By Vince Giuliano with inputs from James P Watson, Victor, and Melody Winnig
Many of our most basic biological processes including gene activation are affected by circadian and other body and cell-level clock rhythms. In January 2012, Victor posted a blog entry Circadian Regulation,NMN, Preventing Diabetes, and Longevity – an introduction to the various clock processes affecting our cells and systems and to the relationship between epigenetics and circadian regulation. Among matters laid out there was Victor’s argument “The suggestion is that direct supplementation of NMN, which is the rate-limiting step in NAD production, could have therapeutic potential, while effectively bypassing the need for NAMPT and its potentially harmful, proinflammatory effects. ” That observation was published in January 2012, almost two years before the current focus on NMN supplementation triggered by David Sinclair’s important December 2013 paper.
This current blog entry highlights selected additional important insights and recent discoveries. Because the area of circadian rhythm-related research is so vast, I am forced to be very selective in deciding on the research to be covered here. I have possibly left out some important findings. One area I do touch seriously on is gene expression. It appears that expression of at least 10% of our genes is regulated by our biological clocks. So, the biological impacts of many things we do – including exercise and other interventions affecting aging – are highly dependent on the states of our clocks when we do them. And those actions may in turn affect the states and functioning of our clocks. Recent discovery after discovery brings home the importance of clock functions to our health and the necessity of respecting them. We are making discoveries about our mechanisms for modifying and resetting our clocks and gaining insights as to how clocks respond to metabolic conditions in cells. And a vast area of new research is just opening up related to how expression of various RNAs affect our circadian rhythms and vice-a-versa.
From the time when our original predecessors crawled out of the ocean onto land, it became important for organisms to coordinate and optimize their metabolic and other internal activities with daily rhythms of night and dark. This applied to both plants and animals. The eventual evolutionary result was the emergence of numerous biological clocks linked to master clocks in higher organisms like ourselves. Mechanisms developed for resetting, synchronizing and controlling these clocks. Reading on here, it should become clear that conforming our activities to respecting those clocks is important for maintaining our health and modulating our aging. However, our modern lifestyles involve many factors that can disrupt our regular circadian rhythms, including but not confined to: air travel over time zones, night work, irregular sleeping patterns, irregular and late-night meals, alcohol, binge eating and drinking, stimulants like caffeine, stresses, bright lights during normal dark night times, and exposure to bright TV and computer screens before bed. Even the color temperatures of our bedroom and bathroom lights may be relevant. And aging itself may be accompanied by or accelerated by clock dysregulation. That is the relevance of this blog topic.
Some basics in review
– Living biological organisms respond to multiple temporal rhythms, the science involved being Chronobiology. Chronobiological rhythms include daily, weekly, seasonal, and annual rhythms and special ones such as connected with tides and reproduction. The rhythms are coordinated by internal clocks on the cellular level, and it may be that the periodicity of one such clock in higher organisms is the entire lifespan.
– The most studied rhythms and the ones I focus on here are the daily (circadian) ones.
– “A circadian clock, or circadian oscillator, is a biochemical mechanism that oscillates with a period of 24 hours and is coordinated with the day-night cycle. Circadian clocks are the central mechanisms which drive circadian rhythms. Viewed classically, they consist of three major components: a) A central oscillator with a period of about 24 hours that keeps time, b). A series of input pathways to this central oscillator to allow entrainment of the clock, and c) A series of output pathways tied to distinct phases of the oscillator that regulate overt rhythms in biochemistry, physiology, and behavior throughout an organism(ref)”
– The clock is reset or regulated (entrained) “as the environment changes through an organism’s ability to sense external time cues of which the primary one is light. Circadian oscillators are ubiquitous in tissues of the body where they are synchronized by both endogenous and external signals to regulate transcriptional activity throughout the day in a tissue-specific manner.[1] The circadian clock is intertwined with most cellular metabolic processes and it is affected by organism aging.[2]
– the body has a number of circadian clocks and a master clock which to some extent are coordinated. “Although circadian rhythms are endogenous (“built-in”, self-sustained), they are adjusted (entrained) to the local environment by external cues called zeitgebers, commonly the most important of which is daylight(ref)”.
