By Vince Giuliano
Among infectious bacteria, talk is not cheap.
Quorum sensing (QS) is a fundamental biological process involving how cells in assemblages sense and communicate and cooperate with each other. It applies to bacterial colonies as well as to animal cells in organs. This Part 1 blog entry relates to survival-based signaling among bacteria. It is possible that the ravages of many infectious diseases can be averted by the use of substances to interrupt QS signaling. Plant-based substances hold particular promise in this regard. This blog covers current and recent research related to this hypothesis. Much of the relevant research is quite new and I cite several 2013 publications. A Quorum Sensing Part 2 blog entry is concerned with:
- The importance of molecular packing of signaling molecules into vesicles and exosomes,
- The highly social multi-bacteria nature of important infectious diseases,
- The roles of QS in biofilm formation,
- Molecular messaging of cells in response to infection and stresses,
- The role of QS in wound healing, and
- The roles of QS in periodontal disease and the formation of tooth cavities.
Along with the discovery of quorum sensing came the realization that infectious bacteria are highly social entities who cooperate in multiple ways for their survival. The 2013 publication Impacts of quorum sensing on microbial metabolism and human health provides an overview: “Bacteria were considered to be lonely ‘mutes’ for hundreds of years. However, recently it was found that bacteria usually coordinate their behaviors at the population level by producing (speaking), sensing (listening), and responding to small signal molecules. This so-called quorum sensing (QS) regulation enables bacteria to live in a ‘society’ with cell-cell communication and controls many important bacterial behaviors. In this chapter, QS systems and their signal molecules for Gram-negative and Gram-positive bacteria are introduced. Most interestingly, QS regulates the important bacterial behaviors such as metabolism and pathogenesis. QS-regulated microbial metabolism includes antibiotic synthesis, pollutant biodegradation, and bioenergy production, which are very relevant to human health. QS is also well-known for its involvement in bacterial pathogenesis, such as iin nfections by Pseudomonas aeruginosa and Staphylococcus aureus. Novel disease diagnosis strategies and antimicrobial agents have also been developed based on QS regulation on bacterial infections. In addition, to meet the requirements for the detection/quantification of QS signaling molecules for research and application, different biosensors have been constructed, which will also be reviewed here. QS regulation is essential to bacterial survival and important to human health. A better understanding of QS could lead better control/manipulation of bacteria, thus making them more helpful to people.”
What is quorum sensing? The basics related to bacteria
Quorum sensing (QS) is a signaling process used by many Gram-negative and Gram-positive bacteria to monitor their own population density and coordinate gene regulation so as to determine collective behavior. By knowing how many other bacterial cells are present, bacteria can trigger genes that radically affect individual cell behavior and as a result behavior of a whole colony of cells. For example a colony of bioluminescent bacteria can remain dark until quorum sensing determines there is a critical density of such bacteria, at which point the entire colony lights up at once. Another example of action that can result from a quorum sensing trigger point is cells starting to secrete substances that will make up a protective biofilm for a cell colony. Another cell action that can be triggered by quorum sensing in many bacteria is the release of cytotoxic chemicals that attack a host organism. Based on the quorum sensing, essentially all the bacteria switch at once to being aggressive warriors.. This strategy is used, for example by Pseudomonas aeruginosa, which causes infections in patients with compromised immune systems and cystic fibrosis. The bacteria do not launch an attack on a patient until their quorum sensing mechanisms tell them that they are in sufficient numbers to possibly succeed. Then they attack all at once. The evolutionary advantage to the bacteria is that they can hide out without being noticed in the host organism until they have multiplied sufficiently. If they don’t maintain a low profile, they could be wiped out in the initial stage by the hosts immune system. These are natural “sleeper cells.”
Signaling is via cell-secreted signaling molecules called autoinducers. Each autoinducer signaling molecule is highly specific to the bacterial species involved, and activates cell-membrane sensors in other bacteria of the same species. In the case of Gram-negative bacteria N-acyl homoserine lactone (AHL) autoinducer signal molecules are employed. Quorum-sensing circuits have been identified in over 25 species of Gram-negative bacteria. “Gram-positive bacteria employ secreted peptides processed from precursors, that can be used as autoinducers for QS — Signals are actively exported outside the cell, where they interact with the external domains of membrane-bound sensor proteins. The transduction of a signal generated by a phosphorylation cascade culminates in the activation of a DNA-binding protein, that influences the transcription of specific genes, so that each sensor protein is highly selective for a given peptide signal. In Gram-positive bacteria, a peptide signal precursor locus is translated into a precursor protein that is cleaved to produce the processed peptide autoinducer signal. This signal is then usually transported out of the cell via an ABC transporter(from Quorum Sensing and Phytochemicals, 2013).”
