By Vince Giuliano
Longevity is the art of not dying. This art in turn draws heavily on the various arts of staying well. Chronic rhinosinusitis (CRS), though not usually a life-threatening illness in itself, can not only compromise quality of life but also affect the immune system and other body systems, invite other illnesses and result in shortened lives. This blog entry cites recent research about the nature of chronic sinusitis, the frequent role of MRSA infections in CRS, the role of microbial biofilms in keeping MRSA and other sinus infections drug-resistant and, finally, how manuka honey might provide a basis for treatment for CRS.
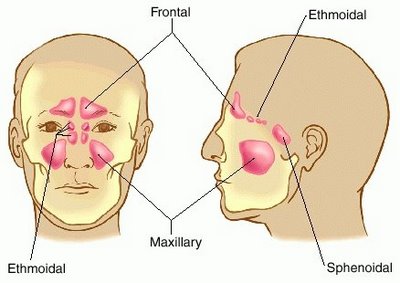
Image of sinuses from Slightlyodd Sinus Facts
About chronic rhinosinusitis
“CRS is now defined as a group of disorders characterized by inflammation of the mucosa of the nose and paranasal sinuses of at least 12 weeks duration. The group of CRS disorders annually accounts as many as 22 million office visits and more than 500,000 emergency department visits in the U.S., according to some estimates. Annual CRS-related healthcare expenditures may reach as much as $3.5 billion(ref).” It is a nasty condition. I know since, having a deviated septum (nose structure), I have been susceptible to it and have had several bouts of CRS in my life. CRS is not a simple disease but describes a complex of possible diseases, sometimes ones difficult to diagnose. It can be caused by bacteria and fungi or be the result of an allergic reaction. It can be caused by a deviated septum which inhibits proper sinus drainage or sinus polyps. Depending on the cause the appropriate treatment can also vary including nasal irrigation, anti-allergy medication, antibiotics, anti-fungal treatments and surgery.
“The word Rhinosinusitis is replacing the term sinusitis because sinusitis is often preceded by rhinitis and rarely occurs without concurrent nasal airway inflammation. Rhinosinusitis has been defined by the American Academy of Otolaryngology as an inflammation of the nose and sinuses. It is believe that this condition comprise a spectrum of inflammatory and infectious diseases. — Medical treatment is the initial treatment choice before opting for surgery in patients who do not improve. Many medical treatments have been recommended or employed. Evidence for their efficacy is rarely strong, partly because of the poor-quality trials in unselected groups of patients2. Chronic rhinosinusitis involves multifactorial etiology. The condition does not respond by simply making an empiric antibiotic selection. There are several predisposing factors in chronic rhinosinusitis which include host factors like allergic rhinitis, viral illness (children in daycare), gastroesophageal reflux, anatomic obstruction, immunodeficiency, genetics, congenital. There are also environmental factors such as irritants (cigarette smoke), microbial (viral, fungal and bacterial) and even medication inducing rhinitis medicamentosa. The quest and identification of factors predisposing to chronic rhinosinusitis is key to guide appropriate management(ref).”
In a great many cases antibiotics have been prescribed for CRS that don’t work. If the situation is serious enough or if there are complicating physical factors, then surgery may be tried.
“The most common indication for sinus surgery is failing medical therapy of chronic sinusitis. Approximately 200,000 U.S. adults undergo sinus surgery per year. Relative indication include persisting obstruction to sinus aeration (e.g., polyp, concha, septum), specific area of recurring disease and chronic or recurrent acute rhinosinusitis who have not responded adequately to medical therapy. Absolute indication include complications like brain abscess, meningitis, subperiosteal abscess, sinus mucocele or pyocele, fungal sinusitis (all varieties), massive polyposis (obstructing sinuses) and neoplasm or suspected neoplasm (causing sinus obstruction)(ref).” Even after surgery serious problems may persist or show up worsened. In fact, research I cite below indicates that bacterial colonization of the sinuses can be worse after surgery.
Patients with CRS are likely to have a diversity of bacteria in their sinuses and nasal passages.
The 2011 publication Characterization of bacterial community diversity in chronic rhinosinusitis infections using novel culture-independent techniques reports: “Background: Chronic rhinosinusitis (CRS) with or without polyps is a common chronic upper airway condition of multifactorial origin. Fundamental to effective treatment of any infection is the ability to accurately characterize the underlying cause. Many studies have shown that only a small fraction of the total range of bacterial species present in CRS is detected through conventional culture-dependent techniques. Consequently, culture data are often unrepresentative of the true diversity of the microbial community within the sample. These drawbacks, along with the length of time required to complete the analysis, strongly support the development of alternative means of assessing which bacterial species are present. As such, molecular microbiological approaches that assess the content of clinical samples in a culture-independent manner could significantly enhance the range and quality of data obtained routinely from such samples. We aimed to characterize the bacterial diversity present in tissue and mucus samples taken from the CRS setting using molecular nonculture-dependent techniques. Methods: Through 16S ribosomal RNA (rRNA) gene clone sequencing and terminal restriction fragment length polymorphism (T-RFLP) analysis, the bacteria present in 70 clinical samples from 43 CRS patients undergoing endoscopic sinus surgery were characterized. Results: Bacterial T-RFLP profiles were generated for 70 of 73 samples and a total of 48 separate bands were detected. Species belonging to 34 genera were identified as present by clone sequence analysis. Of the species detected, those within the genera Pseudomonas, Citrobacter, Haemophilus, Propionibacterium, Staphylococcus, and Streptococcus were found numerically dominant, with Pseudomonas aeruginosa the most frequently detected species. Conclusion: This study has validated the use of the culture-independent technique T-RFLP in sinonasal samples. Preliminary characterization of the microbial diversity in CRS suggests a complex range of common and novel bacterial species within the upper airway in CRS, providing further evidence for the polymicrobial etiology of CRS.”
About bacterial colonization and biofilms in chronic and recurrent rhinosinusitis
“A biofilm is an aggregate of microorganisms in which cells adhere to each other on a surface. These adherent cells are frequently embedded within a self-produced matrix of extracellular polymeric substance (EPS). Biofilm EPS, which is also referred to as slime (although not everything described as slime is a biofilm), is a polymeric conglomeration generally composed of extracellular DNA, proteins, and polysaccharides. Biofilms may form on living or non-living surfaces and can be prevalent in natural, industrial and hospital settings.[1][2] The microbial cells growing in a biofilm are physiologically distinct from planktonic cells of the same organism, which, by contrast, are single-cells that may float or swim in a liquid medium(ref).” The cells in a biofilm form cell colonies and when a pathogen like a MRSA forms a biofilm colony on a human tissue like the wall of a maxillary sinus, the process is referred to as bacterialcolonization.
