the interactions among nutrients, epigenetics, and prostate cancer susceptibility. In particular, we focus on epigenetic dysregulation and the impact of specific nutrients and food components on DNA methylation and histone modifications that can alter gene expression and influence prostate cancer progression.”
Another 2012 publication presenting a compatible picture is Chemoprevention of Prostate Cancer with Cruciferous Vegetables: Role of Epigenetics: “Globally, prostate cancer is the second most frequently diagnosed cancer in men although the incidence of cancer varies greatly throughout the world. Nutrition and diet are important modifiable risk factors for prostate cancer development. Epidemiological studies have shown an inverse association between cruciferous vegetable intake and the risk of developing prostate cancer. Here we focus specifically on the molecular mechanisms by which phytochemicals in cruciferous vegetables, sulforaphane (SFN), indole-3-carbinol (I3C) and its derivative 3,3′-diindolylmethane (DIM), may prevent the initiation of prostate cancer and slow tumorigenesis. We have particularly emphasized a possible role for epigenetics in this process as many dietary factors can modulate epigenetic alterations and alter susceptibility to disease. We have identified known and possible epigenetic mechanisms by which these phytochemicals can alter detoxification pathways, sex hormone signaling, and genes that regulate cell cycle, apoptosis, inflammation, angiogenesis and metastasis. The ability of SFN, I3C or DIM to target aberrant epigenetic patterns, in addition to their effects on detoxification/carcinogen metabolism, may make them effective chemoprevention agents at multiple stages of the prostate carcinogenesis pathway. The identification of dietary epigenetic modulators and their use either alone or in combination, may increase efficacy of anti-cancer therapies and prevention strategies, without serious side effects.” Although this abstract does not mention Nrf2, the active ingredient discussed, sulforaphane, is a powerful activator of Nrf2.
About prostate cancer stem cells
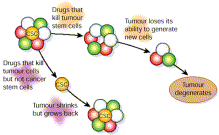
Going back over more than six years now, it has been recognized that prostate cancer tumor masses contain cancer cells that have stem-cell like properties. Killing cancer cells may not provide an adequate therapy for the disease as long as cancer stem cells remain alive.
The 2006 publication Prostate cancer stem cellsreported “Prostate cancer is the most frequently diagnosed cancer in men. Despite recent advances in the detection of early prostate cancer there is little effective therapy for patients with locally advanced and/or metastatic disease. The majority of patients with advanced disease respond initially to androgen ablation therapy. However, most go on to develop androgen-independent tumours that inevitably are fatal. A similar response is seen to chemotherapeutic and radiotherapy treatments. As a result, metastatic prostate cancer remains an incurable disease by current treatment strategies. Recent reports of cancer stem cells have prompted questions regarding the involvement of normal stem/progenitor cells in prostate tumour biology, their potential contribution to the tumour itself and whether they are the cause of tumour initiation and progression. Although still controversial, the cancer stem cell is likely to be the most crucial target in the treatment of prostate cancer, and a thorough understanding of its biology, particularly of how the cancerstemcell differs from the normal stemcell, might allow it to be targeted selectively and eliminated, thus improving therapeutic outcome.”
The 2008 publication Prostate cancer stem cellsreported “Despite the discovery over 60 years ago by Huggins and Hodges 1 that prostate cancers respond to androgen deprivation therapy, hormone-refractory prostate cancer remains a major clinical challenge. There is now mounting evidence that solid tumours originate from undifferentiated stem cell-like cells coexisting within a heterogeneous tumour mass that drive tumour formation, maintain tumour homeostasis and initiate metastases. This review focuses upon current evidence for prostate cancer stem cells, addressing the identification and properties of both normal and transformed prostate stem cells. — Despite recent advances in the detection of early prostate cancer, there remains little effective therapy for patients with locally advanced and/or metastatic disease. The majority of patients with advanced disease respond initially to androgen ablation therapy, due to the androgen-dependent nature of the vast majority of prostate cancer cells. However, with very high frequency, androgen-independent cancers emerge and subsequently widespread metastasis occur. — The development of more effective treatment strategies for prostate cancer must target all the cells within a tumour. Gene expression profiling from our laboratory has highlighted key cell signalling pathways that are over-represented in the cancer stem cell population. Abrogation of these pathways, leading to disruption of self-renewal, should be a key area of research. More sophisticated modes of therapy may be necessary, such as combination of a DNA damaging agent with a DNA repair inhibitor. Ultimately, it would be desirable to have a treatment against prostate cancer stem cells that could be used in combination with androgen ablation therapy to reduce tumour mass.”