– “In mammals, the suprachiasmatic nucleus (SCN) of the hypothalamus constitutes the central circadian pacemaker. The SCN receives light signals from the retina and controls peripheral circadian clocks (located in the cortex, the pineal gland, the liver, the kidney, the heart, etc.). This hierarchical organization of the circadian system ensures the proper timing of physiological processes. In each SCN neuron, interconnected transcriptional and translational feedback loops enable the circadian expression of the clock genes(ref)”
– “The peripheral clocks are entrained by metabolic factors, like feeding and physical activity, while the central clock in the suprachiasmatic nucleus (SCN) of the hypothalamus is entrained by ambient conditions, such as light and to a lesser extent temperature(ref).”
As we see below, recent research challenges the above classical notions in certain respects. For example, the clocks do not necessarily respond to each other in a hierarchical manner. There appears to be clocks in every cell and the various clocks may or may not be synchronized to master clocks or to each other.
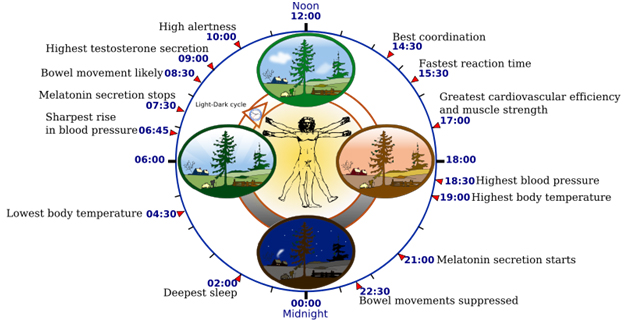
Image source “Biological clock affects the daily rhythm of many physiological processes. This diagram depicts the circadian patterns typical of someone who rises early in morning, eats lunch around noon, and sleeps at night (10 p.m.). Although circadian rhythms tend to be synchronized with cycles of light and dark, other factors – such as ambient temperature, meal times, stress and exercise – can influence the timing as well.”
Every human cell contains a 24-hour circadian clock which epigenetically regulates expression of thousands of genes.
The 2012 publication Transcriptional Architecture and Chromatin Landscape of the Core Circadian Clock in Mammals reports on the molecular clock mechanisms within cells: “The mammalian circadian clock involves a transcriptional feedback loop in which CLOCK and BMAL1 activate the Period and Cryptochrome genes, which then feed back and repress their own transcription. We have interrogated the transcriptional architecture of the circadian transcriptional regulatory loop on a genome scale in mouse liver and find a stereotyped, time-dependent pattern of transcription factor binding, RNA polymerase II (RNAPII) recruitment, RNA expression, and chromatin states. We find that the circadian transcriptional cycle of the clock consists of three distinct phases: a poised state, a coordinated de novo transcriptional activation state, and a repressed state. Only 22% of messenger RNA (mRNA) cycling genes are driven by de novo transcription, suggesting that both transcriptional and posttranscriptional mechanisms underlie the mammalian circadian clock. We also find that circadian modulation of RNAPII recruitment and chromatin remodeling occurs on a genome-wide scale far greater than that seen previously by gene expression profiling.”
The article goes on to explain what goes on in the course of a day: “The circadian clock in mammals is cell autonomous and is composed of an autoregulatory transcriptional network with Unterlocked feedback loops (1, 2). At the core, the basic helix-loop-helix–PER-ARNT-SIM (PAS) transcriptional activators BMAL1,CLOCK, and NPAS2 activate the Period (Per1 and Per2) and Cryptochrome (Cry1 and Cry2) genes, whose transcripts and proteins slowly accumulate during the daytime (3–7). The PER and CRY proteins associate and translocate into the nucleus during the evening and physically interact with the LOCK/NPAS2:BMAL1 complex to repress their own transcription (5, 7, 8). As the PER and CRY proteins are progressively phosphorylated during the night, they are targeted for ubiquitination by specific E3 ligases and are eventually degraded by the proteasome (9–12). The waxing and waning of this transcriptional feedback loop takes ~24 hours to complete and represents the core mechanism of the circadian clock in mammals. Biochemical analysis on a small set of target genes has shown that CLOCK, BMAL1, and CRY1 bind in a diurnal manner to Regulatory regions; interact with p300 andCREB-binding protein (CBP); and are accompanied by rhythmic changes in histone H3 Lys4 trimethylation (H3K4me3) and H3 Lys9 acetylation (H3K9ac) (13–19) characteristic of active promoter regions (20–23). — Although the majority of the core components of the circadian gene network are likely known (1) and many thousands of transcripts have been shown to express circadian oscillations in various tissues (24), the genome-wide architecture of the transcriptional network regulated by the core circadian clock remains to be defined (25, 26).” —-
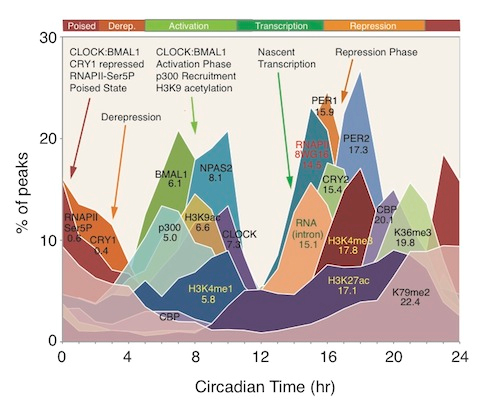
Image source “Circadian transcriptional landscape. Phase distributions of circadian transcriptional regulators, nascent RNA transcripts, RNA polymerase II (RAPII) occupancy, and histone modification rhythms in mouse liver. Mean circular phase of peak binding is indicated under the name.” © 2012 American Association for the Advancement of Science.
