By Vince Giuliano
Life depends on molecular messaging. Cells and organisms – simple ones like c-elegans and complicated ones like humans – are constantly exchanging internal molecular messages. These messages are critical for articulating development, tissue homeostasis, repairs of various kinds, fighting infectious bacteria and other essential functions of life, Members of different species – including us humans – use molecular messages to sense and gain information about members of other species, to communicate with them, and to jockey for a survival edge with them. We sometimes use such messaging to fool or blindside each other. This applies to bacteria-bacteria interactions, to bacteria-plant interactions, to bacteria-animal interactions and to all combinations of the above. This blog entry covers recent research regarding such forms of messaging
The Quorum Sensing Part 1 blog entry described how many species of bacteria use quorum sensing (QS) molecular messaging to determine the best survival strategy for members of that species under particular conditions, and to drive implementation actions. Moreover, that blog entry discussed how blocking of quorum sensing through use of phytochemicals could be an effective antibacterial strategy that might work well against colony-forming antibiotic resistant bacterial strains. The focus was on intra-species communications. This blog entry, instead, focuses on molecular communications and quorum sensing in inter-species communications and among animal and human cells.
Topics that will be touched on include:
- The importance of molecular packing of signaling molecules into vesicles and exosomes,
- The roles of QS in biofilm formation,
- Molecular messaging of cells in response to infection and stresses,
- The role of QS in wound healing, and
- The roles of QS in periodontal disease and the formation of tooth cavities
On inter-species molecular communications among biological organisms
Biological organisms may use a wide variety of means for communication including visual, sound, tactical, and olfactory signals, and languages of many kinds. A chameleon can change his color, and octopus can change his spots, bees can engage in flight acrobatics to signal danger, flowers can use bright colors and light-polarizing molecules to attract bees for pollination, humans can shout at each other or write treatises like this one. The list of communications strategies used by biological entities goes on and on.
This blog entry is concerned with molecular communications. Some examples are very familiar, for example a skunk squirting out butyl mercaptan, a very smelly substance, when the skunk senses danger. Members of many species emit and sense pheromones, chemical messengers addressed to members of the same species that can affect social behavior such as the presence of danger or opportunities for sexual engagement. Ants leave trails of chemical messengers to delineate pathways to food. Here, I am concerned with less-familiar molecular communications that originate at the cell level and that may affect health and longevity. Some of the key things I will be discussing are:
- Molecular communications in many instances take place through transmission of molecules packaged into microcapsules known as vesicles or exosomes.
- Quorum’s sensing enters in for both intra-and inter-species communications when cells act in concert, such as Staphylococcus bacteria in an infection deciding to form a biofilm or release virulence factors, or when immune system cells coordinate a defense.
- The single-pathogen disease model is inadequate In many real-world infectious disease situations in humans, such as a case of wound healing, of gum infection of lung infection and chronic rhinosinusitis, Instead, grasping what is going on and identifying an effective therapy requires understanding that pathogens exist in multi-species communities that form biofilms. In such a community, multiple species of bacteria and microflora as well as human cells communicate and exercise quorum sensing among each other and the net result is determined by complex signaling interactions.
- The concepts treated here, of exosome communications, of quorum sensing, of rich interspecies mutual involvement and communications, are important for lending new understanding in many areas related to infectious diseases, including wound healing,chronic rhinosinusitis, lung infections, and periodontal disease.
- It is very interesting that the publications which discuss exosome communications in disease processes rarely if ever mentioning quorum sensing while the publications which discuss quorum sensing in disease processes rarely mention exosome communications. I assume that this reflects the fact that different communities of researchers are involved in exosome communications and quorum sensing and that these two bodies of researchers probably don’t communicate with each other very well. I try to combine knowledge from these two areas of research together in this blog entry.
Molecular communications from a cell may be via direct release of messenger proteins into the cell’s immediate environment or via nano-sized vesicles, particularly exosomes, which enclose molecular signaling cargo within protective membranes.
The 2011 publication Microvesicles: Intercellular Vectors of Biological Messages relates” Cells communicate directly by cell-cell contact and indirectly via the release of mediators. But gaining a greater appreciation recently is the identification and characterization of intercellular communication through the secretion of microvesicles (MVs). MVs—small vesicles that comprise microparticles (MPs) and exosomes (1) —released from a wide variety of cells, can be considered micro-messengers. Whereas exosomes are released into the extracellular compartment by exocytosis, MPs are shed from the blebbing plasma membrane, and the composition and effects of both on target cells differ depending on the cell from which originate and the type of stimulus involved in their formation. MVs are obtained after several steps of centrifugation, whereby MPs are defined as the MVs obtained by centrifugation at < 100,000 × g, whereas exosomes are isolated by centrifugation at > 100,000 × g. Because the processes by which exosomes and MPs work are different, some researchers have claimed that such broad observations impede the defining and understanding of MV actions (2). — MPs are small vesicles, heterogeneous in size [(0.05–1 μm, a characteristic which is often used to distinguish MPs from exosomes (< 0.1 μm) and platelets (> 1 μm), respectively] and composition, with pro-coagulant and pro-inflammatory properties. Although all cell types can theoretically release MPs, the determination of the their origins has established that MPs can be released from the plasma membrane of circulating cells (such as platelets, erythrocytes, T and B cells, and monocytes); cells from the vascular wall (endothelial and smooth muscle cells); and tumor cells (3, 4). Even if the mechanisms governing MP formation are complex and not well understood, it is well appreciated that cell activation and apoptosis are the two main cellular processes that lead to their formation (Figure 1). — Exosomes are small, natural membrane vesicles released by a wide variety of cell types into the extracellular compartment by exocytosis. Exosomes were described initially during the secretion of MVs of endocytic origin by reticulocytes (8). Indeed, exosomes are formed within endosomes by invagination of the limiting membranes, resulting in the formation of multivesicular bodies. Subsequently, multivesicular bodies fuse with the plasma membrane and release exosomes into the extracellular environment (9). Secretion of exosomes can be spontaneous or induced depending on the cell type. Reticulocytes, T cells, mastocytes, and resting B cells secrete detectable levels of exosomes following the activation of a cell surface receptor. In contrast, dendritic cells, macrophages, and epithelial cells constitutively secrete exosomes in vitro, as do most tumor cells (10). The identification of MVs as exosomes is based on morphological and biochemical criteria. Thus, exosomes are obtained after high speed centrifugation (10); to confirm their presence and purity, immunoblotting analysis with antibody directed against exosomal markers like tetraspanins, heat shock protein (HSP)70, HSP90, or elongation factor-1α are needed. Also, electron microscopy or flow cytometry may be used for detection of exosomes, although there are claims that exosomes would be too small to be detected by flow cytometry. — Although exosomes do not contain any proteins from nuclear, mitochondrial, endoplasmic reticulum or Golgi apparatus, as a consequence of their endosomal origin, they are also selectively enriched in mRNA and miRNA, allowing genetic exchange between cells (11). Because MVs “hijack” the cytoplasm, capturing cytoplasm components and miRNA prior to release from the cell, the packaging of miRNAs in the cell may be random (12). — The mechanisms by which MPs and exosomes transfer biological information to recipient cells are not completely understood, but they may involve direct interaction with surface-expressed ligands or the transfer of surface receptors, proteins, mRNA, miRNA, and bioactive lipids. Moreover, they may serve as vehicles to transfer infectious particles (e.g., “Trojan horse” mechanisms) and to deliver intact organelles. Up to now, four mechanisms (ligand-receptor interaction, transfer, fusion, and internalization) by which information is transmitted by MVs have been reported (Figure 1, Table 1).”