“Formation of a biofilm begins with the attachment of free-floating microorganisms to a surface. These first colonists adhere to the surface initially through weak, reversible adhesion via van der Waals forces. If the colonists are not immediately separated from the surface, they can anchor themselves more permanently using cell adhesion structures such as pili.[6]— The first colonists facilitate the arrival of other cells by providing more diverse adhesion sites and beginning to build the matrix that holds the biofilm together. Some species are not able to attach to a surface on their own but are often able to anchor themselves to the matrix or directly to earlier colonists. It is during this colonization that the cells are able to communicate via quorum sensing using such products as AHL. Once colonization has begun, the biofilm grows through a combination of cell division and recruitment. The final stage of biofilm formation is known as development, and is the stage in which the biofilm is established and may only change in shape and size. The development of a biofilm may allow for an aggregate cell colony (or colonies) to be increasingly antibiotic resistant(ref).“ Once in an established biofilm colony, pathogenic bacteria are much harder to kill with most antibiotics, and this is thought to be a likely reason why several pathogenic forms of CRS are resistant to antibiotic therapy.
A particular bacteria often found in patients with chromic rhinosinusitis which forms biofilms is MRSA, e.g. Methicillin-resistant S. aureus. “Staphylococcus aureus (/ˌstæfɨlɵˈkɒkəsˈɔriəs/) is a bacterial species named from Greekσταφυλόκοκκος meaning the “golden grape-cluster berry”. Also known as “golden staph” and Oro staphira, it is a facultative anaerobicGram-positivecoccalbacterium. It is frequently found as part of the normal skin flora on the skin and nasal passages.[1] It is estimated that 20% of the human population are long-term carriers of S. aureus.[1]S. aureus is the most common species of staphylococcus to cause Staph infections. The reason S. aureus is a successful pathogen is a combination of bacterial immuno-evasive strategies. One of these strategies is the production of carotenoidpigmentstaphyloxanthin, which is responsible for the characteristic golden colour of S. aureus colonies. This pigment acts as a virulence factor, primarily by being a bacterial antioxidant which helps the microbe evade the reactive oxygen species which the host immune system uses to kill pathogens.[2][3] MRSA –is one of a number of greatly-feared strains of S. aureus which have become resistant to most antibiotics. MRSA strains are most often found associated with institutions such as hospitals, but are becoming increasingly prevalent in community-acquired infections(ref).”
As will emerge in the discussion below, MRSA is not the only biofilm-generating pathogen found in chronic rhinosinusitis, Another commonly occuring pathogen that forms biofilms in CRS is pseudomonas aeruginosa. Bacteria resistant to antibiotics, which may involve other mechanisms in addition to formation of biofilms, sometimes called superbugs, include 3.1 Staphylococcus aureus, 3.2 Streptococcus and Enterococcus, 3.3 Pseudomonas aeruginosa, 3.4 Clostridium difficile, 3.5 Salmonella and E. coli, 3.6 Acinetobacter baumannii and 3.7 Mycobacterium tuberculosis (ref).
Biofilms of pathogens such as S, aureus and pseudomonas aeruginosa have effective ways to protect the bacteria from antibiotics.
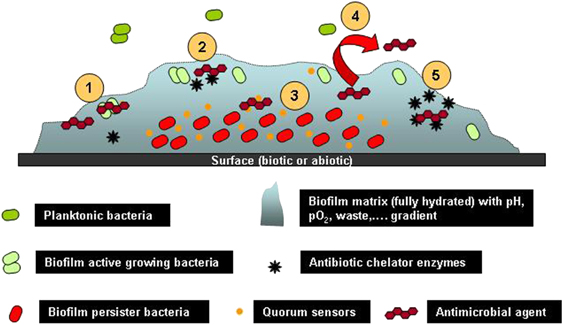
Image and following passage from The Challenge of Treating Biofilm-associated Bacterial Infections J L del Pozo and R Patel, as reported in Nature.com: “Some proposed-biofilm associated resistance mechanisms: (1) Antimicrobial agents may fail to penetrate beyond the surface layers of the biofilm. Outer layers of biofilm cells absorb damage. Antimicrobial agents action may be impaired in areas of waste accumulation or altered environment (pH, pCO2, pO2, etc). (2) Antimicrobial agents may be trapped and destroyed by enzymes in the biofilm matrix. (3) Altered growth rate inside the biofilm. Antimicrobial agents may not be active against nongrowing microorganisms (persister cells). (4) Expression of biofilm-specific resistance genes (e.g., efflux pumps). (5) Stress response to hostile environmental conditions (e.g., leading to an overexpression of antimicrobial agent-destroying enzymes).”
The existence of bacterial biofilms has been known for a long time but their roles in drug resistance have been explored only recently. The newer studies of bacterial colonization and biofilms appears to lend a new dimension of understanding of CRS and potential treatments for it. By about 2010 is was starting to be widely recognized that if biofilms are indeed a factor responsible for antibiotic resistance in CRS, then whole new forms of therapy for CRS might be developed based on dissolving the biofilms.
Starting over 6 years ago, researchers have implicated bacterial biofilms as culprits in maintaining drug resistance in chronic rhinosinusitis.
The 2006 publication Bacterial biofilms in chronic rhinosinusitis reported: “Chronic sinusitis is a prevalent, debilitating condition, and a subpopulation of patients fails to respond to either medical or surgical intervention. Bacterial biofilms are 3-dimensional aggregates of bacteria that have special properties due to their group structure, including increased resistance to antibiotics in some forms. They have been shown to play a major role in many chronic infections, including cystic fibrosis, endocarditis, and otitis media. Evidence now suggests that they may play an important role in chronic sinusitis. Our laboratory has identified the presence of biofilms in sinonasal mucosa isolated from human patients and on stents removed after frontal sinus surgery. In addition, biofilms have been found on the sinus epithelium of rabbits infected with Pseudomonas aeruginosa, but not in rabbits infected with non-biofilm-forming P. aeruginosa mutants. This animal model can provide opportunities to address the functional significance of biofilm production in the sinus cavities. A further understanding of the role of bacterial biofilms may lead to the development of more appropriate therapies for the treatment and prevention of chronic sinusitis.”