Quorum sensing is now recognized as a critical phenomenon in many infectious disease processes and Pubmed.org now lists 4194 research publications on QS going back to 1994. A lively and easily understandable video describing QS in bacteria can be found here.
There are additional kinds of quorum sensing and functions for it. An example is allowing members of a bacterial species to sense not only how many of its own kind are in the environment but also the total number of bacteria of any kind in the environment. This can allow the cell to feel out what the competitive environment is like and help it in deciding whether and when to trigger gene activation actions. Moreover, actions can go beyond biofilm formation or releasing biotoxic factors. The publication Quorum sensing and disease reports: “Much has been learned in the past decade about the complex behavior of bacteria under the influence of various Quorum Sensing systems. Many bacteria possess multiple QS systems, which control different aspects of virulence or other behaviors at different times in the infection process. And each bacteria species responds differently to QS stimuli, depending on environmental pressures, the presence or absence of competing bacterial species, the immune response of the host and other conditions. — For example, many bacteria form dense collections of cells called biofilms that are enclosed in a protective barrier. Such biofilms are highly resistant to host immune responses, anti-bacterial agents and other threats. The creation of these biofilms is regulated by QS signaling, and they generally are created after very large numbers of bacteria are present. — Some bacteria, however, use quorum sensing to end the biofilm stage as well as begin it. Cholera is one such example. In the final stages of infection, cholera uses QS signaling to dissolve existing biofilms and begin a massive dispersal of individual bacteria. This creates a highly infectious condition within the host and aids in cholera’s ability to spread. — There are many such examples of different bacteria responding to QS signaling in diverse ways at different times during the infection process. — What seems to be nearly universal, however, is the bacteria’s tendency to maintain as low a profile as possible when the host is first infected. It is believed that this behavior minimizes the host’s immune response while the bacteria build sufficient numbers to mount an effective offense. When a sufficient number is reached, the bacteria manifest their pathogenic (disease causing) arsenal. The timing of this transition reveals an exquisite genetic intelligence: when pathogenic bacteria have reached a density that would trigger the host’s full-scale immune response whether or not there existed a disease symptom stimulus, the bacteria, using QS signaling, begin their disease-causing phase. The disease stresses the host, thereby weakening the host’s ability to respond to the infection. This gives advantage to the bacteria.”
Among the actions resulting from quorum sensing signaling, certain bacteria can generate surfactants and extra flagella which facilitate bacterial “swarming” across solid surfaces(ref)(ref).
A diversity of related quorum sensing populations can exist even for a single pathogen.
The 2009 e-publication Instantaneous within-patient diversity of Pseudomonas aeruginosa quorum-sensing populations from cystic fibrosis lung infections reported: “To obtain insight into the instantaneous within-patient diversity of QS, we assayed a panel of 135 concurrent P. aeruginosa isolates from eight different adult CF patients (9 to 20 isolates per patient) for various QS-controlled phenotypes. Most patients contained complex mixtures of QS-proficient and -deficient isolates. Among all patients, deficiency in individual phenotypes ranged from 0 to about 90%. Acyl-HSL, sequencing, and complementation analyses of variants with global loss-of-function phenotypes revealed dependency upon the central QS circuitry genes lasR, lasI, and rhlI. Deficient and proficient isolates were clonally related, implying evolution from a common ancestor in vivo. Our results show that the diversity of QS types is high within and among patients, suggesting diverse selection pressures in the CF lung. A single selective mechanism, be it of a social or nonsocial nature, is unlikely to account for such heterogeneity. The observed diversity also shows that conclusions about the properties of P. aeruginosa QS populations in individual CF infections cannot be drawn from the characterization of one or a few selected isolates.”
There is much more to be said about quorum sensing in bacteria as will be unfolding in the course of this and the Part 2 blog entries.
To maintain perspective, we need to keep in mind that most microbes humans encounter are either positive or neutral for our health, and that quorum sensing is an evolutionarily-conserved property of many families of microbes, including all colony-forming ones.
Much of the research and literature related to quorum sensing has focused on pathological bacteria and how inhibiting quorum sensing might be a good strategy for averting or curing diseases. However, dealing with infectious diseases is only part of what must be taken into account to ensure health and longevity. Maintaining quorum sensing in beneficial bacteria is important too.