Most but not all circadian gene regulation is post-transcriptional.
“Although global circadian rhythms in steady-state mRNA levels have been known for a decade (35, 41, 42), here we find that only ~22% of cycling mRNA transcripts are driven by transcription (Fig. 2). Therefore, posttranscriptional regulatory events must contribute significantly to the generation of steady-state cycling mRNA levels (46, 47). Unexpectedly the most pervasive circadian regulation observed on a genome scale are rhythms in RNAPII recruitment and initiation, H3K4me3, H3K9ac, and H3K27ac, which occur at thousands of expressed genes whether or not RNA cycling was detectable. What accounts for these genome-wide circadian rhythms in RNAPII occupancy and histone modifications? Examination of the correlation between circadian transcription factor occupancy and gene expression shows that about 90% of genes bound by these factors are expressed whereas only 1 to 5% of unexpressed genes are similarly bound (table S10). These results demonstrate that gene expression per se, rather than rhythmicity of gene expression, is tightly correlated with circadian transcription factor binding. Rhythmic circadian transcription factor occupancy in turn could then be responsible for RNAPII recruitment and initiation on a genome-wide basis, which would then lead to the global rhythmic histone modifications seen here. Thus, circadian transcriptional regulators appear to be involved in the initial stages of RNAPII recruitment and initiation and the histone modifications associated with these events to set the stage for gene expression on a global scale, but additional control steps must then determine the ultimate transcriptional outputs from these sites(ref).”
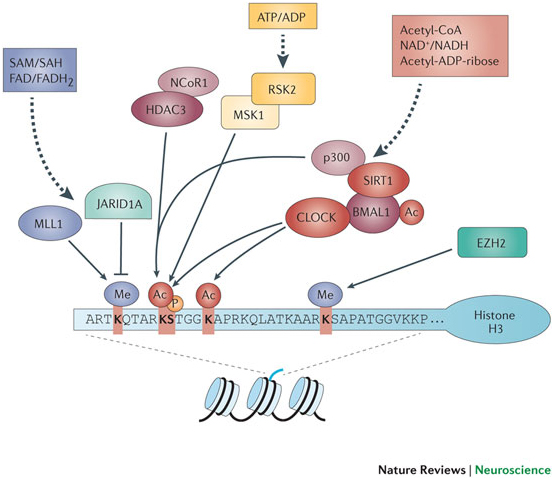
Image source “Schematic representation of the histone H3 tail, the relevant post-translational modifications and the chromatin remodellers involved in circadian control. At least 10% of the genes in any given cell are expressed in a cyclic manner under circadian control mediated by cyclic chromatin modifications at the promoters of clock-controlled genes. Phosphorylation (P; serine 10 (S10)), acetylation (Ac; lysine 9/14 (K9/14)) and methylation (Me; K4 and K27) are associated with circadian transcription. Some chromatin modifiers may be directly or indirectly modulated by the circadian system (Table 1). Methylation at K4 is consistently associated with gene activation and might be crucial for circadian gene transcription and recruitment of circadian chromatin remodelling complexes87. Non-histone proteins can also undergo clock-dependent acetylation, as is the case for BMAL1 (Table 1). Several chromatin remodellers can be considered as metabolic sensors, as they use metabolites for their enzymatic function; one example is the NAD+-dependent deacetylase sirtuin 1 (SIRT1), which revealed a link between the circadian clock and cellular metabolism through its role in circadian control and its physical interaction with CLOCK. For enzymatic functions of these chromatin modifiers and references, see Table 1. See Refs 88,89 for further details. EZH2, enhancer of zeste homologue 2; FAD, flavin adenine dinucleotide, quinone form; FADH2, flavin adenine dinucleotide, hydroquinone form; HDAC, histone deacetylase; JARID1A, Jumonji/ARID domain-containing protein 1A; MLL1, mixed lineage leukaemia protein 1; MSK1, mitogen- and stress-activated protein kinase 1; NCoR1, nuclear receptor co-repressor 1; RSK2, ribosomal S6 kinase 2; SAH, S-adenosylhomocysteine; SAM, S-adenosylmethionine.”