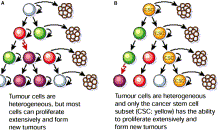
Dozens of other publications have validated the existence and importance of cancer stem cells. These two illustrations from the 2007 publication Cancer stem cells: A new paradigm for understanding tumor progression and therapeutic resistance illustrate the functional differences between cancer stem cells and the thereapeutic implications connected with their existence.
+Fig 1. The cancer stem cell theory. The traditional thinking about how neoplasms develop is shown in (A), where most tumor cells can proliferate extensively and form new tumors. In the cancer stem cell theory (B), tumor cells are heterogenous, but only cancer stem cells are able to proliferate extensively and form new tumors. These cells are termed cancer stem cells because like normal stem cells, they can both self renew and produce differentiated progeny. With permission from Reya T, Morrison SJ, Clarke MF, et al. Stem cells, cancer, and cancer stem cells. Nature 2001;414:105-11(ref).”
“Fig 2. Cancer stem cells are resistant to standard therapies. Only treatments that specifically target cancer stem cells will result in cancer cure. With permission from Reya T, Morrison SJ, Clarke MF, et al. Stem cells, cancer, and cancer stem cells. Nature 2001;414:105-11(ref).”
Toxic metals induce genetic transformation of prostate cells into prostate cancer stem cells. And further, arsenic-transformed malignant epithelial cells recruit nearby normal stem cells into a cancer phenotype thereby potentially increasing cancer stem cell number.
The March 2012 publication Arsenic-Transformed Malignant Prostate Epithelia Can Convert Noncontiguous Normal Stem Cells into an Oncogenic Phenotype reports: “Background: Cancer stem cells (CSCs) are likely critical to carcinogenesis, and, like normal stem cells (NSCs), are impacted by microenvironment. Malignant cells release extracellular factors modifying tumor behavior. Inorganic arsenic, a human carcinogen, over-produces CSCs in various model systems of carcinogenesis. Here, we determine if NSCs are influenced by nearby arsenic-transformed malignant epithelial cells (MECs) as a possible factor in arsenic associated CSC overabundance. Methods: Transwell non-contact co-culture allowed the study of the effects of non-contiguous, arsenic-transformed prostate MECs on the isogenic human prostate NSC line, WPE-stem. Cancer phenotype was assessed by secreted MMPs, invasiveness, colony formation and spheroid formation. Gene expression was assessed at the protein (western blot) or mRNA (RT-PCR) levels. Results: Non-contact co-culture of MECs and NSCs rapidly (≤ 3 weeks) caused hyper-secretion of MMPs and marked suppression of the tumor suppressor gene PTEN in NSCs. NSCs co-cultured with MECs also showed increased invasiveness and clonogenicity and formed more free-floating spheroids and highly branched ductal-like structures in Matrigel, all typical for CSCs. MEC co-culture caused dysregulated self-renewal and differentiation-related gene expression patterns and epithelial-to-mesenchymal transition in NSCs consistent with acquired cancer phenotype. Interleukin-6, a cytokine involved in tumor microenvironment control, was hyper-secreted by MECs and interleukin-6 exposure duplicated several responses in NSCs of conversion to CSCs via MEC co-culture (MMP hyper-secretion, decreased PTEN, etc.). Conclusions: These results indicate that arsenic-transformed MECs recruit nearby NSCs into a cancer phenotype thereby potentially increasing CSC number. This may be a factor in arsenic-induced CSC overabundance seen in multiple model systems.”