It is estimated that there are approximately a10 times more foreign cells in the human body than our actual human cells, some 100 trillion of them compared to 10 trillion of our own. These foreign cells are of thousands of species and exist in multiple biomes including the gut biome, the skin biome, the mouth biome, the vaginal biome, etc. I discussed just the gut biome in the blog entry Gut microbiota, probiotics, prebiotics and synbiotics – keys to health and longevity. A human typically carries thousands of species of such microbes, and the species can vary significantly depending on the local biome concerned. For example, the skin biomes for the left-hand and for the right hand may mostly consist of different bacteria. The “not us” microbes fall generally into three categories: 1. ones that exist in harmony with us that are known to play positive roles in health maintenance, 2. bacteria that generally play a positive or neutral role with respect to our health but which can turn to play negative roles(ref). Such bacteria are called commensal bacteria, where the bacteria depends on a symbiotic relationship with the host with little or no harm to the host. And 3. bacteria associated with pathologies, ones like Salmonella, Listeria, Pseudomonas, Staphylococcus and Lactobacillus. Traditional medical research has focused mainly on bacteria in category 3. The study of human biomes did not get really underway until the late 1990s, and the roles these biomes play with regard to our health are only now being explored.
In general, animal biomes and the bacteria in them have co-evolved. Bacteria have generally worked out a friendly or neutral symbiotic state with the host organism and with each other. Bacteria in the human biomes contribute in multiple ways to both our health and longevity. Many of these bacteria employ quorum sensing as part of their basic survival approach, certainly the ones that are colony-forming. Human cells and multiple bacteria may exist in the same organ, either cooperating with each other, indifferent or competing, and all using their own mechanisms of quorum sensing. In this blog entry, consistent with emphasis in the literature, we concentrate on the relatively small number of bacteria which are pathological, and focus on mechanisms for controlling or curing diseases via inhibiting quorum sensing. However, the last thing we want to do is inhibit quorum sensing across-the-board. Doing that would stop the actions of multiple beneficial microbes as well as essential QS among our own cells and probably be disastrous for health.
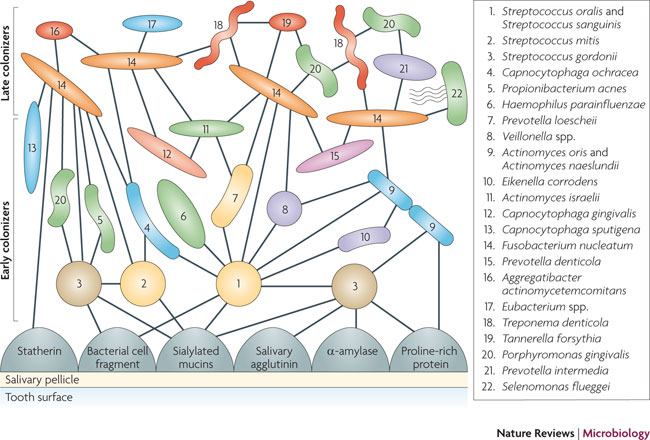
Image source Bacterial species involved with dental biofilm accumulation and tooth decay. All employ quorum sensing.
The mechanisms and activities of QS are being actively researched. QS is a hot topic.
For example, the June 2013 publication Novel quorum-sensing peptides mediating interspecies bacterial cell death reports: “Escherichia coli mazEF is a toxin-antitoxin stress-induced module mediating cell death. It requires the quorum-sensing signal (QS) “extracellular death factor” (EDF), the penta-peptide NNWNN (EcEDF), enhancing the endoribonucleolytic activity of E. coli toxin MazF. Here we discovered that E. coli mazEF-mediated cell death could be triggered by QS peptides from the supernatants (SN) of the Gram-positive bacterium Bacillus subtilis and the Gram-negative bacterium Pseudomonas aeruginosa. In the SN of B. subtilis, we found one EDF, the hexapeptide RGQQNE, called BsEDF. In the SN of P. aeruginosa, we found three EDFs: the nonapeptide INEQTVVTK, called PaEDF-1, and two hexadecapeptides, VEVSDDGSGGNTSLSQ, called PaEDF-2, and APKLSDGAAAGYVTKA, called PaEDF-3. When added to a diluted E. coli cultures, each of these peptides acted as an interspecies EDF that triggered mazEF-mediated death. Furthermore, though their sequences are very different, each of these EDFs amplified the endoribonucleolytic activity of E. coli MazF, probably by interacting with different sites on E. coli MazF. Finally, we suggest that EDFs may become the basis for a new class of antibiotics that trigger death from outside the bacterial cells. IMPORTANCE Bacteria communicate with one another via quorum-sensing signal (QS) molecules. QS provides a mechanism for bacteria to monitor each other’s presence and to modulate gene expression in response to population density. Previously, we added E. coli EDF (EcEDF), the peptide NNWNN, to this list of QS molecules. Here we extended the group of QS peptides to several additional different peptides. The new EDFs are produced by two other bacteria, Bacillus subtilis and Pseudomonas aeruginosa. Thus, in this study we established a “new family of EDFs.” This family provides the first example of quorum-sensing molecules participating in interspecies bacterial cell death. Furthermore, each of these peptides provides the basis of a new class of antibiotics triggering death by acting from outside the cell.”