Clock genes exercise epigenetic control of gene expression on clock-controlled genes via chromatin remodeling such as DNA methylation or histone tail modifications
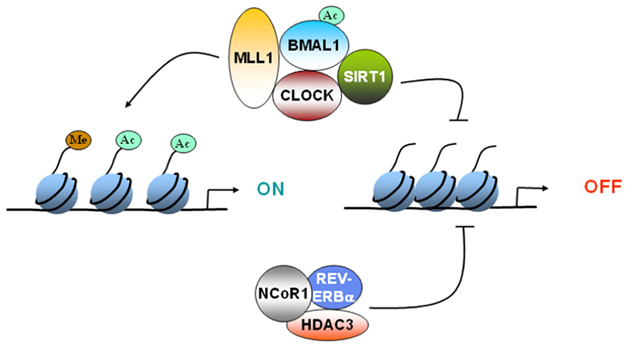
The 2012 publication Circadian rhythms and memory formation: regulation by chromatin remodeling describes the basic mechanism through which clock genes exercise epigenetic control of gene expression on clock controlled genes via chromatin remodeling: “Epigenetic changes, such as DNA methylation or histone modification, can remodel the chromatin and regulate gene expression. Remodeling of chromatin provides an efficient mechanism of transducing signals, such as light or nutrient availability, to regulate gene expression. CLOCK:BMAL1 mediated activation of clock-controlled genes (CCGs) is coupled to circadian changes in histone modification at their promoters. Several chromatin modifiers, such as the deacetylases SIRT1 and HDAC3 or methyltransferase MLL1, have been shown to be recruited to the promoters of the CCGs in a circadian manner. Interestingly, the central element of the core clock machinery, the transcription factor CLOCK, also possesses histone acetyltransferase activity. Rhythmic expression of the CCGs is abolished in the absence of these chromatin modifiers. Recent research has demonstrated that chromatin remodeling is at the cross-roads of circadian rhythms and regulation of metabolism and aging. It would be of interest to identify if similar pathways exist in the epigenetic regulation of memory formation.”
Image source “FIGURE 1. Epigenetic regulation of gene expression by circadian clock. CLOCK can acetylate histones to induce gene expression. CLOCK interacts with MLL1 (a histone methyltransferase) and SIRT1 (a deacetylase).These epigenetic regulators can modify the chromatin according the environmental stimuli, such as nutrient availability. Furthermore, REV-ERBα, a clock-controlled gene, can cause recruitment of HDAC3 and deacetylate histones. Circadian regulation of either the expression or the activity of these epigenetic regulators determines whether the gene gets turned “ON” or “OFF.”
The circadian rhythms associated with sleep are very important for the health of humans and have been studied from multiple perspectives
There is extensive body of research literature relating to circadian rhythms, sleep, sleep disorders and related pathologies(ref). There are various types of circadian rhythm sleep disorders, such as outlined in this document from the American Academy of Sleep Medicine.