In prostate cancers, deregulated expression of microRNA’s belonging to the let-7 family results in upregulation of cancer stem cell activity
The March 2012 publication Loss of Let-7 Up-Regulates EZH2 in Prostate Cancer Consistent with the Acquisition of CancerStem Cell Signatures That Are Attenuated by BR-DIM reports: “Castration-resistant prostate cancer (CRPC) contributes to the high mortality of patients diagnosed with prostate cancer (PCa), which in part could be attributed to the existence and the emergence of cancer stem cells (CSCs). Recent studies have shown that deregulated expression of microRNAs (miRNAs) contributes to the initiation and progression of PCa. Among several known miRNAs, let-7 family appears to play a key role in the recurrence and progression of PCa by regulating CSCs; however, the mechanism by which let-7 family contributes to PCa aggressiveness is unclear. Enhancer of Zeste homolog 2 (EZH2), a putative target of let-7 family, was demonstrated to control stem cell function. In this study, we found loss of let-7 family with corresponding over-expression of EZH2 in human PCa tissue specimens, especially in higher Gleason grade tumors. Overexpression of let-7 by transfection of let-7 precursors decreased EZH2 expression and repressed clonogenic ability and sphere-forming capacity of PCa cells, which was consistent with inhibition of EZH2 3’UTR luciferase activity. We also found that the treatment of PCa cells with BR-DIM (formulated DIM: 3,3′-diindolylmethane by Bio Response, Boulder, CO, abbreviated as BR-DIM) up-regulated let-7 and down-regulated EZH2 expression, consistent with inhibition of self-renewal and clonogenic capacity. Moreover, BR-DIM intervention in our on-going phase II clinical trial in patients prior to radical prostatectomy showed upregulation of let-7 consistent with down-regulation of EZH2 expression in PCa tissue specimens after BR-DIM intervention. These results suggest that the loss of let-7 mediated increased expression of EZH2 contributes to PCa aggressiveness, which could be attenuated by BR-DIM treatment, and thus BR-DIM is likely to have clinical impact.”
Radiation, a traditional therapy for prostate cancer, may damage cancer stem cells, but they have a significant capability to recover and repopulate the cancer.
Another publication that appeared online this morning (April; 2012) is Long-term recovery of irradiated prostate cancer increases cancer stem cells. “Background: Despite improvements in treatment, prostate cancer (PC) remains the second-leading cause of cancer death in men. Radiotherapy is among the first-line treatments for PC, but a significant number of patients relapse. Recent evidence supports the idea that PC is initiated by a subset of cells, termed cancer stem cells (CSCs). CSCs have also been implicated in radioresistance in various malignancies, but their role in PC has not yet been investigated. Methods: We compared the relative radiosensitivity of isolated CSCs to the total population of their corresponding cell lines, and examined the relative numbers of CSCs in irradiated cell lines following long-term recovery and in recurrent human PC. Results: Here, we show that while irradiation does not immediately favor increased survival of CSCs, irradiated PC cell lines showed an increase in CSC properties with long-term recovery. These data suggest that, although CSCs are initially damaged by radiation, they possess a greater capacity for recovery and regrowth. Conclusions: The combination of radiotherapy with a CSC-targeted therapeutic strategy may prevent tumor recurrence.” Again the bottom line seems to be that to really vanish the cancer, it is necessary to go after the cancer stem cells as well as the normal cancer cells.
Phytosubstances can target prostate cancer stem cells.
The March 2012 publication Novel epigallocatechin gallate (EGCG) analogs activate AMP-activated protein kinase pathway and target cancer stem cells reports: “AMP-activated protein kinase (AMPK) is a critical monitor of cellular energy status and also controls processes related to tumor development, including cell cycle progression, protein synthesis, cell growth and survival. Therefore AMPK as an anti-cancer target has received intensive attention recently. It has been reported that the anti-diabetic drug metformin and some natural compounds, such as quercetin, genistein, capsaicin and greentea polyphenol epigallocatechin gallate (EGCG), can activate AMPK and inhibit cancer cell growth. Indeed, natural products have been the most productive source of leads for the development of anti-cancer drugs but perceived disadvantages, such as low bioavailability and week potency, have limited their development and use in the clinic. In this study we demonstrated that synthetic EGCG analogs 4 and 6 were more potent AMPK activators than metformin and EGCG. Activation of AMPK by these EGCG analogs resulted in inhibition of cell proliferation, up-regulation of the cyclin-dependent kinase inhibitor p21, down-regulation of mTOR pathway, and suppression of stem cell population in human breast cancer cells. Our findings suggest that novel potent and specific AMPK activators can be discovered from natural and synthetic sources that have potential to be used for anti-cancer therapy in the clinic.”