The 2010 publication Biofilms reported: “Bacterial biofilms are 3-dimensional aggregates of bacteria that have been shown to play a major role in many chronic infections. Evidence is growing that bacterial biofilms may play a role in certain cases of recalcitrant chronic sinusitis that do not respond to traditional medical and surgical therapies. Novel therapies may have clinical applications to prevent and destabilize biofilms. Future research will determine if topical antimicrobials, surfactants, and other adjuvant therapies can be used to treat biofilm-associated chronic rhinosinusitis.”
The 2011 review publication Bacterial biofilms and the pathophysiology of chronic rhinosinusitis reported: “Purpose Of Review: To review the evidence for the presence of bacterial biofilms in chronic rhinosinusitis (CRS) and mechanisms by which they may contribute to the chronic inflammation characteristic of this disease. Lastly, to provide an overview of the current and potential future treatments for bacterial biofilms in CRS. Recent Findings: Advances in the techniques for identifying biofilms have confirmed the presence of bacterial biofilms on the sinonasal mucosa of patients with CRS. The impact on mucosal inflammation of the polymicrobial or multiorganism milieu is not yet well understood. Numerous novel topical therapies for the treatment of bacterial biofilms in CRS have been suggested with some demonstrating clinical efficacy. Blocking of quorum sensing represents a potential future therapy for biofilm treatment in CRS and biofilm infection at large. Summary: Biofilms represent an important influence on the pathophysiology of CRS. Further understanding of biofilm interactions and microbial organism behavior will provide us with future treatment modalities for this disease.”
A number of studies have looked at the frequency of occurrence of biofilm-forming bacteria in CRS.
The 2008 publication Prevalence of biofilm-forming bacteria in chronic rhinosinusitis relates: “Background: Recently, biofilms have been implicated in the pathogenesis of recalcitrant chronic rhinosinusitis (CRS). We sought to determine the prevalence of biofilm-forming cultures obtained from patients with CRS and clinical factors that may contribute to biofilm formation. Methods: Endoscopically guided sinonasal cultures were obtained in duplicate from CRS patients with evidence of mucopurulence. Bacterial swabs were sent for microbiological characterization and were simultaneously evaluated for biofilm-forming capacity by a modified Calgary Biofilm Detection Assay. Biofilm formation was based on concomitant values of biofilm-forming Pseudomonas aeruginosa O1 (PAO1) (positive control) and non-biofilm-forming mutants sad-31 (type IV pili) and sad-36 (flagella K; negative control). Samples, with growth greater than the sad-31 mutant, were designated as biofilm formers. Results: Sinonasal cultures were obtained from 157 consecutive patients (83 female patients) over a 4-month period. Forty-five samples (28.6%) showed biofilm formation. Among patients with a prior history of functional endoscopic sinus surgery (FESS), 30.7% (n = 42) showed biofilm growth. For patients naive to surgical intervention (n = 20), only 15% showed biofilm formation. A positive, statistically significant correlation existed between biofilm formation and number of prior FESS procedures. Polymicrobial cultures, Pseudomonas aeruginosa, and/or Staphylococcus aureus comprised 71% of samples. Chi-squared analysis showed an association with prior infections, but not with any pharmacologic therapy or comorbidies. Conclusion: We show a high percentage of CRS patients (28.6%) whose sinonasal mucopurulence has biofilm-forming capacity. Postsurgical patients had a high prevalence of biofilm-forming bacteria, a possible reflection of the severe nature of their disease. Additional studies are warranted.”
See also the 2012 publication Role of bacterial and fungal biofilms in chronicrhinosinusitis and the 2011 publication Are biofilms associated with an inflammatory response in chronic rhinosinusitis? The new April 2012 publication Prevalence of Biofilms and Their Response to Medical Treatment in ChronicRhinosinusitis without Polypsreports: “Objective: The aim of this study was to investigate the prevalence of biofilms and the effects of medical treatment modalities in chronicrhinosinusitis (CRS) patients without nasal polyps. Study Design:. Randomized controlled trial. Settings. Tertiary referral hospital. Subjects and Methods. The authors randomly divided 32 adult patients with CRS without nasal polyps into 2 groups. — . Results. Biofilms were detected in 24 of 32 patients (75%) before the treatment (grades 1-3). Biofilms were detected in 14 of 32 patients (43.8%) after the treatment (grades 1-2). When each group was evaluated independently, there was a significant improvement after the treatment in both groups I and II. When the biofilm grades of group I were compared to those of group II, there was no significant difference both in the pre- and posttreatment evaluation. Conclusion. The prevalence of biofilms in CRS without polyps was 75% in our study. Regression of biofilms to 43% was observed under medical treatment. Adding nasal steroids to macrolides gave no further benefit.”
The 2008 publication Characterization of bacterial and fungal biofilms in chronic rhinosinusitis reports: “Background: Conclusive evidence exists that biofilms are present on the mucosa of chronic rhinosinusitis (CRS) patients. Less is known about the species constituting these biofilms. This study developed a fluorescence in situ hybridization (FISH) protocol for characterization of bacterial and fungal biofilms in CRS. Methods: Fifty CRS patients and 10 controls were recruited. Bacteria FISH probes for Staphylococcus aureus, Haemophilus influenzae, and Pseudomonas aeruginosa and a universal probe for fungi were applied to sinus mucosal specimens and then analyzed using confocal scanning laser microscopy. Results: Thirty-six of 50 CRS patients had biofilms present in contrast to 0/10 controls, suggesting a role for biofilms in the pathogenesis of this disease. S. aureus was the most common biofilm-forming organism. Eleven of 50 CRS patients had characteristic fungal biofilms present. Conclusion: This is the largest study of biofilms in CRS. It has validated mucosal tissue cryopreservation for delayed biofilm analysis. Fungal biofilms have been identified and the importance of S. aureus biofilms in the polymicrobial etiology of CRS is highlighted.”