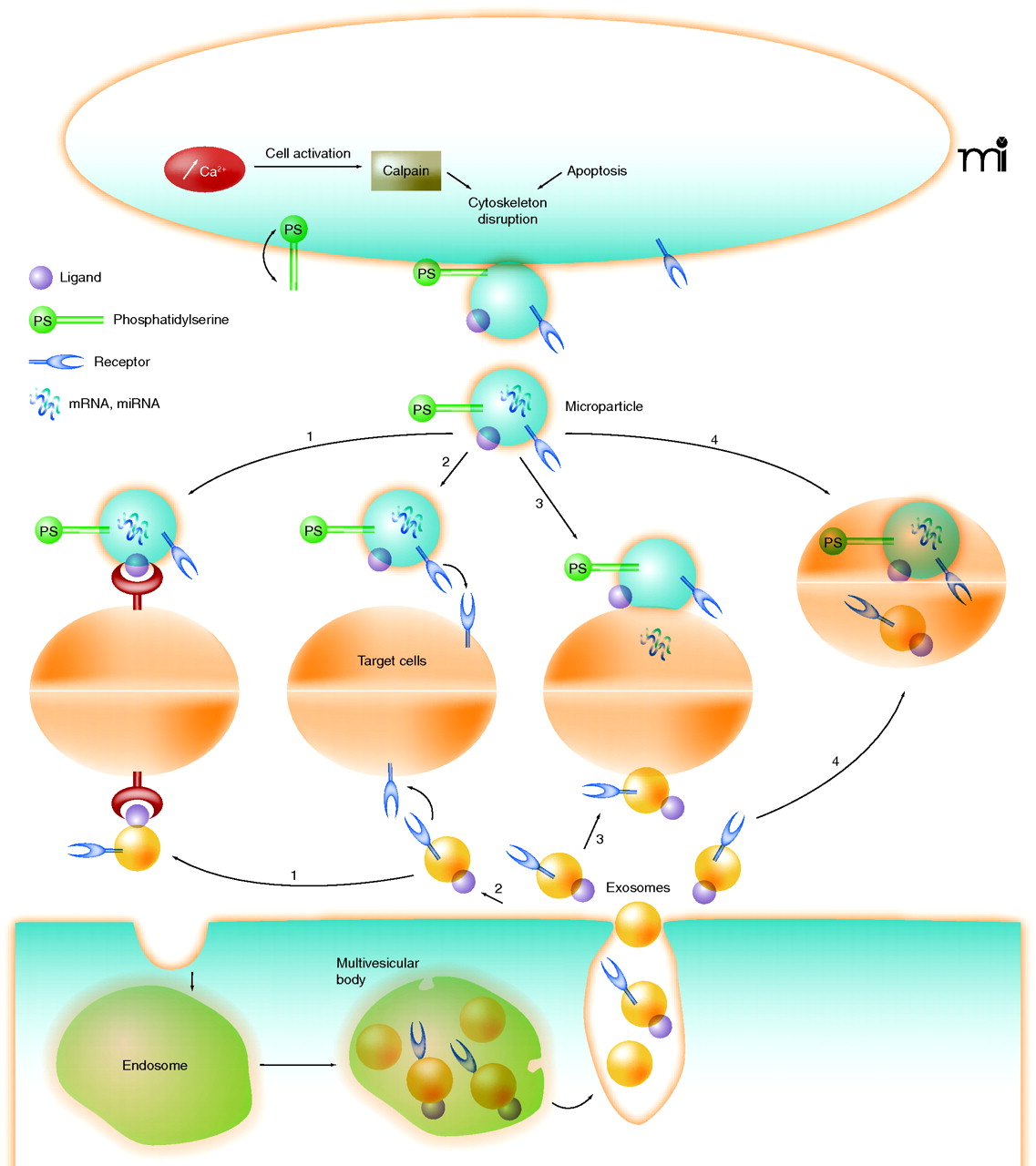
“Figure 1 Schematic representation of mechanisms for the formation of microparticles and exosomes and the different ways by which they interact with target cells Once microparticles (top) and exosomes (bottom) are released, they can interact with target cells through four mechanisms: (1) ligand/receptor interaction, (2) protein transfer, (3) membrane fusion, or (4) internalization.
Exosome packaging of molecular messages offers a number of advantages including protection of signaling cargo from being degraded due to factors in the extra-cellular medium and an ability to target the cargo messages to particular cells.
There is a lot more to be said about exosomeshow and when they are produced and what they do. Besides being conveyors of signaling molecules, exosomes can perform other functions such as ridding cells of lipofuscin trash. My colleague Jim Watson has been studying them recently and is likely to generate one or more blog entries on them in the near future. I mention them here because of their critical role in molecular inter and intra-species communications.
When the Japanese attacked Pearl Harbor, it was a clear message of war One form of bacterial interspecies messaging is an attack. Pathogenic bacteria and parasites can use exosomes to transport virulence factors into cells of a host organism and infect them.
The 2010 e-publication An exosome-based secretion pathway is responsible for protein export from Leishmania and communication with macrophages reported: “Specialized secretion systems are used by numerous bacterial pathogens to export virulence factors into host target cells. Leishmania and other eukaryotic intracellular pathogens also deliver effector proteins into host cells; however, the mechanisms involved have remained elusive. In this report, we identify exosome-based secretion as a general mechanism for protein secretion by Leishmania, and show that exosomes are involved in the delivery of proteins into host target cells. Comparative quantitative proteomics unambiguously identified 329 proteins in Leishmania exosomes, accounting for >52% of global protein secretion from these organisms. Our findings demonstrate that infection-like stressors (37 degrees C +/- pH 5.5) upregulated exosome release more than twofold and also modified exosome protein composition. Leishmania exosomes and exosomal proteins were detected in the cytosolic compartment of infected macrophages and incubation of macrophages with exosomes selectively induced secretion of IL-8, but not TNF-alpha. We thus provide evidence for an apparently broad-based mechanism of protein export by Leishmania. Moreover, we describe a mechanism for the direct delivery of Leishmania molecules into macrophages. These findings suggest that, like mammalian exosomes, Leishmania exosomes function in long-range communication and immune modulation.”
Exosome release of virulence factors appears to be a general property of mycobacterial species
The 2011 publication Mycobacteria release active membrane vesicles that modulate immune responses in a TLR2-dependent manner in mice speaks to a similar point: “Bacteria naturally release membrane vesicles (MVs) under a variety of growth environments. Their production is associated with virulence due to their capacity to concentrate toxins and immunomodulatory molecules. In this report, we show that the 2 medically important species of mycobacteria, Mycobacterium tuberculosis and Mycobacterium bovis bacille Calmette-Guérin, release MVs when growing in both liquid culture and within murine phagocytic cells in vitro and in vivo. We documented MV production in a variety of virulent and nonvirulent mycobacterial species, indicating that release of MVs is a property conserved among mycobacterial species. Extensive proteomic analysis revealed that only MVs from the virulent strains contained TLR2 lipoprotein agonists. The interaction of MVs with macrophages isolated from mice stimulated the release of cytokines and chemokines in a TLR2-dependent fashion, and infusion of MVs into mouse lungs elicited a florid inflammatory response in WT but not TLR2-deficient mice. When MVs were administered to mice before M. tuberculosis pulmonary infection, an accelerated local inflammatory response with increased bacterial replication was seen in the lungs and spleens. Our results provide strong evidence that actively released mycobacterial vesicles are a delivery mechanism for immunologically active molecules that contribute to mycobacterial virulence. These findings may open up new horizons for understanding the pathogenesis of tuberculosis and developing vaccines.” Other publications listed below describe how infected macrophages are the ones which alert the immune system and promote the inflammatory response. They also do this via a system of exosome communications, in this case intra-species.”
Animal and human cells stressed by bacterial or parasitic infection or other stressors generate exosome communications to alert other cells of their plight and to initiate responsive actions. Stressed cells at an injury site generate signals to immune system cells calling for an inflammatory response. In particular, macrophages may emit exosomes containing chemokines, cytokines and chemotaxic factors that initiate neutrophil, macrophage, and dendritic cell migration to the injury site.
A number of recent publications have been addressed to this point. I listed a few examples. Later, I will discuss how signaling from infectious bacteria in biofilm colonies can interfere with or inhibit healing human exosome communications.
The 2007 publication Exosomes released from macrophages infected with intracellular pathogens stimulate a proinflammatory response in vitro and in vivo relates “Intracellular pathogens and the molecules they express have limited contact with the immune system. Here, we show that macrophages infected with intracellular pathogens Mycobacterium tuberculosis, M bovis BCG, Salmonella typhimurium, or Toxoplasma gondii release from cells small vesicles known as exosomes which contain pathogen-associated molecular patterns (PAMPs). These exosomes, when exposed to uninfected macrophages, stimulate a proinflammatory response in a Toll-like receptor– and myeloid differentiation factor 88–dependent manner. Further, exosomes isolated from the bronchoalveolar lavage fluid (BALF) of M bovis BCG–infected mice contain the mycobacteria components lipoarabinomannan and the 19-kDa lipoprotein and can stimulate TNF-α production in naive macrophages. Moreover, exosomes isolated from M bovis BCG– and M tuberculosis–infected macrophages, when injected intranasally into mice, stimulate TNF-α and IL-12 production as well as neutrophil and macrophage recruitment in the lung. These studies identify a previously unknown function for exosomes in promoting intercellular communication during an immune response to intracellular pathogens, and we hypothesize that extracellular release of exosomes containing PAMPs is an important mechanism of immune surveillance.”