A favorite bacterium for study of QS dynamics has been Chromobacterium violaceum –“a Gram-negative, facultative anaerobic, non-sporing coccobacillus. It is part of the normal flora of water and soil of tropical and sub-tropical regions of the world. It produces a natural antibiotic called violacein, which may be useful for the treatment of colon and other cancers.[1] It grows readily on nutrient agar, producing distinctive smooth low convex colonies with a dark violet metallic sheen (due to violacein production). Its full genome was published in 2003.[2] It has the ability to break down tarballs.[3] (ref)”(ref). For example, Chromobacterium violaceum CV026 is sometimes used as a reporter strain for measuring QS inhibitory activity of candidate substances.
Inhibiting quorum sensing could be a fast and effective way of stopping and clearing up an infectious disease. The need is very significant.
Starting more than10 years ago, there has been an increasing interest in averting or curing infectious diseases through inhibition of quorum sensing. One reason for this has been increasing resistance of strains of pathogenic bacteria to conventional antibiotics, and an inability of the pharmaceutical industry to develop conventional-antibiotics to replace ones that are no longer effective against bacteria which have evolved to becoming drug-resistant. Antibiotic resistance is a growing crises(ref). Another reason is that interfering with pathogen signaling seems to be novel, potentially both safe and effective, and a possible way to outwit bacteria who are smart at evolving.
The March 2013 publication Quorum sensing inhibitors: an overview provides a summary: “Excessive and indiscriminate use of antibiotics to treat bacterial infections has lead to the emergence of multiple drug resistant strains. Most infectious diseases are caused by bacteria which proliferate within quorum sensing (QS) mediated biofilms. Efforts to disrupt biofilms have enabled the identification of bioactive molecules produced by prokaryotes and eukaryotes. These molecules act primarily by quenching the QS system. The phenomenon is also termed as quorum quenching (QQ). In addition, synthetic compounds have also been found to be effective in QQ. This review focuses primarily on natural and synthetic quorum sensing inhibitors (QSIs) with the potential for treating bacterial infections. It has been opined that the most versatile prokaryotes to produce QSI are likely to be those, which are generally regarded as safe. Among the eukaryotes, certain legumes and traditional medicinal plants are likely to act as QSIs. Such findings are likely to lead to efficient treatments with much lower doses of drugs especially antibiotics than required at present.”
Another attractive aspect of QS inhibition as a potential strategy against infectious diseases appeared to be how quickly and effectively it could work, potentially with no side effects. Again, from Quorum sensing and disease: “The second, nearly universal trait, is that even after a bacterium has entered its pathogenic phase, it quickly returns to its non-pathogenic state when we disable its QS mechanisms. This is another clear sign of genetic intelligence, for when a bacterium in its pathogenic state is passed on to a new host, the new host would quickly locate and dispatch the invader if the invader continued in its pathogenic form. What happens when such transfers do occur is that, from the bacterium’s point of view, there is an immediate cessation of the quorum. This is the indication to the bacterium that it is in a new host, and that the infection process is starting over. All pathogenic activities of the bacterium then cease, giving the bacterium time to multiply and, later, mount another offense. This is the beauty of the anti-QS approach to the treatment of infection. It terminates the disease causing activities, thereby returning the body to strength and health. The body can then deal with the infection through its normal immune system responses, without inflammation or other deleterious effects. In contrast, current antibiotic treatments attempt to kill the bacteria while the body is in a state that is already compromised by the disease. The combination of the disease and the stress caused by many antibiotics can further weaken the system to dangerous levels.”
But the road was discovered to be bumpy. The 2004 document Quorum-sensing control in Staphylococci – a target for antimicrobial drug therapy? expresses this perception and warns of a case where QS inhibition did not appear to work: “Today, we find ourselves in an urgent need for novel antibacterial drugs, as many important human pathogens have acquired multiple antibiotic resistance factors. Among those, Staphylococcus aureus and S. epidermidis play a major role as the leading sources of nosocomial infections. Recently, it has been suggested to develop therapeutics that attack bacterial virulence rather than kill bacteria. Such drugs are called “antipathogenic” and are believed to reduce the development of antibiotic resistance. Specifically, cell-density-dependent gene regulation (quorum-sensing) in bacteria has been proposed as a potential target. While promising reports exist about quorum-sensing blockers in gram-negative bacteria, the use of the staphylococcal quorum-sensing system as a drug target is now seen in an increasingly critical way. Inhibition of quorum-sensing in Staphylococcus has been shown to enhance biofilm formation. Furthermore, down-regulation or mutation of the Staphylococcus quorum-sensing system increases bacterial persistence in device-related infection, suggesting that interference with quorum-sensing would enhance rather than suppress this important type of staphylococcal disease. The chemical nature and biological function of another proposed staphylococcal quorum-sensing inhibitor, named “RIP”, are insufficiently characterized. Targeting quorum-sensing systems might in principle constitute a reasonable way to find novel antibacterial drugs. However, as outlined here, this approach requires careful investigation in every specific pathogen and type of infection.”