The 2010 publication Evidence of bacterial biofilms in nasal polyposis looked at patients with chronic rhinosinusitis and nasal polyps. “Introduction: The pathogeny of chronic rhinosinusitis with nasal polyposis (CRS/NP) has not been elucidated. Bacterial exotoxins have been implicated in many inflammatory chronic diseases, such as chronic otitis, chronic tonsillitis, cholesteatomas, and more recently CRS/NP. We propose that the bacteria in CRS/NP are not only present in a planktonic state, but also occur in microbial communities as biofilms. Objective: To determine and characterize the presence of biofilms in CRS/NP. Methods: We performed a prospective study in 12 patients undergoing endoscopic sinus surgery for nasal polyposis. Ten patients without CRS/NP who underwent septoplasty were included as a control group. Tissue samples were obtained from the inferior turbinate mucosae. The bacteria were isolated and typified and the material was examined in vitro using a spectrophotometer, and in vivo using optical microscopy and confocal scanning laser microscopy. Results: Moderate to high in vitro biofilm-forming capacity was detected in 9 out of 12 patients with CRS/NP (mean [SD] optical density values of between 0.284 [0.017] and 3.337 [0.029]). The microorganisms isolated were Staphylococcus (5 patients), Streptococcus viridans, Pseudomonas aeruginosa, Enterococcus faecalis and Streptococcus viridans/Corynebacterium. Biofilms were demonstrated in vivo in 2 patients and no biofilm structures were evident in any of the controls. Conclusion: This study demonstrates the presence of bacterial biofilms in patients with CRS/NP. This chronic inflammatory factor might contribute to nasal mucosa damage, increased inflammatory cells in tissue, and the subsequent hyperplasic process.”
The 2011 publication Clinical factors associated with bacterial biofilm formation in chronic rhinosinusitis reports “Objectives: Bacterial biofilms appear to contribute to chronic rhinosinusitis. However, the mechanism behind biofilm formation in chronic rhinosinusitis remains poorly defined. The aim of this study is to evaluate clinical factors that may be associated with bacterial biofilm formation in chronic rhinosinusitis. Study design: Cross-sectional study: Setting: Department of Otorhinolaryngology-Head and Neck Surgery at the Hospital of the University of Pennsylvania. Subjects And Methods: Five hundred eighteen patients with chronic rhinosinusitis were enrolled from 2007 to 2010. Samples were taken to evaluate for biofilm formation in vitro using a modified Calgary Biofilm Detection Assay. Clinical data were collected from chart review. Pearson’s χ(2) and logistic regression were used for the analyses. Results: Of the patients, 108 (20.9%) showed biofilm formation in vitro. Bacterial biofilm formation in vitro was not significantly associated with polyps, allergy, Samter’s triad, sleep apnea, smoking status, age, or gender. However, it was significantly associated with positive culture results (odds ratio [OR] = 3.13; 95% confidence interval [CI], 1.85-5.29; P < .001), prior sinus surgeries (1.93; 1.01-3.69; P = .046), and nasal steroid use in the month prior to sample collection (2.09; 1.07-4.08; P = .030). Polymicrobial cultures, Pseudomonas aeruginosa, and Staphylococcus aureus comprised most of the samples. Conclusion: The results of this study suggest that the probability of bacterial biofilm formation is independent of many clinical factors considered to be risk factors for chronic rhinosinusitis. Further studies are needed to clarify the nature of the associations between prior sinus surgeries, nasal steroid use, and biofilm formation.”
Bacterial biofilm is generally associated with a negative impact on prognosis in chronic rhinosinusitis.
The Nov-Dec 2011 publication Factors affecting bacterial biofilm expression in chronic rhinosinusitis and the influences on prognosis reports: “Purpose: The purpose of the study was to investigate the expression of bacterial biofilm (BF) for chronicrhinosinusitis (CRS) and to find out factors affecting BF expression and the influences on its prognosis. Materials And Methods: All specimens were analyzed by a scanning electron microscope and bacterial cultivation from a panel of 93 patients with CRS, 20 with nasal septum deviation, and 17 with nasal bone fractures as the control group. Referring to the grade classification criteria from the sinusitis-specific questionnaire Sino-Nasal Outcome Test-20, patients were assessed preoperatively based on common clinical manifestations. Patients were followed up for condition improvement as assessed by visual analogue scale and nasal endoscopic examination. Results: In the experimental group, among all the patients, 3 were lost (lost 3.2%). Bacterial biofilm was positive in 64 (71.1%) of 90 patients. Mucosal cilia were observed in varying degrees of injury. Bacterial culture was positive in 60 (66.7%) patients. In the control group, no BF was found and no bacterium was cultured. Bacterial biofilm expression was correlated with the bacterial culture. The BF(+) patients’ visual analogue scale scores 6 months and 1 year postoperation were lower than the BF(-) patients’ scores (P < .05). The BF(+) patients’ Lund-Kennedy scores at 6 months and 1 year postoperation were higher than the BF(-) patients’ scores (P < .05). Conclusions: Bacterial biofilm is involved in the pathogenesis of CRS and is associated with the bacterial culture. Bacterial biofilm has a certain impact on patients’ prognosis.”
In chronic rhinosinusitis, biofilm formation likely represents the latter phase of an inflammatory process that leads to complete epithelial destruction.
The 2008 publication Damage to ciliated epithelium in chronic rhinosinusitis: what is the role of bacterial biofilms? reports: “Objectives: We assess the association between the presence of biofilms and cilial damage in patients with chronic rhinosinusitis (CRS), describe the microorganisms associated with samples that exhibited cilial loss and biofilms, and demonstrate the absence of ciliary injury and biofilms in similarly prepared “normal” controls. Methods: We examined samples of ethmoid mucosa obtained from 24 patients who underwent functional endoscopic sinus surgery for CRS. Samples from a control group (20 healthy subjects) were also examined. The specimens were divided into 2 fragments; the first was processed for bacterial cultures, and the second was subjected to scanning electron microscopy. Statistical analysis was performed. Results: All CRS samples had positive bacterial cultures. The scanning electron microscopy analysis showed bacterial biofilms in 10 of the 24 specimens. A marked destruction of the epithelium was observed in samples positive for biofilms (p < 0.001), and the presence of Haemophilus influenzae was associated with ciliary abnormalities (partial damage in 55.6% and absence of cilia in 50%; p = 0.041). Conclusions: The high percentage of biofilms in our specimens confirms the association between biofilms and CRS. Our data support the hypothesis that biofilm formation represents the latter phase of an inflammatory process that leads to complete epithelial destruction.”
Endoscopic sinus surgery can have a negative outcome because of persistence of staphylococcus aureus biofilms.