A 2013 publication Microparticles from mycobacteria-infected macrophages promote inflammation and cellular migration reports: “Mycobacterium tuberculosis infection is characterized by a strong inflammatory response whereby a few infected macrophages within the granuloma induce sustained cellular accumulation. The mechanisms coordinating this response are poorly characterized. We hypothesized that microparticles (MPs), which are submicron, plasma membrane-derived vesicles released by cells under both physiological and pathological conditions, are involved in this process. Aerosol infection of mice with M. tuberculosis increased CD45(+) MPs in the blood after 4 wk of infection, and in vitro infection of human and murine macrophages with mycobacteria enhanced MP release. MPs derived from mycobacteria-infected macrophages were proinflammatory, and when injected into uninfected mice they induced significant neutrophil, macrophage, and dendritic cell recruitment to the injection site. When incubated with naive macrophages, these MPs enhanced proinflammatory cytokine and chemokine release, and they aided in the disruption of the integrity of a respiratory epithelial cell monolayer, providing a mechanism for the egress of cells to the site of M. tuberculosis infection in the lung. In addition, MPs colocalized with the endocytic recycling marker Rab11a within macrophages, and this association increased when the MPs were isolated from mycobacteria-infected cells. M. tuberculosis-derived MPs also carried mycobacterial Ag and were able to activate M. tuberculosis-specific CD4(+) T cells in vivo and in vitro in a dendritic cell-dependent manner. Collectively, these data identify an unrecognized role for MPs in host response against M. tuberculosis by promoting inflammation, intercellular communication, and cell migration.”
One of the pro-inflammatory signaling molecules released in exosomes from mycobacteria-infected cells is HSP70. See the 2010 publication Exosomal Hsp70 induces a pro-inflammatory response to foreign particles including mycobacteria. HSP 70 is an important pluripotent heat shock protein involved in certain hormetic responses and discussed in multiple other blog entries. See, for starters, HSP70 to the rescue.
A May 2013 article Immunomodulatory impact of leishmania-induced macrophage exosomes: a comparative proteomic and functional analysis reports: “Released by many eukaryotic cells, the exosomes are 40–100 nm vesicles shown to operate over the complex processes of cell-cell communication. Among the metazoan cell lineages known to generate exosomes is the mononuclear phagocyte lineage, a lineage that parasites such as Leishmania are known to subvert as host cells. We previously reported that mouse macrophage signaling and functions are modified once co-incubated with exoproteome of Leishmania promastigotes. Using mass spectrometry analysis, we were curious to further compare the content of purified exosomes released by the J774 mouse macrophage cell line exposed or not to either LPS or to stationary phase Leishmania mexicana promastigotes. Collectively, our analyses resulted in detection of 248 proteins, ~50–80% of which were shared among the three sources studied. Using exponentially modified protein abundance index (emPAI) and network analyses, we found that the macrophage exosomes display unique signatures with respect to composition and abundance of many functional groups of proteins, such as plasma membrane-associated proteins, chaperones and metabolic enzymes. Moreover, for the first time, L. mexicana surface protease GP63 is shown to be present in exosomes released from J774 macrophages exposed to stationary phase promastigotes. We observed that macrophage exosomes are able to induce signaling molecules and transcription factors in naive macrophages. Finally, using qRT-PCR, we monitored modulation of expression of multiple immune-related genes within macrophages exposed to exosomes. We found all three groups of exosomes to induce expression of immune-related genes, the ones collected from macrophages exposed to L. mexicana sharing properties with exosomes collected from macrophage left unexposed to any agonist. Overall, our results allowed depicting that protein sorting into macrophage-derived exosomes depends upon the cell status and how such distinct protein sorting can in turn impact the functions of naive J774 cells. — Author Summary: Secreted vesicles, such as exosomes, are now considered as an important route of communication among eukaryotic cells. Depending on the donor cell source and protein content, these vesicles are expected to distinctly impact the recipient cell properties. Here, three groups of exosomes released by the mouse macrophage cell line J774 exposed or not – naive exosomes – to either Leishmania mexicana promastigotes or to LPS were compared through proteomic analysis. Also, their biological activities on naive J774 macrophages were tested. Regardless of the source, the three groups of exosomes shared 50–80% of their proteins, although their relative abundances differed, especially those associated with the plasma membrane. Post exposure to one out of the three groups of exosomes, naive J774 recipient macrophages were compared for their profile of immune transcripts. Of note, whether they were exposed to either naive exosomes or to L. mexicana– induced exosomes, the naive J774 macrophages shared similar immune transcriptional signatures, the latter being distinct from the ones displayed by the macrophages exposed to LPS-induced exosomes. These data are discussed within the context of the unique cross talk that accounts for the early establishment of an immunomodulatory parasite such as Leishmania in its mammalian host.”
Not only can macrophages generate signaling molecules when stressed, but also signaling molecules in exosomes can directly influence the behavior of macrophages.
The 2011 publication [A preliminary study on the influence of human plasma exosomes-like vesicles on macrophage Wnt5A-Ca²+ pathway] reports: “OBJECTIVE: To study the influence of human plasma exosomes-like vesicles on the regulatory function of macrophages. METHODS: The exosomes-like vesicles were purified from healthy donors plasma with a series of high-speed centrifugation and ultrafiltration. Macrophages were derived from cultured human blood monocytes. The molecular markers of macrophages were assayed by FACS. After cultured with exosomes-like vesicles, the changes of macrophages cytoplasma Ca(2+), and related genes and proteins were assayed by FACS, RT-PCR and Western Blot, respectively. RESULTS: After cultured with exosomes-like vesicles, mean fluorescent intensity (MFI) of macrophages cytoplasma Ca(2+) was increased. The vesicles enhanced macrophages to express cytokines genes, the expression of IL-1β and TNF-α genes being increased by 0.85 and 1.69 times respectively at 2 h, and that of IL-6 gene 3.7 times compared with the control at 8 h. However, the vesicles inhibited the expression of macrophages IL-10 gene, had no influence on the Frizzled5 receptor expression and could induce CaMKII phosphorylation. CONCLUSIONS: Exosomes-like vesicles can up-regulate macrophages expression of inflammatory cytokines genes, and increase the secretion of inflammatory cytokines by activating the Wnt5A-Ca(2+) signaling pathway.”
Exosomes from infected macrophages may contain many different proteins including antigenic (antibody-producing) ones. The 2010 publication Proteomic analysis identifies highly antigenic proteins in exosomes from M. tuberculosis-infected and culture filtrate protein-treated macrophages relates: “Exosomes are small 30-100 nm membrane vesicles released from hematopoietic and nonhematopoietic cells and function to promote intercellular communication. They are generated through fusion of multivesicular bodies with the plasma membrane and release of interluminal vesicles. Previous studies from our laboratory demonstrated that macrophages infected with Mycobacterium release exosomes that promote activation of both innate and acquired immune responses; however, the components present in exosomes inducing these host responses were not defined. This study used LC-MS/MS to identify 41 mycobacterial proteins present in exosomes released from M. tuberculosis-infected J774 cells. Many of these proteins have been characterized as highly immunogenic. Further, since most of the mycobacterial proteins identified are actively secreted, we hypothesized that macrophages treated with M. tuberculosis culture filtrate proteins (CFPs) would release exosomes containing mycobacterial proteins. We found 29 M. tuberculosis proteins in exosomes released from CFP-treated J774 cells, the majority of which were also present in exosomes isolated from M. tuberculosis-infected cells. The exosomes from CFP-treated J774 cells could promote macrophage and dendritic cell activation as well as activation of naïve T cells in vivo. These results suggest that exosomes containing M. tuberculosis antigens may be alternative approach to developing a tuberculosis vaccine.”
A pathogen can also sometimes trick host cells into sending microvesicle or exosome messages that actually help the pathogen.
The 2013 publication Interplay of host-pathogen microvesicles and their role in infectious disease relates “The release of extracellular vesicles, whether MVs (microvesicles) or exosomes, from host cells or intracellular pathogens is likely to play a significant role in the infection process. Host MVs may fuse with pathogen surfaces to deliver host complement regulatory proteins. They may also deliver cytokines that enhance invasion. Decoy functions are also possible. Whereas host MVs may direct pathogens away from their target cells, pathogen MVs may in turn redirect complement membrane-attack complexes away from their target pathogen. An understanding of the mechanisms of this interplay, bringing about both immune evasion and enhanced invasion, will help to direct future research with a view to rendering pathogens more susceptible to immune attack or in improving drug efficacy. It should also be possible to use MVs or exosomes isolated directly from the pathogens, or from the cells infected with pathogens, to provide alternative vaccination strategies.”