It appears that a number of phytosubstances are effective at inhibiting QS.
It has been known for a long time that a number of phyto substances (plant-based substances) have been effective in combating a number of infectious diseases(ref)(ref). And this wisdom is basic in traditions of folk and herbal medicine. However, the mechanisms used by phytochemicals to do this have not been clear. Research during the last ten years indicates that many of these phytochemicals effectively inhibit quorum sensing in pathogenic target bacteria, as will be elaborated below. This hitherto unidentified mechanism could possibly become very important for dealing with infectious diseases. We have written in this blog extensively about the potential roles of phyto substances in averting cancer and aging(ref)(ref)(ref)(ref), epigenetic pathways through which phyto substances work(ref)(ref), and the potential role they could play in treating age-related neurological diseases(ref). This is the first blog entry, however, seriously relating phyto substances to infectious diseases.
The fact that phytochemicals, substances developed in plants as stress defenses, can also work in humans to deal with similar stresses via inhibition of QS is another example of Xenohormesis. See the discussion in the Xenohormesis section of this blog entry.
Starting around ten years ago, researchers have been looking into various medicinal plants and phytochemicals to determine whether they employ QS as an antimicrobial strategy. They have identified a rather large number of them. It appears that QS inhibition of pathogenic bacteria is an important survival strategy used by many plants.
The 2007 publication Dietary phytochemicals as quorum sensing inhibitors reported: “Quorum sensing (QS) is a cell density dependent expression of species in bacteria mediated by hormone like compounds called autoinducers (AI). Several processes responsible for successful establishment of bacterial infection are mediated by QS. Inhibition of QS is therefore being considered as a new target for antimicrobial chemotherapy. Dietary phytochemicals are secondary metabolites in plants known to have several health benefits including antimicrobial activity. However, their ability to inhibit QS has never been studied. Our objective was to investigate the effect of sub-lethal concentrations (SLC) of bioactive dietary phytochemical extracts from common dietary fruit, herb and spice extracts on modulating QS mediated by AI in model bioassay test systems. QS inhibition was measured in violacein pigment producing Chromobacterium violaceum O26 (CVO26) and CV 31532 system, mediated by AI known as acylated homeserine lactone (AHL). We also investigated the effect of the sub-lethal concentrations of the extracts on swarming motility of pathogens Escherichia coli (EC)O157:H7 and Pseudomonas aeruginosa (PA-01). Our results indicate that all extracts significantly inhibited quorum sensing. The mechanism of inhibition appeared to be combination of interfering with AHL activity and modulating the synthesis of AHL’s. Our results also indicated that various phytochemical extracts which inhibited QS also inhibited swarming of pathogenic bacteria, known to be modulated by QS. The observation that phytochemicals from foods can inhibit QS related processes opens up an exciting new strategy for antimicrobial chemotherapy and lead to the discovery of new category of antibiotics which can overcome the issues related to antimicrobial resistance.”
It makes sense. Because QS is a common evolutionary-driven strategy employed by pathogenic bacteria, plants in turn have evolved a broad repertory of phytosubstances which inhibit QS.
The May 2013 review Plant-derived natural products as sources of anti-quorum sensing compounds explains this point: “Just like animals and humans, plants are constantly exposed to bacterial infections, it is therefore logical to expect that plants have developed sophisticated chemical mechanisms to combat pathogens. In this review, we have surveyed the various types of plant-based natural products that exhibit anti-quorum sensing properties and their anti-quorum sensing mechanisms.”
The 2013 publication Quorum Sensing and Phytochemicals reviews mechanisms of QS employed by both gram-negative and gram-positive bacteria and discusses another important topic: the potential role of QS inhibition to slow or prevent food spoilage due to bacteria. “Most infectious diseases are caused by bacteria, which proliferate within quorum sensing (QS)-mediated biofilms. Efforts to block QS in bacteria and disrupt biofilms have enabled the identification of bioactive molecules that are also produced by plants. This mini review primarily focuses on natural QS inhibitors, which display potential for treating bacterial infections and also enhance the safety of food supply.” A great many specific phytochemicals are mentioned in the review.