The April 2012 publication Genistein inhibits the stemness properties of prostate cancer cells through targeting Hedgehog-Gli1 pathway reports: “Cancer stem cells (CSCs) are involved in tumorigenesis and progression of prostate cancer (PCa). Conventional anticancer therapeutics failed to eradicate CSCs, which may eventually lead to the disease relapse and metastasis. Therefore, targeting prostate CSCs may be an ideal strategy to cure PCa. — Genistein is a major isoflavone constituent of soybeans and soy products, which has been shown to exhibit potent anticancer effect on many cancers. We have previously reported that genistein can inhibit PCa cell invasion by reversing epithelial to mesenchymal transition, suggesting that genistein may be effective against metastatic PCa. In addition, we have recently demonstrated that PCa tumorsphere cells (TCs) possess CSC properties. Here, we found that tumorsphere formation and colony formation of Pca cells were noticeably suppressed in the presence of genistein. Pretreatment of PCa TCs with genistein also suppressed tumorigenicity in vivo. Additionally, genistein treatment inhibited tumor growth of PCa TCs. Further studies showed that genistein treatment not only led to the down-regulation of PCa CSC markers CD44 in vitro and in vivo, but also inhibited Hedgehog-Gli1 pathway, which may contribute to the anti-CSC effect of genistein in PCa TCs. Therefore, our findings demonstrated that genistein may be a dietary phytochemical with potential to target prostate CSCs.”
Milk thistle may be protective against prostate cancer.
The 2005 publication Milk thistle and prostate cancer: differential effects of pure flavonolignans from Silybum marianum on antiproliferative end points in human prostate carcinoma cellsreported: “Extracts from the seeds of milk thistle, Silybum marianum, are known commonly as silibinin and silymarin and possess anticancer actions on human prostate carcinoma in vitro and in vivo. Seven distinct flavonolignan compounds and a flavonoid have been isolated from commercial silymarin extracts. Most notably, two pairs of diastereomers, silybin A and silybin B and isosilybin A and isosilybin B, are among these compounds. In contrast, silibinin is composed only of a 1:1 mixture of silybin A and silybin B. With these isomers now isolated in quantities sufficient for biological studies, each pure compound was assessed for antiproliferative activities against LNCaP, DU145, and PC3 human prostate carcinoma cell lines. Isosilybin B was the most consistently potent suppressor of cell growth relative to either the other pure constituents or the commercial extracts. Isosilybin A and isosilybin B were also the most effective suppressors of prostate-specific antigen secretion by androgen-dependent LNCaP cells. Silymarin and silibinin were shown for the first time to suppress the activity of the DNA topoisomerase IIalpha gene promoter in DU145 cells and, among the pure compounds, isosilybin B was again the most effective. These findings are significant in that isosilybin B composes no more than 5% of silymarin and is absent from silibinin. Whereas several other more abundant flavonolignans do ultimately influence the same end points at higher exposure concentrations, these findings are suggestive that extracts enriched for isosilybin B, or isosilybin B alone, might possess improved potency in prostate cancer prevention and treatment.”
A publication that appeared online this morning (April 2012) documents the effects of milk thistle on PCa further Angiopreventive Efficacy of Pure Flavonolignans from Milk Thistle Extract against Prostate Cancer: Targeting VEGF-VEGFR Signaling. “The role of neo-angiogenesis in prostate cancer (PCA) growth and metastasis is well established, but the development of effective and non-toxic pharmacological inhibitors of angiogenesis remains an unaccomplished goal. In this regard, targeting aberrant angiogenesis through non-toxic phytochemicals could be an attractive angiopreventive strategy against PCA. The rationale of the present study was to compare the anti-angiogenic potential of four pure diastereoisomeric flavonolignans, namely silybin A, silybin B, isosilybin A and isosilybin B, which we established previously as biologically active constituents in Milk Thistle extract. Results showed that oral feeding of these flavonolignans (50 and 100 mg/kg body weight) effectively inhibit the growth of advanced human PCA DU145 xenografts. Immunohistochemical analyses revealed that these flavonolignans inhibit tumor angiogenesis biomarkers (CD31 and nestin) and signaling molecules regulating angiogenesis (VEGF, VEGFR1, VEGFR2, phospho-Akt and HIF-1α) without adversely affecting the vessel-count in normal tissues (liver, lung, and kidney) of tumor bearing mice. These flavonolignans also inhibited the microvessel sprouting from mouse dorsal aortas ex vivo, and the VEGF-induced cell proliferation, capillary-like tube formation and invasiveness of human umbilical vein endothelial cells (HUVEC) in vitro. Further studies in HUVEC showed that these diastereoisomers target cell cycle, apoptosis and VEGF-induced signaling cascade. Three dimensional growth assay as well as co-culture invasion and in vitro angiogenesis studies (with HUVEC and DU145 cells) suggested the differential effectiveness of the diastereoisomers toward PCA and endothelial cells. Overall, these studies elucidated the comparative anti-angiogenic efficacy of pure flavonolignans from Milk Thistle and suggest their usefulness in PCA angioprevention.”