The 2011 publication Exosomes released from M. tuberculosis infected cells can suppress IFN-γ mediated activation of naïve macrophages describes another situation where human macrophage cell exosome communications are negatively affected by a pathogen so as to compromise immune system response. “BACKGROUND: Macrophages infected with Mycobacterium tuberculosis (M.tb) are known to be refractory to IFN-γ stimulation. Previous studies have shown that M.tb express components such as the 19-kDa lipoprotein and peptidoglycan that can bind to macrophage receptors including the Toll-like receptor 2 resulting in the loss in IFN-γ responsiveness. However, it is unclear whether this effect is limited to infected macrophages. We have previously shown that M.tb-infected macrophages release exosomes which are 30-100 nm membrane bound vesicles of endosomal origin that function in intercellular communication. These exosomes contain mycobacterial components including the 19-kDa lipoprotein and therefore we hypothesized that macrophages exposed to exosomes may show limited response to IFN-γ stimulation. METHODOLOGY/PRINCIPAL FINDINGS: Exosomes were isolated from resting as well as M.tb-infected RAW264.7 macrophages. Mouse bone marrow-derived macrophages (BMMØ) were treated with exosomes +/- IFN-γ. Cells were harvested and analyzed for suppression of IFN-γ responsive genes by flow cytometry and real time PCR. We found that exosomes derived from M.tb H37Rv-infected but not from uninfected macrophages inhibited IFN-γ induced MHC class II and CD64 expression on BMMØ. This inhibition was only partially dependent on the presence of lipoproteins but completely dependent on TLR2 and MyD88. The exosomes isolated from infected cells did not inhibit STAT1 Tyrosine phosphorylation but down-regulated IFN-γ induced expression of the class II major histocompatibility complex transactivator; a key regulator of class II MHC expression. Microarray studies showed that subsets of genes induced by IFN-γ were inhibited by exosomes from H37Rv-infected cells including genes involved in antigen presentation. Moreover, this set of genes partially overlapped with the IFN-γ-induced genes inhibited by H37Rv infection. CONCLUSIONS: Our study suggests that exosomes, as carriers of M.tb pathogen associated molecular patterns (PAMPs), may provide a mechanism by which M.tb may exert its suppression of a host immune response beyond the infected cell.”
Other publications that treat host-cell exosome messaging in response to infection or other stressors include:
Prion-infected cells regulate the release of exosomes with distinct ultrastructural features. (2012)
TLR2-mediated expansion of MDSCs is dependent on the source of tumor exosomes. (2011)
Again, I saw no mention of quorum sensing while looking at the above exosome-related publications. Later I will cite publications showing that quorum sensing is deeply involved.
Immune system cells likely use quorum sensing for a number of purposes, for example establishing homeostasis of numbers of T cells in a niche.
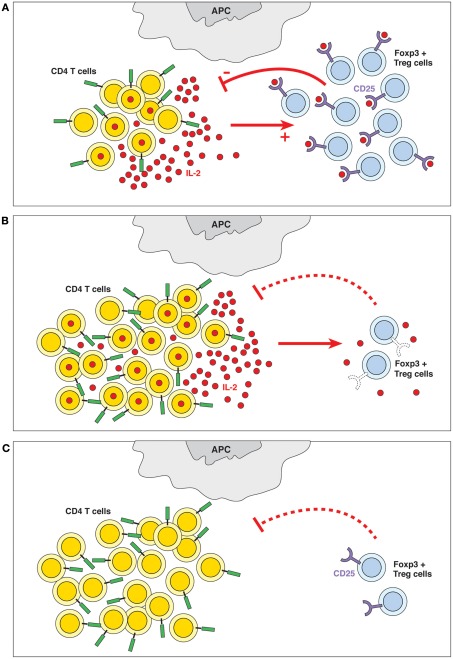
The 2012 e-publication Quorum-Sensing in CD4(+) T Cell Homeostasis: A Hypothesis and a Model reports “Homeostasis of lymphocyte numbers is believed to be due to competition between cellular populations for a common niche of restricted size, defined by the combination of interactions and trophic factors required for cell survival. Here we propose a new mechanism: homeostasis of lymphocyte numbers could also be achieved by the ability of lymphocytes to perceive the density of their own populations. Such a mechanism would be reminiscent of the primordial quorum-sensing systems used by bacteria, in which some bacteria sense the accumulation of bacterial metabolites secreted by other elements of the population, allowing them to “count” the number of cells present and adapt their growth accordingly. We propose that homeostasis of CD4(+) T cell numbers may occur via a quorum-sensing-like mechanism, where IL-2 is produced by activated CD4(+) T cells and sensed by a population of CD4(+) Treg cells that expresses the high-affinity IL-2Rα-chain and can regulate the number of activated IL-2-producing CD4(+) T cells and the total CD4(+) T cell population. In other words, CD4(+) T cell populations can restrain their growth by monitoring the number of activated cells, thus preventing uncontrolled lymphocyte proliferation during immune responses. We hypothesize that malfunction of this quorum-sensing mechanism may lead to uncontrolled T cell activation and autoimmunity. Finally, we present a mathematical model that describes the key role of IL-2 and quorum-sensing mechanisms in CD4(+) T cell homeostasis during an immune response.”
“Quorum-sensing. The presence of IL-2 and the ability of the Treg cells to detect its levels are crucial to the homeostasis of the immune system. Quorum-sensing in this case is defined as an indirect feedback loop where the IL-2 produced by a subpopulation of activated T cells (among others) is detected (sensed) by a subpopulation of CD4+ Treg cells expressing the high-affinity IL-2Rα-chain; these cells contribute to controlling the number of CD4+ T cells. In other words, the overall CD4+ T cell populations sense the produced quantities of IL-2 and adapt their behavior accordingly. (B) Failure of quorum-sensing by defective sensor molecule. The inability to detect IL-2 because of defects in IL-2R expression (in IL-2Rα−/− or IL-2Rβ−/− mice) or signaling (in STAT5−/− mice) leads to lymphoid hyperplasia and autoimmune disease. (C) Failure of quorum-sensing due to absence of the sensed molecule. In the absence of IL-2, Treg cells do not survive, which causes lymphoid hyperplasia and autoimmune pathology.”
Many forms of human bacterial pathologies – whether they be in the lung, in the mouth, or in nonhealing wounds – involve biofilms in which multiple species of bacteria cooperate for their mutual benefit and there is a rich network of quorum sensing communications.
The 2008 publication Quorum Sensing and Bacterial Social Interactions in Biofilms relates: “ — The ability of bacteria to communicate and behave as a group for social interactions like a multi-cellular organism has provided significant benefits to bacteria in host colonization, formation of biofilms, defense against competitors, and adaptation to changing environments. Importantly, many QS-controlled activities have been involved in the virulence and pathogenic potential of bacteria. Therefore, understanding the molecular details of quorum sensing mechanisms and their controlled social activities may open a new avenue for controlling bacterial infections.”
It is time to get beyond looking at infections in terms of single pathogens.
Continuing (ref) “ — It was believed for many years that bacteria, unlike eukaryotic organisms, behaved as self-sufficient individuals and maintained a strictly unicellular life-style [1–3]. During infections, bacterial mass was considered nothing more than the sum of these individuals. Our perception of bacteria as unicellular life-style was deeply rooted in the pure culture paradigm of Robert Koch’s era, when Koch established his “golden criteria” to define a bacterial pathogen by using pure-culture approaches [3]. Indeed, Koch’s concept has led to the great success in the identification of bacterial pathogens and development of antibiotic treatments in acute bacterial infections [3,4]. However, pure-culture planktonic growth of bacteria rarely exists in natural environments. In fact, bacteria in Nature largely reside in a complex and dynamic surface-associated community called a biofilm [3,5,6]. If viewing an intact biofilm under microscope, one may immediately find that bacteria in biofilms do not randomly stick together, but rather form a well-organized community with numerous specialized configurations [5,6]. One may also find that bacterial cells in biofilms physically interact with each other and maintain ‘intimate’ relationships [5,6]. Even without physical contact, bacteria living at the same community likely secrete small extra-cellular molecules to interact with each other [7–10]. It was not until the last three decades that our view of self-sufficient unicellular lifestyle of bacteria has changed. The advances from at least two major research areas, biofilm development and bacterial quorum sensing, have led us to begin to appreciate, in much more detail for the first time, the concept that bacteria can organize into groups, form well-organized communities, and communicate for coordinated activities or social life that was once believed to be restricted to multi-cellular organisms [3,6–13]. — Microbiologists have discovered an unexpectedly high degree of coordinated multi-cellular behaviors that have led to the perception of biofilms as “cities” of microorganisms [6]. Especially, many bacteria have been found to regulate diverse physiological processes and group activities through a mechanism called quorum sensing, in which bacterial cells produce, detect and respond to small diffusible signal molecule [7–14]. It has long been known that in infectious diseases the invading bacteria need to reach a critical cell density before they express virulence and overwhelm the host defense mechanisms before they initiate an infectious disease [1–3]. Since quorum-sensing mechanisms are widespread in both prokaryotic and single-celled eukaryotic organisms such as fungi [7–9,15], it is not surprising that cell-cell communication through quorum sensing has important implications in microbial infections. A growing body of excellent reviews has highlighted.”
Biofilms are involved in many important infectious disease processes, if not most of them. Studied examples include infectious kidney stones, bacterial endocarditis, cystic fibrosis lung infections, non-healing wounds, chronic rhinosinusitis, periodontal disease and dental caries.