A sample of recent research publications descriptive of or related to QS inhibition by phytosubstances is:
[Biotechnological cultivation of edible macrofungi: an alternative for obtaining nutraceutics].
Antibiofilm activity of Dendrophthoe falcata against different bacterial pathogens.
Anti-quorum sensing potential of crude Kigelia africana fruit extracts.
Anti-quorum sensing potential of the mangrove Rhizophora annamalayana.
Assessment of anti-quorum sensing activity for some ornamental and medicinal plants native to egypt.
Caffeine as a potential quorum sensing inhibitor.
Drimendiol, a drimane sesquiterpene with quorum sensing inhibition activity.
Eugenol inhibits quorum sensing at sub-inhibitory concentrations.
Microbial regulation in gorgonian corals.
Submerged cultivation of medicinal mushrooms: bioprocesses and products (review).
Synthesis of cembranoid analogues and evaluation of their potential as quorum sensing inhibitors.
Quorum sensing inhibitors: an overview.
Submerged cultivation of medicinal mushrooms: bioprocesses and products (review).
Synthesis of cembranoid analogues and evaluation of their potential as quorum sensing inhibitors.
Marine organisms like sponges might also be good sources of QS inhibitors that would work against human pathogens. The 2012 publication Inhibition of Quorum Sensing Mediated Virulence Factors Production in Urinary Pathogen Serratia marcescens PS1 by Marine Sponges reports: “The focal intent of this study was to find out an alternative strategy for the antibiotic usage against bacterial infections. The quorum sensing inhibitory (QSI) activity of marine sponges collected from Palk Bay, India was evaluated against acyl homoserine lactone (AHL) mediated violacein production in Chromobacterium violaceum (ATCC 12472), CV026 and virulence gene expressions in clinical isolate Serratia marcescens PS1. Out of 29 marine sponges tested, the methanol extracts of Aphrocallistes bocagei (TS 8), Haliclona (Gellius) megastoma (TS 25) and Clathria atrasanguinea (TS 27) inhibited the AHL mediated violacein production in C. violaceum (ATCC 12472) and CV026. Further, these sponge extracts inhibited the AHL dependent prodigiosin pigment, virulence enzymes such as protease, hemolysin production and biofilm formation in S. marcescens PS1. However, these sponge extracts were not inhibitory to bacterial growth, which reveals the fact that the QSI activity of these extracts was not related to static or killing effects on bacteria. Based on the obtained results, it is envisaged that the marine sponges could pave the way to prevent quorum sensing (QS) mediated bacterial infections.”
Another, March 2013, publication relates to marine organisms as sources for pathogen QS inhibitors: Attenuation of Pseudomonas aeruginosa virulence by marine invertebrate-derived Streptomyces sp.
Some conventional drugs also inhibit QS in pathogenic bacteria.
The antibiotic doxycycline appears to have interference with QS as one of its mechanisms of action. The publication Doxycycline interferes with quorum sensing-mediated virulence factors and biofilm formation in Gram-negative bacteria reports: “Inhibition of quorum sensing (QS)-regulated virulence factors including biofilm is a recognized anti-pathogenic drug target. The search for safe and effective anti-QS agents is expected to be useful to combat diseases caused by multidrug-resistant bacteria. In this study, effect of a commonly used antibiotic, doxycycline on QS was evaluated using sensor strains of Chromobacterium violaceum (ATCC 12472 and CVO26) and Pseudomonas aeruginosa PAO1. Sub-MICs of doxycycline reduced QS-controlled violacein production in C. violaceum to a significant degree (70 %) and showed a significant reduction of LasB elastase (67.2 %), pyocyanin (69.1 %), chitinase (69.8 %) and protease (65 %) production and swarming motility (74 %) in P. aeruginosa PAO1 over untreated controls. Similar results were also recorded against a clinical strain of P. aeruginosa (PAF-79). Interestingly, doxycycline at respective sub-MICs (4 and 32 μg ml(-1)) significantly reduced the biofilm-forming capability and exopolysaccharide production in both the strains of P. aeruginosa (PAO1 and PAF-79) over untreated controls. The results of this study highlight the multiple actions of doxycycline against QS-linked traits/virulence factors and its potential to attenuate virulence of P. aeruginosa.”
Also you can see New life for an old drug: the anthelmintic drug niclosamide inhibits Pseudomonas aeruginosa quorum sensing. “Microarray analysis showed that niclosamide affects the transcription of about 250 genes, with a high degree of target specificity toward the QS-dependent regulon. Phenotypic assays demonstrated that niclosamide suppresses surface motility and production of the secreted virulence factors elastase, pyocyanin, and rhamnolipids, and it reduces biofilm formation. In accordance with the strong antivirulence activity disclosed in vitro, niclosamide prevented P. aeruginosa pathogenicity in an insect model of acute infection.”