The July 2011 publication Staphylococcus aureus biofilms: Nemesis of endoscopic sinus surgery reports: “Chronic rhinosinusitis (CRS) patients with biofilms have persistent postoperative symptoms, ongoing mucosal inflammation, and recurrent infections. Recent evidence suggests that biofilms of differing species confer varying disease profiles in CRS patients. We aimed to prospectively investigate the effects of Staphylococcus aureus, Pseudomonas aeruginosa, Haemophilus influenzae, and fungal biofilms on outcomes following endoscopic sinus surgery (ESS). Study Design: Prospective blinded study. Methods: In this prospective blinded study, 39 patients undergoing ESS for CRS assessed their symptoms preoperatively using internationally accepted standardized symptom scoring systems and quality-of-life measures (10-point visual analog scale, Sino-Nasal Outcome Test-20, global severity of CRS). Their sinonasal mucosa was graded (Lund-Kennedy scale) and extent of radiologic disease on computed tomography scans scored (Lund-McKay scale). Random sinonasal tissue samples were assessed for different bacterial species forming biofilms by using fluorescent in-situ hybridization and confocal laser microscopy. For 12 months after surgery, CRS symptoms, quality of life, and objective evidence of persisting disease were assessed by using the preoperative tools. Results: Different bacterial species combinations were found in 30 of 39 patients; 60% of these 30 biofilms were polymicrobial biofilms and 70% had S aureus biofilms. Preoperative nasendoscopy and radiologic disease severity were significantly worse in patients with multiple biofilms (P = .02 and P = .01, respectively), and they had worse postsurgery mucosal outcomes on endoscopy (P = .01) requiring significantly more postoperative visits (P = .04). Those with S aureus biofilms progressed poorly with their symptom scores and quality-of-life outcomes, with significant differences in nasendoscopy scores (P = .007). Conclusions: S. aureus biofilms play a dominant role in negatively affecting outcomes of ESS with persisting postoperative symptoms, ongoing mucosal inflammation, and infections.”
It sounds like many patients with S. aureus biofilms to start still had them after endoscopic sinus surgery and were not very well off. Starting 2-3 years ago, there was a growing feeling that perhaps the best thing for patients with such biofilms would be to get rid them in the first place.
In chronic rhinosinusitis when there are S. aureus biofilms, it appears that there is a systematic shift in the adaptive immune response. The T cell response is skewed toward the T-helper(2) pathway.
This skewing has been noted in several publications. The November 2011 publication Adaptive immune responses in Staphylococcus aureus biofilm-associated chronic rhinosinusitis reports: “Background: The etiopathogenesis of chronic rhinosinusitis (CRS) is currently an area of intense debate. Recently, biofilms have been proposed as a potential environmental trigger in this disease. In particular, Staphylococcus aureus biofilms appear to be a predictor of severe disease recalcitrant to current treatment paradigms. However, direct causal links between biofilms and host immune activation are currently lacking. This study aimed to document both the adaptive immune responses that characterize S. aureus biofilm-associated CRS and the relative contributions of staphylococcal superantigens and S. aureus biofilms in the inflammatory make-up of this disease. Methods: total of 53 disease subjects and 15 controls were recruited. Sinonasal mucosa was collected for the determination of S. aureus and Haemophilus influenzae biofilms and presence of total and superantigen-specific IgE and for the measurement of cytokines that characterize the T-helper pathways. Results: Staphylococcus aureus biofilms and superantigens are significantly associated in CRS patients, suggesting the biofilm may be a nidus for superantigen-eluting bacteria. The presence of S. aureus biofilms is associated with eosinophilic inflammation, across the spectrum of CRS, on the back of a T-helper(2) skewing of the host’s adaptive immune response (elevated Eosinophilic Cationic Protein and IL-5). This can be distinguished from the superantigenic effect resulting in the induction of IgE. Conclusion: This study provides novel evidence of a link between S. aureus biofilms and skewing of the T-cell response toward the T-helper(2) pathway that is independent of superantigen activities. Further research is required to confirm the cause-effect relationship of this association.”
Manuka honey can dissolve biofilms created by S. aureus and pseudomonas aeruginosa and kill those microbes, including MRSA. It might be the basis for a topical irrigation treatment for chronic rhinosinusitis.
My first reaction when I heard about honey treatment for chronic rhinosinusitis was “This sounds like another new-age treatment and shady marketing pitch.” But I found an impressive amount of research which backs up the above statements. I will lay that research out here. The studies are all by otolaryngology physicians and researchers situated in Western hospital and university research institutions, many in Canada and Australia.
The antimicrobial properties of manuka honey are attributed to the nature of the nectar obtained by the bees from manuka flowers which grows on manuka bushes, a scrub species that grows only in New Zeeland. The Maori natives of New Zeeland used parts of the plant as natural medicine(ref). It is also known as the Tea Tree.
Manuka tree and flowers (source) Bee on manuka flower (source)
The 2009 publication Effectiveness of honey on Staphylococcus aureus and Pseudomonas aeruginosa biofilms reports: “Objectives: Biofilms formed by Pseudomonas aeruginosa (PA) and Staphylococcusaureus (SA) have been shown to be an important factor in the pathophysiology of chronic rhinosinusitis (CRS). As well, honey has been used as an effective topical antimicrobial agent for years. Our objective is to determine the in vitro effect of honey against biofilms produced by PA and SA. Study Design: In vitro testing of honey against bacterial biofilms. Methods: We used a previously established biofilm model to assess antibacterial activity of honey against 11 methicillin-susceptible SA (MSSA), 11 methicillin-resistant SA (MRSA), and 11 PA isolates. Honeys were tested against both planktonic and biofilm-grown bacteria. Results: Honey was effective in killing 100 percent of the isolates in the planktonic form. The bactericidal rates for the Sidr and Manuka honeys against MSSA, MRSA, and PA biofilms were 63-82 percent, 73-63 percent, and 91-91 percent, respectively. These rates were significantly higher (P<0.001) than those seen with single antibiotics commonly used against SA.” Conclusion: Honey, which is a natural, nontoxic, and inexpensive product, is effective in killing SA and PA bacterial biofilms. This intriguing observation may have important clinical implications and could lead to a new approach for treating refractory CRS.” My reaction was “Wow, honey is more effective than antibiotics for killing S aureus?”
Manuka honey can inhibit cell division of MRSA bacteria.