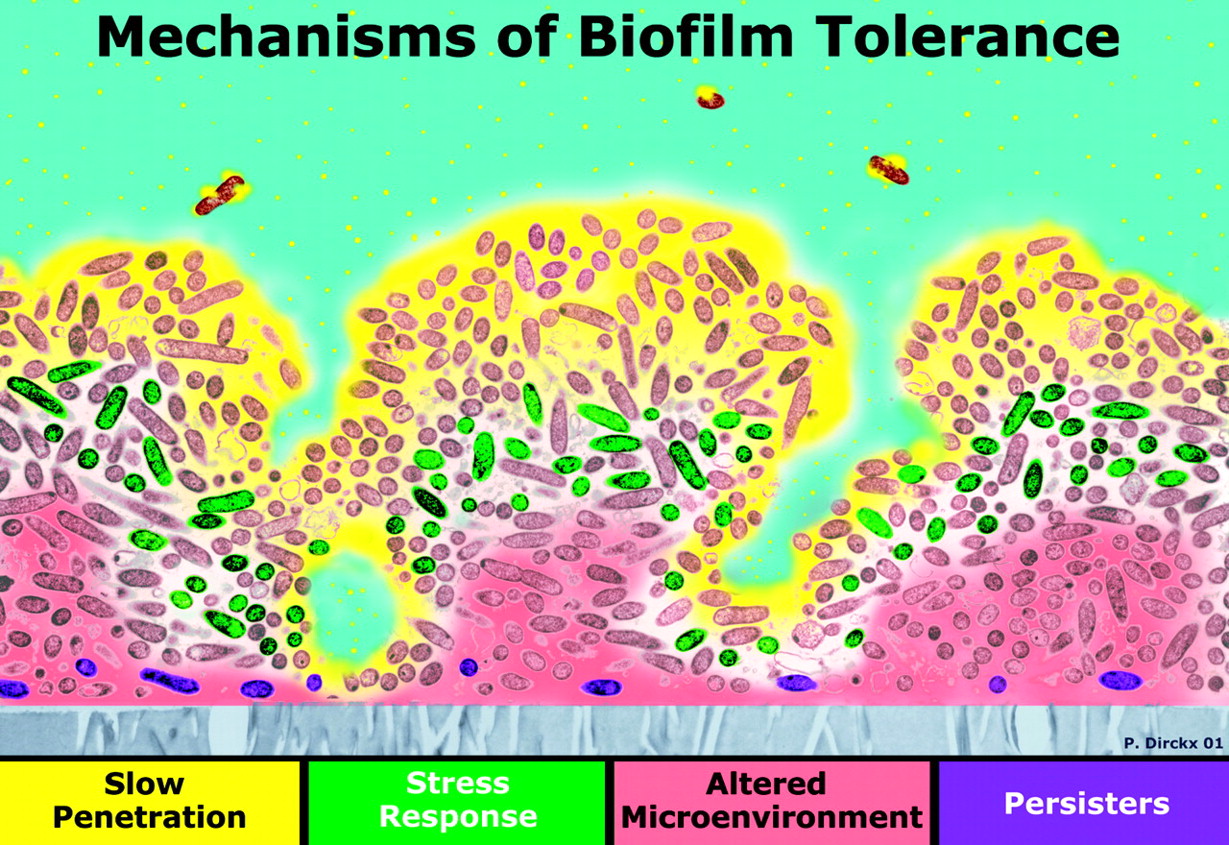
Infectuous bacteria in biofilm communities have developed effective strategies for resisting antibiotics. The illustration is from a PowerPoint presentation A Three-Dimensional Computer Model of Four Hypothetical Mechanisms Protecting Biofilms from Antimicrobials
“Four possible mechanisms of biofilm antibiotic resistance. The image is a cross section of a biofilm with the attachment surface (gray) at the bottom and the aqueous phase containing the antibiotic (yellow) at the top. In zones where there is nutrient depletion (red), antibiotic action may be antagonized. Some bacteria may activate stress responses (green), while others may differentiate into a protected phenotype (purple).”
Biofilms occur naturally in nature, such as in the beds of streams, and some are beneficial to humans. You can find out all about biofilms in a nice tutorial here. You can also check out an earlier blog entry Chronic rhinosinusitis, MRSA, biofilms and manuka honey.
Because of molecular communications, there are no true bystander cells when it comes to infectious attacks.
The “bystander effect” has long puzzled biologists. You do something to some cells like expose them to radiation, and the impact definitely shows up in not only the irradiated cells but also in neighboring cells that did not get irradiated. But the fact is that because of molecular communications from the irradiated cells, the others are affected too. This bystander effect effect shows up in many domains of biology, including attacks by infectious bacteria.. For example, the 2011 publication Mannosylated lipoarabinomannan balances apoptosis and inflammatory state in mycobacteria-infected and uninfected bystander macrophages. “To assess the role of mannosylated lipoarabinomannan (ManLAM) in the inflammatory and apoptotic response of mycobacteria-infected and uninfected, bystander cells we applied a mouse macrophage model of infection with avirulent strains–Mycobacterium bovis BCG, Mycobacterium tuberculosis (MTB) H37Ra and compared with a virulent MTB H37Rv strain infection. ManLAM contributed to the infection of macrophages by protection from apoptosis with stabilized Bcl-2 expression and down-regulated Bax expression for infected cells (BCG) or with stabilized Bcl-2 expression for uninfected bystander target cells (H37Ra). Additionally, ManLAM up-regulated FasL expression on the infected cells. Active extracellular signal-regulated kinase (ERK1/2) in BCG and H37Rv infection provided an anti-apoptotic effect by stabilization of anti-apoptotic Bcl-2 expression in the infected cells. Inhibitors specific for c-Jun-NH2-terminal kinase or stress-activated kinase (JNK) and p38 kinase decreased apoptosis of infected cells (BCG, H37Ra) and of uninfected bystanders (H37Ra) by down-regulating Bax. ManLAM significantly down-regulated production of pro-inflammatory IL-12 and TNF-alpha and activation of JNK by both avirulent strains. We conclude that by stabilization of Bcl-2 expression, down-regulation of JNK activity and down-regulation of pro-inflammatory cytokines production ManLAM can contribute to suppression of apoptosis and inflammatory reaction of uninfected, bystander cells.” In rich networks of biological communications, there are no real bystanders.
I hypothesize that most basic decisions of cells that drive cell behavior and responses are not decisions made by individual cells but are common decisions made by the community of cells involved, triggered by quorum sensing and likely communicated by exosome signaling. These cell communities can include pathogenic bacteria of a given species attacking a wound or organ, human cells of given types such as T cells and stem cell types, microbiota, and plant cells of a given type.
This is a rather sweeping conjecture of my own and I will argue for it by citing examples.
- In the Quorum Sensing Part 1 blog entry I have already cited for multiple infectious bacteria how quorum sensing, counting the relative number of bacteria present, drives behavior which determines whether the bacteria lie low, whether they form protective biofilms, or whether they go on the attack and express virulence. These are community decisions and the bacteria tend to act in concert. Publications like ones cited here above suggest that the quorum sensing communications at least partially involve messages encapsulated in exosomes or other microvesicles.
- In healthy individuals, T cells make community decisions as to which cells represent “self,” and which ones represent “foreign invader” and should therefore be attacked. The 2012 publication Quorum sensing allows T cells to discriminate between self and nonself reports: “T cells orchestrate pathogen-specific adaptive immune responses by identifying peptides derived from pathogenic proteins that are displayed on the surface of infected cells. Host cells also display peptide fragments from the host’s own proteins. Incorrectly identifying peptides derived from the body’s own proteome as pathogenic can result in autoimmune disease. To minimize autoreactivity, immature T cells that respond to self-peptides are deleted in the thymus by a process called negative selection. However, negative selection is imperfect, and autoreactive T cells exist in healthy individuals. To understand how autoimmunity is yet avoided, without loss of responsiveness to pathogens, we have developed a model of T-cell training and response. Our model shows that T cells reliably respond to infection and avoid autoimmunity because collective decisions made by the T-cell population, rather than the responses of individual T cells, determine biological outcomes. The theory is qualitatively consistent with experimental data and yields a criterion for thymic selection to be adequate for suppressing autoimmunity.”
- 3. “Bystander” cellular responses are only explainable in terms of this hypothesis. Consider the radiation bystander effect for example. When a hormetic dose of radiation is applied ti human cells, not only do the cells that receive that radiation experience upgrading of their stress responses, but also neighboring cells that themselves received no radiation also get their stress responses upgraded. If the radiation dose is super-hormetic and the irradiated cells experience damage, so will the un-irradiated bystander cells be damaged. This bystander effect might be immune system mediated and it might induce a hormetic response (ref). Also the effect could be a direct result of cell to cell signaling (ref)(ref). In any case, quorum-sensing decision is made by the community of cells and this decision is communicated to all cells, radiated and not-radiated alike. The bystander cells go along with what the community decides. Based on other publications, I surmise that it is highly probable that communication originates with exosomes released from the radiation-stressed cells. Quorum sensing appears to be a necessary mechanism for decisions to be made among all the cells in the community A quorum sensing decision could be 1. to ignore the stress signals, 2. To upgrade their stress responses, 4. To become damaged, or 4. To scream like crazy for the immune system to weigh in and inflammation to be triggered. It appears that cells are very socialist and lemming-like in their behavior. They appear to respond collectively depending on how cells in the community feel at the moment. This is probably one of the reasons why stress responses are dependent on circadian rhythms and not just a function of the stress itself.