Some antibiotics, acting as signaling molecules, can in a concentration-dependent manner actually promote QS in bacteria
The April 2013 publication Antibiotics at subinhibitory concentrations improve the quorum sensing behavior of Chromobacterium violaceum relates: “Increasing evidence has shown that antibiotics function as intermicrobial signaling molecules instead of killing weapons. However, mechanisms and key factors that are involved in such functions remain poorly understood. Earlier findings have associated antibiotic signaling with quorum sensing (QS); however, results varied among experiments, antibiotics, and bacterial strains. In this study, we found that antibiotics at subinhibitory concentrations improved the violacein-producing ability of Chromobacterium violaceum ATCC 12472. Quantitative real-time polymerase chain reaction of QS-associated gene transcripts and bioassay of violacein production in a QS mutant strain demonstrated that antibiotics enhanced the production of N-acyl-L-homoserine lactones (AHLs; QS signaling molecules) and increased AHL-inducing QS-mediated virulence, including chitinase production and biofilm formation. Moreover, a positive flagellar activity and an increased bacterial clustering ability were found, which are related to the antibiotic-induced biofilm formation. Our findings suggested that antibiotic-mediated interspecific signaling also occurs in C. violaceum, thereby expanding the knowledge and language of cell-to-cell communication.” The implication is that some antibiotics may exercise their influence via a hormetic biphasic dose-response curve, killing bacteria when the dose is super-hormetic and acting as signaling molecules when the dose is in the low hormetic range.
There is also work on synthesizing compounds for inhibiting QS.
For example, the March 2013 publication Design, synthesis and biological evaluation of N-sulfonyl homoserine lactone derivatives as inhibitors of quorum sensing in Chromobacterium violaceum reports “A novel series of N-sulfonyl homoserine lactone derivatives 5a-l has been designed, synthesized and evaluated for quorum sensing inhibitory activities towards violacein production. Of the compounds synthesized, compound 5h was found to possess an excellent level of enantiopurity (99.2% e.e.). The results indicated that compounds bearing an ortho substituent on their phenyl ring exhibited excellent levels of inhibitory activity against violacein production. Compounds 5h and 5k in particular, with IC₅₀ values of 1.64 and 1.66 µM, respectively, were identified as promising lead compounds for further structural modification.”
There are other important applications of biofilm inhibition by inhibiting QS using natural substances
Inhibition of QS has uses beyond those addressing human bacterial pathologies. I have already mentioned the food-preservation application. There are also marine applications. An example is keeping boat bottoms and propellers free of fouling by colonies of marine bacteria, For example, the June 2013 publication Coral-associated bacteria, quorum sensing disrupters, and the regulation of biofouling reports: “Marine biofouling, the settlement of microorganisms and macroorganisms on structures submerged in seawater, although economically detrimental, is a successful strategy for survival in hostile environments, where coordinated bacterial communities establish biofilms via the regulation of quorum sensing (QS) communication systems. The inhibition of QS activity among bacteria isolated from different coral species was investigated to gain further insight into its potency in the attenuation, or even the prevention, of undesirable biofouling on marine organisms. It is hypothesized that coral mucus/microorganism interactions are competitive, suggesting that the dominant communities secrete QS disruptive compounds. One hundred and twenty bacterial isolates were collected from healthy coral species and screened for their ability to inhibit QS using three bioreporter strains. Approximately 12, 11, and 24% of the isolates exhibited anti-QS activity against Escherichia coli pSB1075, Chromobacterium violaceum CV026, and Agrobacterium tumefaciens KYC55 indicator strains, respectively. Isolates with positive activity against the bioluminescent monitor strains were scanned via a cytotoxic/genotoxic, E. coli TV1061 and DPD2794 antimicrobial panel. Isolates detected by C. violaceum CV026 and A. tumefaciens KYC55 reporter strains were tested for their ability to inhibit the growth of these reporter strains, which were found to be unaffected. Tests of the Favia sp. coral isolate Fav 2-50-7 (>98% similarity to Vibrio harveyi) for its ability to attenuate the formation of biofilm showed extensive inhibitory activity against biofilms of Pseudomonas aeruginosa and Acinetobacter baumannii. To ascertain the stability and general structure of the active compound, cell-free culture supernatants exposed to an increasing temperature gradient or to digestion by proteinase K, were shown to maintain potent QS attenuation and the ability to inhibit the growth of biofilms. Mass spectrometry confirmed the presence of a low molecular mass compound. The anti-QS strategy exemplified in the coral mucus is a model with potentially wide applications, including countering the ecological threat posed by biofilms. Manipulating synchronized bacterial behavior by detecting new QS inhibitors will facilitate the discovery of new antifouling compounds.”