The November 2011 publication Manuka honey inhibits cell division in methicillin-resistant Staphylococcus aureus reports: “Objectives: The aim of this study was to investigate the effect of manuka honey, artificial honey and an antibacterial component (methylglyoxal) on cell division in methicillin-resistant Staphylococcus aureus (MRSA). Methods: Viability of epidemic MRSA-15 NCTC 13142 incubated with manuka honey, artificial honey and methylglyoxal was determined, and structural effects monitored by electron microscopy. Activity of murein hydrolase (a peptidoglycan-degrading enzyme implicated in cell separation, encoded by atl) was estimated by cell wall hydrolysis and zymography; expression of atl was quantified by real-time PCR. Results: Growth of MRSA was inhibited by 5%, 10% and 20% (w/v) manuka honey and 10% (w/v) artificial honey containing methylglyoxal, but not 10% (w/v) artificial honey. Statistically significantly increased numbers of cells containing septa and increased cell diameter (P < 0.001 and P < 0.001, respectively) were found in MRSA exposed to 5%, 10% or 20% (w/v) manuka honey, but not 10% (w/v) artificial honey with and without methylglyoxal. Intracellular activity of murein hydrolase was elevated in MRSA grown in 10% (w/v) artificial honey and at undetectable levels in MRSA treated with 10% (w/v) manuka honey. Increased atl expression was found in MRSA treated with 10% (w/v) manuka honey and 10% artificial honey containing methylglyoxal. Conclusions: Enlarged cells containing septa were observed in MRSA exposed to inhibitory concentrations of manuka honey, suggesting that cell division was interrupted. These changes were not caused by either the sugars or methylglyoxal in honey and indicate the presence of additional antibacterial components in manuka honey.”
The most active anti-microbial ingredient in manuka honey appears to be Methylglyoxal.
The 2008 publication Identification and quantification of methylglyoxal as the dominant antibacterial constituent of Manuka (Leptospermum scoparium) honeys from New Zealandreports: “The 1,2-dicarbonyl compounds 3-deoxyglucosulose (3-DG), glyoxal (GO), and methylglyoxal (MGO) were measured as the corresponding quinoxalines after derivatization with orthophenylendiamine using RP-HPLC and UV-detection in commercially available honey samples. Whereas for most of the samples values for 3-DG, MGO, and GO were comparable to previously published data, for six samples of New Zealand Manuka (Leptospermum scoparium) honey very high amounts of MGO were found, ranging from 38 to 761 mg/kg, which is up to 100-fold higher compared to conventional honeys. MGO was unambigously identified as the corresponding quinoxaline via photodiodearry detection as well as by means of mass spectroscopy. Antibacterial activity of honey and solutions of 1,2-dicarbonyl towards Escherichia coli (E. coli) and Staphylococcus aureus (S. aureus) were analyzed using an agar well diffusion assay. Minimum concentrations needed for inhibition of bacterial growth (minimum inhibitory concentration, MIC) of MGO were 1.1 mM for both types of bacteria. MIC for GO was 6.9 mM (E. coli) or 4.3 mM (S. aureus), respectively. 3-DG showed no inhibition in concentrations up to 60 mM. Whereas most of the honey samples investigated showed no inhibition in dilutions of 80% (v/v with water) or below, the samples of Manuka honey exhibited antibacterial activity when diluted to 15-30%, which corresponded to MGO concentrations of 1.1-1.8 mM. This clearly demonstrates that the pronounced antibacterial activity of New Zealand Manuka honey directly originates from MGO.”
Other studies have also fingured methylglyoxal as the active ingredient in manuka honey including: Isolation by HPLC and characterisation of the bioactive fraction of New Zealand manuka (Leptospermum scoparium) honey (2008) and (2009) The origin of methylglyoxal in New Zealand manuka (Leptospermum scoparium) honey, which reports “Methylglyoxal in New Zealand manuka honey has been shown to originate from dihydroxyacetone, which is present in the nectar of manuka flowers in varying amounts. Manuka honey, which was freshly produced by bees, contained low levels of methylglyoxal and high levels of dihydroxyacetone. Storage of these honeys at 37 degrees C led to a decrease in the dihydroxyacetone content and a related increase in methylglyoxal. Addition of dihydroxyacetone to clover honey followed by incubation resulted in methylglyoxal levels similar to those found in manuka honey. Nectar washed from manuka flowers contained high levels of dihydroxyacetone and no detectable methylglyoxal.”
Methylglyoxal is is an effective antimicrobial agent against both planktonic and biofilm MRSA and PA organisms in vitro.
The September 2011 publication Methylglyoxal: (active agent of manuka honey) in vitro activity against bacterial biofilms reports: “Background: Pseudomonas aeruginosa (PA) and Staphylococcus aureus (SA) biofilms are associated with poor chronic rhinosinusitis (CRS) disease control following surgery. Manuka honey (MH) has been shown to be both an effective in vitro treatment agent for SA and PA biofilms and nontoxic to sinonasal respiratory mucosa. Methylglyoxal (MGO) has been reported to be the major antibacterial agent in MH. The effect of this agent against SA and PA biofilms has yet to be reported. Our objective was to determine the in vitro effect of MGO against biofilms of SA and PA, via in vitro testing of MGO against bacterial biofilms. Methods: An established biofilm model was used to determine the effective concentration (EC) of MGO against 10 isolates of methicillin-resistant SA (MRSA) and PA. The EC of MGO was also determined against planktonic (free-swimming) MRSA and PA. Results: For MRSA, the EC against planktonic organisms was a concentration of 0.08 mg/mL to 0.3 mg/mL whereas against the biofilm MRSA isolates, the EC ranged from 0.5 mg/mL to 3.6 mg/mL. For PA, the EC against planktonic organisms was a concentration of 0.15 mg/mL to 1.2 mg/mL for planktonic organisms whereas against the biofilm PA isolates, the EC ranged from 1.8 mg/mL to 7.3 mg/mL. Conclusion: MGO, a component of MH, is an effective antimicrobial agent against both planktonic and biofilm MRSA and PA organisms in vitro.”
There are other antibiotic substances in manuka honey besides Methylglyoxal and other medicinal honeys.