Human diseases may entail interspecies molecular communications not only between infectious bacteria of a given species and cells of the host human, but also signaling actions of third-party species organisms including microflora. The conventional picture of a bacterial infection being a contest between attacking bacterial cells and defending human cells can be far too simple. Each of multiple co-located species may use quorum sensing for regulation of gene expression and the net pathological impact may depend critically on messages from seemingly innocuous third-party organisms.
This was pointed out back in 2003 in the publication Modulation of Pseudomonas aeruginosa gene expression by host microflora through interspecies communication. “The change in gene expression patterns in response to host environments is a prerequisite for bacterial infection. Bacterial diseases often occur as an outcome of the complex interactions between pathogens and the host. The indigenous, usually non-pathogenic microflora is a ubiquitous constituent of the host. In order to understand the interactions between pathogens and the resident microflora and how they affect the gene expression patterns of the pathogens and contribute to bacterial diseases, the interactions between pathogenic Pseudomonas aeruginosa and avirulent oropharyngeal flora (OF) strains isolated from sputum samples of cystic fibrosis (CF) patients were investigated. Animal experiments using a rat lung infection model indicate that the presence of OF bacteria enhanced lung damage caused by P. aeruginosa . Genome-wide transcriptional analysis with a lux reporter-based promoter library demonstrated that 4% of genes in the genome responded to the presence of OF strains using an in vitro system. Characterization of a subset of the regulated genes indicates that they fall into seven functional classes, and large portions of the upregulated genes are genes important for P. aeruginosa pathogenesis. Autoinducer-2 (AI-2)-mediated quorum sensing, a proposed interspecies signalling system, accounted for some, but not all, of the gene regulation. A substantial amount of AI-2 was detected directly in sputum samples from CF patients and in cultures of most nonpseudomonad bacteria isolated from the sputa. Transcriptional profiling of a set of defined P. aeruginosa virulence factor promoters revealed that OF and exogenous AI-2 could upregulate overlapping subsets of these genes. These results suggest important contributions of the host microflora to P. aeruginosa infection by modulating gene expression via interspecies communications.” – “Broad response in P. aeruginosa seems to be induced by the presence of OF. Although competition for nutrients was expected to be one of the factors, the observed response appears to be a result of more complex interactions between P. aeruginosa and OF bacteria. — The upregulation of a significant number of virulence genes in P. aeruginosa by OF strains is intriguing and probably explains the increased lung damage observed in the presence of the OF strain in the animal experiments. The upregulation of a pathogen’s virulence factors by microflora underscores the importance of bacterial interactions in pathogenicity. The interactions between bacteria and their host are believed to determine the evolution of many of the virulence factors that pathogens possess — Bacterial pathogens, especially those that inhabit both environmental niches as well as animal hosts, may rely on these interactions to maintain their virulence when out of the hosts. On the other hand, these virulence factors may play a role in the development of microbial communities. — The regulation of P. aeruginosa gene expression and virulence involves its own repertoire of cell–cell signaling molecules (Fuqua et al., 2001; Miller and Bassler, 2001; Whitehead et al., 2001; Smith and Iglewski, 2003). The pathways through which the OF strains affect P. aeruginosa gene expression are expected to be multifactorial and complex. One of the signals seems to be autoinducer-2 (AI-2) produced by non-pseudomonad strains. It seems that P. aeruginosa is modulating its behaviour by monitoring the environmental conditions and by eavesdropping on the other bacteria via AI-2 and probably other signals. AI-2 in the sputum samples could reach a substantial amount, which is readily detectable by the V. harveyi assay. The regulation of a number of virulence factor genes by AI-2 indicates that this signal produced by OF strains contributed, at least in part, to the observed modulation of P. aeruginosa gene expression and changes in its pathogenicity. The AI-2-regulated P. aeruginosa virulence factor genes partially overlap those modulated by OF strains, suggesting that, in the co-culture experiments, AI-2 was one of the signals but not the only signal produced by the OF that regulates P. aeruginosa gene expression. In the lungs of CF patients, this AI-2-mediated effect could also originate from secondary pathogens, suggesting that the virulence of secondary pathogens could also be delivered through enhancing the virulence of the primary pathogen, P. aeruginosa. It is also possible that P. aeruginosa could influence virulence factor genes of secondary pathogens or those potentially present in the OF strains.”
The 2006 review publication Communication in bacteria: an ecological and evolutionary perspective comments on this picture: “Individual bacteria can alter their behaviour through chemical interactions between organisms in microbial communities. This is generally referred to as quorum sensing. Frequently, these interactions are interpreted in terms of communication to mediate coordinated, multicellular behaviour. We show that the nature of interactions through quorum-sensing chemicals does not simply involve cooperative signals, but entails other interactions such as cues and chemical manipulations. These signals might have a role in conflicts within and between species. The nature of the chemical interaction is important to take into account when studying why and how bacteria react to the chemical substances that are produced by other bacteria.”
The 2008 treatise Bio-Communication of Bacteria and their Evolutionary Roots in Natural Genome Editing Competences of Viruses offers a nice overview of bacterial communications systems and their evolutionary origins: “Communicative competences and the use of a semiochemical vocabulary enable bacteria to develop, organize and coordinate rich social life with a great variety of behavioral patterns even in which they organise themselves like multicellular organisms. They have existed for almost four billion years and still survive, being part of the most dramatic changes in evolutionary history such as DNA invention, cellular life, invention of nearly all protein types, partial constitution of eukaryotic cells, vertical colonisation of all eukaryotes, high adaptability through horizontal gene transfer and cooperative
multispecies colonisation of all ecological niches. Recent research demonstrates that these bacterial competences derive from the aptitude of viruses for natural genome editing.
In contrast to a book which would be the appropriate space to outline in depth all communicative pathways inherent in bacterial life in this current article I want to give an overview for a broader readership over the great variety of bacterial bio-communication: In a first step I describe how they interpret and coordinate, what semiochemical vocabulary they share and which goals they try to reach. In a second stage I describe transorganismic communication, i.e. the main categories of sign-mediated interactions between bacterial and non-bacterial organisms, and interorganismic communication, i.e. between bacteria of the same or related species. In a third stage I will focus on intraorganismic communication, i.e. the relationship between bacteria and their obligate settlers, i.e. viruses. We will see that bacteria are important hosts for multiviral colonisation and the virally-determined order of nucleic acid sequences, which has implications for our understanding of the evolutionary history of pre-cellular and cellular life.”
Different species of bacteria in a colony may engage in cross talk communications so as to impact the behavior and virulance of each other.
From the 2005 publication Interactions between Oral Bacteria: Inhibition of Streptococcus mutans Bacteriocin Production by Streptococcus gordonii: “Recent studies have shown that quorum sensing modulates both intra- and interspecies cell-cell communications. For example, there is cross-inhibition by quorum-sensing pheromones between Staphylococcus aureus and Staphylococcus epidermidis (21). S. epidermidis pheromones inhibited the S. aureus agr response, which may explain the predominance of S. epidermidis on the skin. Interaction between Bacillus subtilis and Erwinia carotovora provides another example (5), since the former produces an enzyme (AiiA) that inactivates the E. carotovora autoinducers. Inactivation of the autoinducers by AiiA attenuated the virulence of E. carotovora.”
The two central “usual suspect” bacteria for inhibited wound healing are Staphylococcus aureus (SA) and Pseudomonas aeruginosa (PA)
PA Image source
At least, these are the two most-studied bacteria.
Individually and together in a biofilm colony with other bacteria, they appear able to halt the normal progression of wound healing.