Another publication related to QS inhibition for marine fouling is Quorum sensing inhibition by Asparagopsis taxiformis, a marine macro alga: separation of the compound that interrupts bacterial communication.
Although most discussions of QS in bacteria are framed in terms of free-floating signal molecules activating cell surface receptors, there is likely to be more to the picture. These signaling molecules may be in some or most instances packaged into vesicles which play a similar role to that played by exosomes in animal species.
From a 2008 news release in The Times Microbial Membrane Vesicles in Bacteria: Taking Quorum Sensing in New Directions: “Commonly, quorum sensing molecules are lactones, small peptides, or small lipids. It is thought that these small molecules are secreted by the cell and diffuse through the aqueous environment where they can interact with other cells. This is all well and good, IF the molecule of interest is hydrophillic and freely diffusible. But researchers are finding that many quorum sensing molecules are NOT hydrophillic, and instead very hydrophobic, such as a quinilone with a long chain fatty acid attached. One such example of this, is the Pseudomonas aeruginosa molecule called PQS. — How can a molecule be used as an extracellular signal, if it can not diffuse freely? Dr. Marvin Whitely and colleagues have shown that PQS is able to promote the formation of membrane vesicles off the outer Pseudomonas membrane. Pseudomonas is known to naturally produce these vesicles, however PQS directly induces changes in the lipid membrane to form such vesicles. —
Furthermore, these vesicles have the strong potential to be able to capture a variety of macromolecules that are in the vicinity of the membrane bleb. A prime example would be the beta-lactamases that are in the periplasmic space. But we could imagine other molecules being packaged up and delivered. — Molecules that are packaged could be delivered to the same cells in the population, or to foreign cells of different species, perhaps even to humans during pathogenesis. The possibilities are endless as to what these membrane vesicles could be providing instructions for. — One very important piece of data that is missing from this model, is that researchers have yet to observe membrane vesicles of one cell fusing with another cell. This would provide solid evidence that these vesicles could, in fact, deliver signals to other cells.”
See the 2005 publication Membrane vesicles traffic signals and facilitate group activities in a prokaryote: “Many bacteria use extracellular signals to communicate and coordinate social activities, a process referred to as quorum sensing1. Many quorum signals have significant hydrophobic character, and how these signals are trafficked between bacteria within a population is not understood. Here we show that the opportunistic human pathogen Pseudomonas aeruginosa packages the signalling molecule 2-heptyl-3-hydroxy-4-quinolone
(pseudomonas quinolone signal; PQS)2 into membrane vesicles that serve to traffic this molecule within a population. Removal of these vesicles from the bacterial population halts cell–cell communication and inhibits PQS-controlled group behaviour. We also show that PQS actively mediates its own packaging and the packaging of other antimicrobial quinolines produced by P. aeruginosa into vesicles. These findings illustrate that a prokaryote possesses a signal trafficking system with features common to those used by higher organisms and outlines a novel mechanism for delivery of a signal critical for coordinating group behaviour in P. aeruginosa.”
Clinicaltrials.gov shows four clinical trials related to QS, two completed, one terminated and one recruiting.
A few final words
The worlds of quorum sensing and quorum sensing inhibition appears to be just opening up and indeed may offer a powerful new set of weapons against infectious diseases. I am left with a few questions that may be reflective only of my ignorance or of the early state of research in this area.
- To what extent are the QS inhibitors generic across multiple species, and to what extent are they species–specific? A great many of the studies of QS inhibitors have focused on QS inhibition in only one or two species, namely Chromobacterium violaceum and Pseudomonas aeruginosa. This is well and good but leaves open the question of whether the substances equally inhibit QS in a host of other pathogens.
- To the extent that the QS inhibitors are generic, what are the implications of their use in vivo, given that they might also inhibit QS in beneficial bacteria?
- To what extent is the inhibition of quorum sensing the main mechanism of phytochemicals for combating infectious diseases? What other companion mechanisms may be involved?
- What are the extra-cellular vehicles for bacterial QS communications? Do they mainly involve free-floating signal molecules activating cell surface receptors, or are these signaling molecules frequently packaged into vesicles, perhaps even activating bacterial cells via endocytosis? If vesicles are involved, do they commonly carry multiple signaling molecules? I do know that in the case of human QS cell communications, exosome packaging may be involved.
There is much more to be said about quorum sensing and I will continue my discussion of this topic in the Quorum sensing Part 2 blog entry.



Pingback: Quorum Sensing Part 2 – Intra and inter-species molecular communications | AGING SCIENCES – Anti-Aging Firewalls