The 2011 publication Two major medicinal honeys have different mechanisms of bactericidal activity reports: “Honey is increasingly valued for its antibacterial activity, but knowledge regarding the mechanism of action is still incomplete. We assessed the bactericidal activity and mechanism of action of Revamil® source (RS) honey and manuka honey, the sources of two major medical-grade honeys. RS honey killed Bacillus subtilis, Escherichia coli and Pseudomonas aeruginosa within 2 hours, whereas manuka honey had such rapid activity only against B. subtilis. After 24 hours of incubation, both honeys killed all tested bacteria, including methicillin-resistant Staphylococcus aureus, but manuka honey retained activity up to higher dilutions than RS honey. Bee defensin-1 and H₂O₂ were the major factors involved in rapid bactericidal activity of RS honey. These factors were absent in manuka honey, but this honey contained 44-fold higher concentrations of methylglyoxal than RS honey. Methylglyoxal was a major bactericidal factor in manuka honey, but after neutralization of this compound manuka honey retained bactericidal activity due to several unknown factors. RS and manuka honey have highly distinct compositions of bactericidal factors, resulting in large differences in bactericidal activity.”
Manuka honey appears capable of sensitizing MRSA to antibiotics to which the MRSA is otherwise resistant.
At least, this appears to be the case for oxacillin in-vitro. The new March 2012 e-publication Synergy between oxacillin and manuka honey sensitizes methicillin-resistant Staphylococcus aureus to oxacillin reports: “Objectives: Honey is an ancient wound remedy that has recently been introduced into modern clinical practice in developed countries. Manuka honey inhibits growth of methicillin-resistant Staphylococcus aureus (MRSA) by preventing cell division. In Gram-negative bacteria a synergistic interaction between honey and antibiotics has been suggested. We aimed to determine the effect of manuka honey on oxacillin resistance in MRSA. Methods: Inhibition of MRSA by manuka honey and oxacillin separately and in combination was tested by disc diffusion, Etest strips, serial broth dilution, chequerboards and growth curves. Results: Manuka honey and oxacillin interacted synergistically to inhibit MRSA. Manuka honey reversed oxacillin resistance in MRSA, and down-regulation of mecR1 was found in cells treated with manuka honey. Conclusions: Microarray analysis showed that exposure of MRSA to inhibitory concentrations of manuka honey resulted in down-regulation of mecR1. Here we demonstrated that subinhibitory concentrations of honey in combination with oxacillin restored oxacillin susceptibility to MRSA. Other honey and antibiotic combinations must now be evaluated.”
Methylglyoxal also appears to be the substance or one of the substances that breaks up S. aureus biofilms.
The 2011 publication Methylglyoxal-infused honey mimics the anti-Staphylococcus aureus biofilm activity of manuka honey: potential implication in chronic rhinosinusitis .reports: “Objectives/Hypothesis: Low pH, hydrogen peroxide generation, and the hyperosmolarity mechanisms of antimicrobial action are ubiquitous for all honeys. In addition, manuka honey has been shown to contain high concentrations of methylglyoxal (MGO), contributing the relatively superior antimicrobial activity of manuka honey compared to non-MGO honeys. In high concentrations, manuka honey is effective in killing Staphylococcus aureus biofilms in vitro. Lower concentrations of honey, however, are desirable for clinical use as a topical rinse in chronic rhinosinusitis in order to maximize the tolerability and practicality of the delivery technique. This study, therefore, was designed to evaluate the contribution of MGO to the biofilm-cidal activity of manuka honey, and furthermore determine whether the antibiofilm activity of low-dose honey can be augmented by the addition of exogenous MGO. Study Design: In vitro microbiology experiment. Methods: Five S. aureus strains (four clinical isolates and one reference strain) were incubated to form biofilms using a previously established in vitro dynamic peg model. First, the biofilm-cidal activities of 1) manuka honey (790 mg/kg MGO), 2) non-MGO honey supplemented with 790 mg/kg MGO, and 3) MGO-only solutions were assessed. Second, the experiment was repeated using honey solutions supplemented with sufficient MGO to achieve concentrations exceeding those seen in commercially available manuka honey preparations. Results: All honey solutions containing a MGO concentration of 0.53 mg/mL or greater demonstrated biofilm-cidal activity; equivalent activity was achieved with ≥1.05 mg/mL MGO solution. CONCLUSIONS: MGO is only partially responsible for the antibiofilm activity of manuka honey. Infusion of MGO-negative honey with MGO, however, achieves similar cidality to the equivalent MGO-rich manuka honey. Conclusions: MGO is only partially responsible for the antibiofilm activity of manuka honey. Infusion of MGO-negative honey with MGO, however, achieves similar cidality to the equivalent MGO-rich manuka honey.”
Besides inhibiting cell division and penetrating biofilms to fight MRSA infections, another way that manuka honey appears to combat MRSA is downregulating expression of universal stress protein A.
The 2011 publication Effect of manuka honey on the expression of universal stress protein A in meticillin-resistant Staphylococcus aureus reports: “Staphylococcus aureus is an important pathogen that can cause many problems, from impetigo to endocarditis. With its continued resistance to multiple antibiotics, S. aureus remains a serious health threat. Honey has been used to eradicate meticillin-resistant S. aureus (MRSA) strains from wounds, but its mode of action is not yet understood. — Proteomics provides a potent group of techniques that can be used to analyse differences in protein expression between untreated bacterial cells and those treated with inhibitory concentrations of manuka honey. In this study, two-dimensional (2D) electrophoresis was combined with matrix-assisted laser desorption/ionisation time-of-flight mass spectrometry (MALDI-TOF MS) to determine the identities of proteins whose levels of expression were changed at least two-fold following treatment with manuka honey. Protein extracts were obtained from cells grown in tryptone soy broth (with or without manuka honey) by mechanical disruption and were separated on 2D polyacrylamide gels. A protein was isolated in gels prepared from untreated cell extract that was absent from gels made using honey-treated cell extract. Using MALDI-TOF MS, the protein was identified as universal stress protein A (UspA). Downregulation of this protein was confirmed by real-time polymerase chain reaction (PCR), which showed a 16-fold downregulation in honey-treated cells compared with untreated samples. This protein is involved in the stress stamina response and its downregulation could help to explain the inhibition of MRSA by manuka honey.”
As of 2010 least, it appears that pathogen bacteria have not built up resistance to manuka honey.
The 2010 publication Absence of bacterial resistance to medical-grade manuka honey reports: “Clinical use of honey in the topical treatment of wounds has increased in Europe and North America since licensed wound care products became available in 2004 and 2007, respectively. Honey-resistant bacteria have not been isolated from wounds, but there is a need to investigate whether honey has the potential to select for honey resistance. Two cultures of bacteria from reference collections (Staphylococcusaureus NCTC 10017 and Pseudomonas aeruginosa ATCC 27853) and four cultures isolated from wounds (Escherichia coli, methicillin-resistant S. aureus (MRSA), Pseudomonas aeruginosa and S. epidermidis) were exposed to sub-lethal concentrations of manuka honey in continuous and stepwise training experiments to determine whether the susceptibility to honey diminished. Reduced susceptibilities to manuka honey in the test organisms during long-term stepwise resistance training were found, but these changes were not permanent and honey-resistant mutants were not detected. The risk of bacteria acquiring resistance to honey will be low if high concentrations are maintained clinically.”