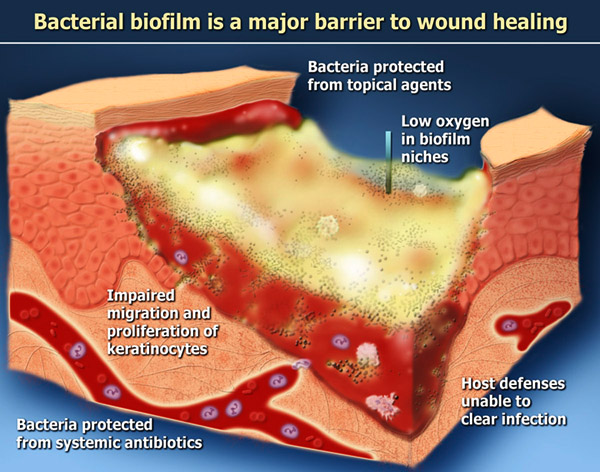
The 2009 publication Staphylococcal biofilms impair wound healing by delaying reepithelialization in a murine cutaneous wound model reported “Bacterial biofilms have gained increasing visibility in recent years as a ubiquitous form of survival for microorganisms in myriad environments. A number of in vivo models exist for the study of biofilms in the setting of medically relevant implanted foreign bodies. Growing evidence has demonstrated the presence of bacterial biofilms in the setting of a number of chronic wound states including pressure sores, diabetic foot ulcers, and venous stasis ulcers. Here we present a novel murine cutaneous wound system that directly demonstrates delayed reepithelialization caused by the presence of a bacterial biofilm. We established biofilms using either Staphylococcus aureus or Staphylococcus epidermidis in splinted cutaneous punch wounds created on the backs of normal C57Bl6/J mice. Wound reepithelialization was significantly delayed by bacterial biofilms. This effect was specifically dependent on the ability of the bacteria to form biofilms as demonstrated by exogenous administration of biofilm inhibiting peptides and the use of mutant Staphylococcus spp. deficient in biofilm formation. This represents the first direct evidence for the effect of bacterial biofilms on cutaneous wound healing. — Biofilms are complex communities of microorganisms associated with surfaces and are characterized by high cell densities associated with an extracellular polymer matrix. In recent years, microbial research has revealed that the bacterial biofilms are far more ubiquitous than previously realized. Indeed, when compared with the traditional view of bacteria growing in planktonic culture with essentially unlimited energy sources, it has become clear that biofilms represent a far more realistic representation of bacterial behavior outside of the laboratory setting.1–5 The genetic signals involved in biofilm formation have been elucidated in several bacterial species of clinical interest, including the Gram-positive cocci Staphylococcus aureus and Staphylococcus epidermidis and the concept of quorum sensing has emerged as an essential means of bacterial communication essential to form the biofilm community.6–12 It is now appreciated that there exist discrete signaling molecules that both promote biofilm assembly as well as biofilm dissolution to facilitate planktonic release of bacteria into another niche.”
The 2012 publication Impact of Pseudomonas aeruginosa quorum sensing on cellular wound healing responses in vitro relates “The virulent capacity of Pseudomonas aeruginosa can largely be ascribed to quorum sensing, i.e. the ability to evade host defence by a coordinated production and secretion of virulence factors. When P. aeruginosa is harboured in chronic wounds, a non-healing condition is often observed. In this study, we examined the in vitro cellular responses of the major cell types of re-epithelialization to supernatants of P. aeruginosa wild-type or an isogenic mutant not expressing quorum sensing-regulated virulence genes. We observed impairment of cell migration in keratinocytes (p = 0.009) and fibroblasts (p = 0.043) when supplementing medium with 20% P. aeruginosa culture supernatants. Cell proliferation was not significantly reduced, except for keratinocytes (p = 0.040). Data show compliance with in vivo observations of proliferating, non-motile epithelial cell behaviour in bacterially contaminated chronic wounds. Our findings suggest that quorum sensing may serve as an interesting target for controlling P. aeruginosa virulence in modern wound care.”
Quorum sensing communications from PA also affect expression in human cells.
The publication The Pseudomonas aeruginosa N-Acylhomoserine Lactone Quorum Sensing Molecules Target IQGAP1 and Modulate Epithelial Cell Migration relates “The human pathogen Pseudomonas aeruginosa and other bacteria communicate with each other using quorum sensing (QS). This is important for their growth, virulence, motility and the formation of biofilms. Furthermore, eukaryotic cells “listen and respond” to QS signaling, but the exact mechanisms and receptors on mammalian cells have not been identified.– We suggest that recognition of IQGAP1 by 3O-C12-HSL is a very early event in the communication between bacteria and human epithelial cells.”
Wound healing is a complex process requiring much signaling among human cells. Impaired wound healing can result from colonization of the wound by multiple biofilm-forming bacteria, not just Staphylococcus aureus and Pseudomonas aeruginosa, and the results of their communications. The process can be far from simple, involving simultaneous quorum sensing cross-talk among several species.
The treatise Understanding the effects of bacterial communities and biofilms on wound healing characterizes the complexity involved. “For survival and reproductive success, species of bacteria often rely on close relationships with other species. A collection of bacteria occupying the same physical habitat is called a ‘community’, and one example of a community of micro-organisms is a ‘biofilm’. Biofilms have been implicated in numerous chronic infections including cystic fibrosis, otitis media and prostatitis. Through interactions within a biofilm, the resident population of bacteria is likely to benefit from increased metabolic efficiency, substrate accessibility, enhanced resistance to environmental stress and inhibitors, and an increased ability to cause infection and disease. Dermal wounds often provide an ideal environment for bacteria to exist as a community, which may have a significant effect on wound healing. — Chronic wounds are invariably polymicrobial, yet most research to date has focused on the role of specific potential pathogens in wounds (eg Pseudomonas aeruginosa) rather than the effect of interactions between different species. Only recently have microbiologists begun to address and recognise the significance and importance of an interacting community and the relationship to the disease process. It is the intention of this review to address the potential role that microbial communities and associated biofilms may play in wound healing. — The composition (chemical, biological and physical) of a surface will determine the initial microbiology that develops at that surface. In the case of a recently formed wound, different micro-organisms from endogenous and exogenous sources will contaminate the wound surface. The properties of the wound surface will predetermine which micro-organisms will attach, grow and remain components of an early biofilm. The initial colonising bacteria on any surface are referred to as the ‘pioneering’ species. Colonising bacteria will modify the habitat and create a ‘microenvironment’ that encourages the attachment and growth of secondary colonising micro-organisms. If unchallenged, and with favourable conditions, a complex community of micro-organisms is likely to develop. Although such a continuum of events has been documented in the oral cavity, microbial development and succession in wounds have not been investigated. It has been proposed that as a microbial biofilm develops the community will ultimately form a more stable ‘climax community’ [7]. As the community is able to adapt appropriately to outside perturbations the term ‘microbial homeostasis’ has been suggested to reflect stability within a ‘climax community’ [7]. — Most infections are caused by a combination of micro-organisms (polymicrobial or mixed infection) [12]. In a wound environment, individual organisms may be unable to satisfy the requirements to cause disease and subsequently combine forces to do this. Examples of this type of situation have been documented for diseases of the gum and peridontium [13] and leg ulcer infections [8]. — In many animal models numerous combinations of anaerobic and aerobic bacteria have been shown to produce levels of sepsis or disease that could not be induced by individual species [13]. Such synergy has been demonstrated in wound pathogens such as: Prevotella melaninogenicus, Porphyromonas asaccharolytica and Peptostreptococcus micros [14]; Porphyromonas asaccharolytica and Klebsiella pneumoniae [8], [15]; Escherichia coli and Bacteriodes fragilis [8], [16] and Staphylococcus aureus [8]. — Mechanisms that constitute pathogenic synergy include communal protection from phagocytosis and intracellular killing, production of essential growth factors, modification of the local environment (eg redox potential) and the protection of sensitive species by the inactivation of inhibitors. This protection has been named ‘indirect pathogenicity’ [17]. In certain situations some pathogens are found to be antibiotic sensitive but are rendered ‘resistant’ by other members of the mixed infection. Treatment in these cases would be targeted at all of the component micro-organisms in a mixed infection [17], [18]. — Opportunistic pathogens are members of the resident microflora that become pathogenic by gaining access to sites that are not normally accessible to them, or by causing changes in the local environmental conditions that favour their prevalence within a community. In addition to synergy, organisms within a wound may interact via quorum sensing.”
Again, the keys to such an incredible process of coordination among species are vesicle/exosome communications and Quorum sensing.
“Quorum sensing involves communication strategies within a mixed community of organisms that ultimately enable them to coordinate their activities, and enhance their pathogenicity and ability to cause disease.
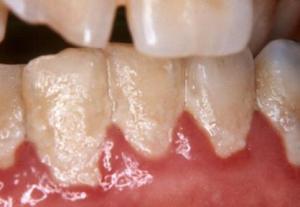
Quorum sensing plays a major role in communications among oral bacteria and is deeply indicated in the propagation of periodontal disease and dental caries. Multiple species of bacteria live in the mouth and can interact via quorum sensing so as to produce multi-species biofilms and induce dental caries or periodontal disease. The process is highly articulated.
Biofilm-formation can lead to infected swollen gums, recession of gums and loss of teeth and accelerated tooth decay.