A proprietary combination of five phyto-substances, Protandim, embodies milk thistle extract along with bacopa extract, ashwagandha, green tea extract and curcumin. The substance appears to have a remarkable ability to enhance expression of Nrf2. As described in the 2011 publication Oxidative stress in health and disease: the therapeutic potential of Nrf2 activation, “in a recent clinical trial of Protandim (a composition of multiple synergistic phytochemical Nrf2 activators) the average individual showed an increase of erythrocyte SOD of 34%. As the entire human body contains roughly 7 g of SOD, this 34% increase, if seen in all organs, would result in a steady-state increase of more than 145 6,000,000 U of SOD activity distributed throughout the body (Nelson et al., 2006). Thus, the Nrf2-induced increase produced more than 100 times the amount of SOD activity provided by a 15 mg injection of the purified enzyme. This, coupled with the fact that hundreds of other so-called ‘‘survival’’ genes are modulated by Nrf2 (in addition to SOD1), makes Nrf2 activation appear to be a very attractive alternative to the use of antioxidant enzymes, or of synthetic mimetics of antioxidant enzymes, or of natural or synthetic molecules touted to be ‘‘antioxidants’’ by virtue of their abilities to react stoichiometrically with oxidants or free radicals.” That publication points out how phytosubstances operate against multiple genes involved in cancers while typically, conventional cancer treatments only address a single gene or pathway. –“In colon carcinoma, IPA analysis revealed 28 genes associated with the disease that were also modulated by Protandim treatment. Of these, the first 25 listed (89%) were regulated by Protandim in the opposing direction to that taken by the colon carcinoma disease process. In addition, Protandim downregulated the one gene targeted by a chemotherapeutic drug, an antimetabolite inhibitor for that gene’s product, thymidylate synthetase.”
Clinical trials galore
A search of clinicaltrials.gov for prostate cancer yielded 2197 studies. Some I thought to be possibly interesting are:
- Selenium in Preventing Prostate Cancer
- Low-Fat Diet and/or Flaxseed in Preventing Prostate Cancer
- Diet in Altering Disease Progression in Patients With Prostate Cancer on Active Surveillance
- Study of Antioxidants on Prostate Tumors in Men Undergoing Radical Prostatectomy for Prostate Cancer
- Everolimus and Gefitinib in Treating Patients With Progressive Glioblastoma Multiforme or Progressive Metastatic Prostate Cancer
- DNA Changes in Patients With Prostate Cancer
- flavones Compared With Lycopene Before Surgery in Treating Patients With Stage I or Stage II Prostate Cancer
- Selenium in Treating Patients Who Are Undergoing Brachytherapy for Stage I or Stage II Prostate Cancer
- Isoflavones in Preventing Further Development of Cancer in Patients With Stage I or Stage II Prostate Cancer
- Selenium in Treating Patients With Prostate Cancer
- Lycopene in Preventing Prostate Cancer in Patients Who Are at High Risk of Developing Prostate Cancer
- Low-Fat Fish Oil Diet for Prostate Cancer Prevention
- Lapatinib in Treating Patients With Prostate Cancer That Did Not Respond to Hormone Therapy
- Adoptive Transfer of Autologous T Cells Targeted to Prostate Specific Membrane Antigen (PSMA) for the Treatment of Castrate Metastatic Prostate Cancer (CMPC)
- Feasibility Study on a Nordic Lifestyle Intervention Trial Among Men With Prostate Cancer
- Hormone Therapy Plus Chemotherapy in Treating Patients With Prostate Cancer
- Isoflavones and Radiation Therapy in Treating Patients With Localized Prostate Cancer
- Low-Fat, High-Fiber Diet Compared to a Standard Diet in Treating Patients With Prostate Cancer
- Valproic Acid in Treating Patients With Progressive, Non-Metastatic Prostate Cancer
- Diet and PSA Levels in Patients With Prostate Cancer
- Study of Cabazitaxel Plus Bavituximab as Second-line Chemotherapy for Patients With Castration-resistant Prostate Cancer
- Study of Metformin With Simvastatin for Men With Prostate Carcinoma
- Soy Supplements in Treating Patients Undergoing Surgery for Localized Prostate Cancer
- Lycopene in Treating Patients Undergoing Radical Prostatectomy for Prostate Cancer
- Hormone Therapy in Treating Patients Who Have Stage I or Stage II Prostate Cancer
- Soy Protein in Preventing Recurrent Cancer in Patients Who Have Undergone Surgery for Stage II Prostate Cancer
- Vitamin D and Soy Supplements in Treating Patients With Recurrent Prostate Cancer
This collection is hardly a comprehensive or representative list of the clinical trials. It lists studies that grabbed my attention out of only the first 300 of the 2197 studies listed
Wrapping it up
- Prostate cancer is a major killer and much research if focused on it. Pubmed.org shows 100,515 research articles related to it. This blog item has selectively focused on epigenetics in the etiology of prostate cancer, the roles of Nrf2, cancer stem cells and the potential roles of phyto-substances to reduce the incidence of PCa through upregulating Nrf2 expression. New results are appearing daily. Three of the citations appearing in the blog just showed up online this morning.