Manuka honey may also be an effective therapy for fungal rhinosinusitis.
The 2011 publication Single-blind study of manuka honey in allergic fungal rhinosinusitis reports: “Background: Some patients continue to suffer from symptoms of sinusitis after maximal topical medical and surgical treatment for allergic fungal rhinosinusitis (AFRS). Manuka honey has well-documented antimicrobial and antifungal properties and is currently being used by physicians across the world for a wide variety of medical problems. Objective: This study aimed to determine the effectiveness of Medihoney Antibacterial Medical Honey in patients who continue to suffer from AFRS resistant to conventional medical treatment after bilateral functional endoscopic sinus surgery and maximal postoperative medical management. Methods: A randomized, single-blind, prospective study was conducted at a tertiary centre. Thirty-four patients with AFRS sprayed one nostril with 2 mL of a 50/50 mixture of honey-saline solution once a day at night for 30 days. Otherwise, patients continued with their regular nasal regimen in both nostrils. A 5-point improvement in our clinic’s endoscopic grading system was considered significant. During their pre- and postassessment, patients’ sinus cavities were cultured, and the patients filled out a Sino-Nasal Outcome Test (SNOT-22) questionnaire to assess subjective nasal symptoms. Results: As a group, the 34 patients who completed the study showed no significant improvement in the treated nostrils versus control nostrils (p = 1.000). However, the nine patients who did respond to the honey treatment relative to their control side responded very well. A number of these patients had high IgE levels in their blood. The manuka honey did not appear to modify the culture results in the ethmoid cavities after 30 days of treatment, but patients who completed the SNOT-22 questionnaire indicated global improvement in their symptoms while receiving the honey spray (p = .0220). Conclusion: Overall, topical manuka honey application in AFRS, despite showing symptomatic benefits, did not demonstrate a global improvement in endoscopically staged disease, but specific patients did show significant positive responses. Further research is needed to determine the factors of the patients who responded well to the honey spray, which may correlate to high IgE levels.”
Other publications of related to the molecular biology of biofilms in CRS are:
Role of biofilms in chronic inflammatory diseases of the upper airways.(Aug 2011)
[Observation of bacterial biofilms in patients with chronicrhinosinusitis] (July 2011)
Aspergillus fumigatus biofilm on primary human sinonasal epithelial culture(July 2011)
Bacterial biofilms in chronicrhinosinusitis and their relationship with inflammation severity (April 2012)
Final comments
I could find no research reports on any completed human trials on manuka honey treatment of chronic rhinosinusitis in humans. A search of Internet will find that a few individuals have tried manuka honey irrigation of sinuses on their own, but the results appear to be sketchy and anecdotal. Three clinical trials are listed currently for manuka honey. One is for Manuka Honey Irrigation After Sinus Surgery. So, despite the New Age sound of manuka honey sinus treatment, it has come to the attention of the medical/pharma establishment. In any event, I predict that soon there will be clinical trials of substances that fight biofilms in CRS.
Manuka honey could possibly provide a sweet approach for treating CRS. However, for the pharma industry there is no money in honey. So the key substance entering medical practice that penetrates biofilms in CRS is likely to be something else.
FROM TIME TO TIME, THIS BLOG DISCUSSES DISEASE PROCESSES. THE INTENTION OF THOSE DISCUSSIONS IS TO CONVEY CURRENT RESEARCH FINDINGS AND OPINIONS, NOT TO GIVE MEDICAL ADVICE. THE INFORMATION IN POSTS IN THIS BLOG IS NOT A SUBSTITUTE FOR A LICENSED PHYSICIAN’S MEDICAL ADVICE. IF ANY ADVICE, OPINIONS, OR INSTRUCTIONS HEREIN CONFLICT WITH THAT OF A TREATING LICENSED PHYSICIAN, DEFER TO THE OPINION OF THE PHYSICIAN. THIS INFORMATION IS INTENDED FOR PEOPLE IN GOOD HEALTH. IT IS THE READER’S RESPONSIBILITY TO KNOW HIS OR HER MEDICAL HISTORY AND ENSURE THAT ACTIONS OR SUPPLEMENTS HE OR SHE TAKES DO NOT CREATE AN ADVERSE REACTION.




Manuka OK.
What to do with it? How to use?
Pait the wall of the shop?
Thank you.
Just wanted to pipe in here. Manuka Honey cured my sinus infections even after surgery failed me. I was getting horrible infections with every cold, having to take 3-6 weeks of Ceftin to clear up until I found out about the study on Manuka Honey irrigations. Ive tried irrigating, and it helps, but if you want to knock out your sinus problems buy some medical grade UMF16 Honey, dunk a QTip in it so its got a big gob of honey covering it, and wipe that whole amount into each nostril. Then like on your back and let it drift all the way back to your throat. Try to keep all that honey in your nose for a half hour or so. Its irritating but do that 2x daily, especially right before you sleep, and it will kill EVERYTHING in your nose in a few days to a week. I mean, every time you blow your nose it will be crystal clear, no green or yellow stuff at all.
Another tip is to skip irrigation’s if you have a cold. Too much water + congestion = infection. Wait until the cold is gone before pumping fluid in there.
Doug
Your experience is of course strictly anectodal but nonetheless very interesting. Thanks for sharing it. As far as I know there is no published research on how to administrate manuka honey for sinus problems, responding also to Ilya’s inquiry.
Vince
Doug’s instructions above were a great help to me, I will try that.
Also, I have tried manuka honey in my nebuliser (diluted in sterile water) to kill infection in my lung. I have bronchiectasis with half left lung removed, amazingly the nebulised manuka improved my breathing and the next sputem sample sent to lab was clear. The infection it seemed to kill was haemophilus influenzae.
su
Pingback: Health & Energy day 1264 (Tue 18 Jun 2013) | JimBaconLive
Pingback: Quorum Sensing Part 2 – Intra and inter-species molecular communications | AGING SCIENCES – Anti-Aging Firewalls
Pingback: My Homepage