The 2002 publication Communication among Oral Bacteria reports: “Human oral bacteria interact with their environment by attaching to surfaces and establishing mixed-species communities. As each bacterial cell attaches, it forms a new surface to which other cells can adhere. Adherence and community development are spatiotemporal; such order requires communication. The discovery of soluble signals, such as autoinducer-2, that may be exchanged within multispecies communities to convey information between organisms has emerged as a new research direction. Direct-contact signals, such as adhesins and receptors, that elicit changes in gene expression after cell-cell contact and biofilm growth are also an active research area. — Several oral bacterial species are amenable to genetic manipulation for molecular characterization of communication both among bacteria and between bacteria and the host. A successful search for genes critical for mixed-species community organization will be accomplished only when it is conducted with mixed-species communities. — Communication is a key element in successful organizations. The bacteria on human teeth and oral mucosa have developed the means by which to communicate and thereby form successful organizations. These bacteria have coevolved with their host to establish a highly sophisticated relationship in which both pathogenic and mutualistic bacteria coexist in homeostasis. The fact that human oral bacteria are not found outside the mouth except as pathogens elsewhere in the body (51) points to the importance of this relationship. Communication among microorganisms is essential for initial colonization and subsequent biofilm formation on the enamel surfaces of teeth and requires physical contact between colonizing bacteria and between the bacteria and their host. Without retention on the tooth surface, the bacteria are swallowed with the saliva. Through retention, these bacteria can form organized, intimate, multispecies communities referred to as dental plaque. — Sequential changes in populations of bacteria associated with tooth eruption (20, 21, 102, 138) as well as with caries development (53) and periodontal disease states (109, 136) are known. Temporal changes in populations of bacteria on tooth surfaces after professional cleaning are ordered and sequential (92, 114, 115). Such sequential changes must occur through attachment and growth of different bacterial species. With the attachment of each new cell type, a nascent surface is presented for the attachment of other kinds of bacteria, resulting in a progression of nascent surfaces and concomitant changes in species diversity (79, 137). Such coordination indicates communication. In the absence of communication, these orderly changes would be random. Due to the dynamics of growth and adherence, the bacterial populations in the oral cavity are constantly changing, even during the intervals between normal daily oral hygiene treatments. It is unlikely that the various species within oral biofilms function as independent, discrete constituents; rather, these organisms function as a coordinated community that uses intra- and interspecies communication. — For the past 40 years, pure cultures of oral bacteria have been isolated from supragingival and subgingival dental plaque removed from healthy and diseased sites. The numbers and variety of bacteria obtained from many clinical conditions have been catalogued (10, 19, 72, 109, 137). Estimates of the bacterial species diversity in the oral cavity, based on both culture-dependent methods (109, 136) and culture-independent methods (83, 123), indicate about 500 species. About 415 species are estimated to be present in subgingival plaque (123), and many of these are also found in supragingival plaque. Cultured species account for about 60% of the organisms identified by molecular methods, indicating that the oral cavity is an environment where most species can be studied by routine culture methods. This is distinct from other environments where less than 1% of the clones obtained by molecular methods represent cultured species (2, 44, 54, 118). Thus, dental plaque is one of the best-described mixed-species bacterial communities. Because it is easily accessible, it is convenient to study this complex model system.”
The 2008 publication Bacterial quorum sensing: signals, circuits, and implications for biofilms and disease reported: “Communication between bacteria, belonging to the same species or to different species, is mediated through different chemical signals that are synthesized and secreted by bacteria. These signals can either be cell-density related (autoinducers) or be produced by bacteria at different stages of growth, and they allow bacteria to monitor their environment and alter gene expression to derive a competitive advantage. The properties of these signals and the response elicited by them are important in ensuring bacterial survival and propagation in natural environments (e.g., human oral cavity) where hundreds of bacterial species coexist. First, the interaction between a signal and its receptor is very specific, which underlies intraspecies communication and quorum sensing. Second, when multiple signals are synthesized by the same bacterium, the signaling circuits utilized by the different signals are coordinately regulated with distinct overall circuit architecture so as to maximize the overall response. Third, the recognition of a universal communication signal synthesized by different bacterial species (interspecies communication), as well that of signals produced by eukaryotic cells (interkingdom communication), is also integral to the formation of multispecies biofilm communities that are important in infection and disease. The focus of this review is on the principles underlying signal-mediated bacterial communication, with specific emphasis on the potential for using them in two applications-development of synthetic biology modules and circuits, and the control of biofilm formation and infection.”
Again from Interactions between Oral Bacteria: Inhibition of Streptococcus mutans Bacteriocin Production by Streptococcus gordonii: “Streptococcus mutans has been recognized as an important etiological agent in human dental caries. Some strains of S. mutans also produce bacteriocins. In this study, we sought to demonstrate that bacteriocin production by S. mutans strains GS5 and BM71 was mediated by quorum sensing, which is dependent on a competence-stimulating peptide (CSP) signaling system encoded by the com genes. We also demonstrated that interactions with some other oral streptococci interfered with S. mutans bacteriocin production both in broth and in biofilms. The inhibition of S. mutans bacteriocin production by oral bacteria was stronger in biofilms than in broth. Using transposon Tn916 mutagenesis, we identified a gene (sgc; named for Streptococcus gordonii challisin) responsible for the inhibition of S. mutans bacteriocin production by S. gordonii Challis. Interruption of the sgc gene in S. gordonii Challis resulted in attenuated inhibition of S. mutans bacteriocin production. The supernatant fluids from the sgc mutant did not inactivate the exogenous S. mutans CSP as did those from the parent strain Challis. S. gordonii Challis did not inactivate bacteriocin produced by S. mutans GS5. Because S. mutans uses quorum sensing to regulate virulence, strategies designed to interfere with these signaling systems may have broad applicability for biological control of this caries-causing organism.”
Inhibiting quorum sensing among oral bacteria may be a strategy for prevention of dental caries and periodontal disease.
The 2010 publication Antimicrobial and antibiofilm activity of quorum sensing peptides and Peptide analogues against oral biofilm bacteria reports: “Widespread antibiotic resistance is a major incentive for the investigation of novel ways to treat or prevent infections. Much effort has been put into the discovery of peptides in nature accompanied by manipulation of natural peptides to improve activity and decrease toxicity. The ever increasing knowledge about bacteria and the discovery of quorum sensing have presented itself as another mechanism to disrupt the infection process. We have shown that the natural quorum sensing (QS) peptide, competence-stimulating peptide (CSP), used by the caries causing bacteria Streptococcus mutans when used in higher than normally present concentrations can actually contribute to cell death in S. mutans. Using an analogue of this quorum sensing peptide (KBI-3221), we have shown it to be beneficial at decreasing biofilm of various Streptococcus species. This chapter looks at a number of assay methods to test the inhibitory effects of quorum sensing peptides and their analogues on the growth and biofilm formation of oral bacteria.”
Sub-gingival biofilms formed on tooth surfaces have full multispecies complexity and are responsible for gum diseases and tooth decay
The 2009 publication Subgingival biofilm formation discusses this point.
More to come
This blog entry is long enough for now and, I believe, contains important messages. There is more to be said about quorum sensing and exosome communications, however. Topics I will likely cover in a future Part 3 quorum sensing blog entry include:
- roles of QS in cancer and how deregulated QS may lead to tumorigenesis,
- The importance of QS in organizing immune system responses, and
- The important roles of QS in stem cell maintenance and differentiation.



Pingback: Quorum sensing Part 1: quorum sensing inhibition via phytochemicals – a new approach against infectious diseases. | AGING SCIENCES – Anti-Aging Firewalls
Great article, though I miss practical recommendation herb/plant-wise. In “Quorum sensing Part 1” a range of compounds are mentioned that may inhibit quorum sensing directly, some are exotic, some are not:
* Garlic
* Caffeine
* Panax ginseng
* Cloves
Would it be possible (possibly in Quorum sensing Part 3) for you do a practical breakdown of which types of bacteria have their communication effectually inhibited by these “common” plants.
Jo:
Thanks for the suggestion. I will see what I can find with respect to your request, recognizing that experimental observations with respect to QS have been limited to only a very small number of bacteria. A general assumption, however, seems to be that bacterial QS mechanisms are widely shared among bacterial types – an assumption that may only be partially correct. There seems to be much still to be learned.
Vince
Vince
The reseach in gut flora seems to be an example of this inter and intra species communication. Our GI systems play an important role in our immune system, inflammation, cancer, and over all signals.
Note the balance on ketogenic diets VS high sugar and grain of the last 10,000 years made hyper in the last 150 to 50 year time frame. Not our grandfathers GI flora percentages? What is a more optimal balance and can it be achieved with diet? Sugars would be high on the list of usuual suspects to be avoided but what about all glucose and insulin spiking foods? can a simple test of the outcome (stool) of the GI flora tell us? maybe look for delta biomarkers in urine or breath or spit? One would thing inter and intra species communication would have varied biomarkers that could be screened with the brut force of modern high through put screens. Eric