- Prostate cancer can be brought on by adverse environmental conditions, like presence of toxic metals, but epigenetic changes are likely both to precede and cause genetic mutations that define the disease
- Defects in expression of Nrf2 are likely to be causal of prostate cancers and increased expression of Nrf2. Among the key epigenetic mechanisms involved in PCa origination is methylation of promoter areas in protective genes like those related to Nrf2, silencing them.
- To cure a case of prostate cancer it is not enough to kill the cancer cells; it is necessary to kill the cancer stem cells as well.
- A number of phytosubstances, plant based chemicals, appear to be affective against prostate cancer, affecting both cancer cells and cancer stem cells. Mechanisms of operation appear to epigenetic, including inhibition of histone deacytelation leading to demethylation of promoter areas if key protective genes and activation of Nrf2.
- Taking all the research evidence into account, I strongly suspect that supplementation with such substances coupled with a diet rich in phytosubstances and prudent lifestyle behavior can avert most incidences of prostate cancer or significantly delay their onset
There is a need to connect the dots to make full sense of the research.
Finally, I point out that many of the individual publications fall into clusters dealing with specific sub-topics and fail to “connect the dots” with publications in other clusters. So, there is a need to read across several clusters of publications to develop an overall understanding. Specifically, the publications in the cluster that relate Nrf2 to prostate cancer generally do not refer to actions the protective phytosubstances (even though the phytosubstances largely work by activating Nrf2). Most of the publications in the cluster that points to the positive actions of phytosubstances in prostate cancer do not refer to Nrf2 (even though that is a main mechanism through which they work). What is further missing is in yet-another set of research publications, those that indicates how phytosubstances promote Nrf2 (which generally do not refer to cancers). See the blog entry The pivotal role of Nrf2. Part 2 – foods, phyto-substances and other substances that turn on Nrf2 and the earlier blog entry Nrf2 and cancer chemoprevention by phytochemicals.
FROM TIME TO TIME, THIS BLOG DISCUSSES DISEASE PROCESSES. THE INTENTION OF THOSE DISCUSSIONS IS TO CONVEY CURRENT RESEARCH FINDINGS AND OPINIONS, NOT TO GIVE MEDICAL ADVICE. THE INFORMATION IN POSTS IN THIS BLOG IS NOT A SUBSTITUTE FOR A LICENSED PHYSICIAN’S MEDICAL ADVICE. IF ANY ADVICE, OPINIONS, OR INSTRUCTIONS HEREIN CONFLICT WITH THAT OF A TREATING LICENSED PHYSICIAN, DEFER TO THE OPINION OF THE PHYSICIAN. THIS INFORMATION IS INTENDED FOR PEOPLE IN GOOD HEALTH. IT IS THE READER’S RESPONSIBILITY TO KNOW HIS OR HER MEDICAL HISTORY AND ENSURE THAT ACTIONS OR SUPPLEMENTS HE OR SHE TAKES DO NOT CREATE AN ADVERSE REACTION.


Pingback: Important recent research on cancer stem cells in lung cancers | AGING SCIENCES – Anti-Aging Firewalls