Insufficient or poor sleep can have a lasting consequence on gene expression
The 2013 publication Effects of insufficient sleep on circadian rhythmicity and expression amplitude of the human blood transcriptome has this reported significance: “Insufficient sleep and circadian rhythm disruption are associated with negative health outcomes, but the mechanisms involved remain largely unexplored. We show that one week of insufficient sleep alters gene expression in human blood cells, reduces the amplitude of circadian rhythms in gene expression, and intensifies the effects of subsequent acute total sleep loss on gene expression. The affected genes are involved in chromatin remodeling, regulation of gene expression, and immune and stress responses. The data imply molecular mechanisms mediating the effects of sleep loss on health and highlight the interrelationships between sleep homeostasis, circadian rhythmicity, and metabolism.” The abstract of the publication is “Insufficient sleep and circadian rhythm disruption are associated with negative health outcomes, including obesity, cardiovascular disease, and cognitive impairment, but the mechanisms involved remain largely unexplored. Twenty-six participants were exposed to 1 wk of insufficient sleep (sleep-restriction condition 5.70 h, SEM = 0.03 sleep per 24 h) and 1 wk of sufficient sleep (control condition 8.50 h sleep, SEM = 0.11). Immediately following each condition, 10 whole-blood RNA samples were collected from each participant, while controlling for the effects of light, activity, and food, during a period of total sleep deprivation. Transcriptome analysis revealed that 711 genes were up- or down-regulated by insufficient sleep. Insufficient sleep also reduced the number of genes with a circadian expression profile from 1,855 to 1,481, reduced the circadian amplitude of these genes, and led to an increase in the number of genes that responded to subsequent total sleep deprivation from 122 to 856. Genes affected by insufficient sleep were associated with circadian rhythms (PER1, PER2, PER3, CRY2, CLOCK, NR1D1, NR1D2, RORA, DEC1, CSNK1E), sleep homeostasis (IL6, STAT3, KCNV2, CAMK2D), oxidative stress (PRDX2, PRDX5), and metabolism (SLC2A3, SLC2A5, GHRL, ABCA1). Biological processes affected included chromatin modification, gene-expression regulation, macromolecular metabolism, and inflammatory, immune and stress responses. Thus, insufficient sleep affects the human blood transcriptome, disrupts its circadian regulation, and intensifies the effects of acute total sleep deprivation. The identified biological processes may be involved with the negative effects of sleep loss on health, and highlight the interrelatedness of sleep homeostasis, circadian rhythmicity, and metabolism.”
Circadian epigenetic clocks determine the best times for expressions of various genes and coordination of metabolic activities. Failure to respect those times, like insufficient sleep, can lead to disorders and diseases.
The 2013 publication Epigenetic regulation of the molecular clockwork reports “Circadian rhythms control a wide range of physiological events in all organisms. Typical of our modern lifestyles is the flexibility to rest, exercise, eat, or socialize at any time of the circadian day or night; yet, these allowances correlate with rising disorders of a metabolic nature, which are thought to be mediated by changes in the molecular events underlying metabolic gene expression. Because the clock confers on gene expression changes in activity that are not necessarily related to changes in DNA sequence, the study of circadian rhythms is inseparable from epigenetics. Increasingly evident is that energy balance at the systems level relies on precise and collaborative circadian timing of epigenetic events within individual cells and tissues of the body. At the center of these rhythms resides the circadian clock machinery, a remarkably well-orchestrated transcription-translation feedback system that incorporates a fluctuating landscape of mRNA expression, protein stability, chromatin state, and metabolite abundance to keep correct time. Understanding more fully the ties that exist between cellular metabolism and the circadian clock at the epigenetic level will produce not only needed insights about circadian physiology but also novel strategies for the pharmacological and nonpharmacological treatment of metabolic disorders.”
The 2013 publication The circadian clock: a framework linking metabolism, epigenetics and neuronal function reports” ““The circadian clock machinery is responsible for biological timekeeping on a systemic level. The central clock system controls peripheral clocks through a number of output cues that synchronize the system as a whole. There is growing evidence that changing cellular metabolic states have important effects on circadian rhythms and can thereby influence neuronal function and disease. Epigenetic control has also been implicated in the modulation of biological timekeeping, and cellular metabolism and epigenetic state seem to be closely linked. We discuss the idea that cellular metabolic state and epigenetic mechanisms might work through the circadian clock to regulate neuronal function and influence disease states.”
Dysregulation of our epigenetic clocks or lifestyle behaviors that do not respect them can lead to a wide variety of disease susceptibilities and processes
This point was made in Victor’s earlier blog entry with respect to Type 2 diabetes. It has also been mentioned in other blog entries such as The pivotal role of Nrf2. Part 1 – a new view on the control of oxidative damage and generation of hormetic effects, and New, emerging and potential treatments for cancers: Part 1 – focus on the mTOR pathway. A plethora of research publications relate circadian patterns to most if not all important disease processes, particularly those characteristic of old age. Most of these 328 publications relate circadian rhythms and sleep deprivation to Parkinson’s disease. A Pubmed search on “circadian rhythms cancer” currently produces 2623 research publications. A Pubmed search on “circadian rhythms Alzheimers” currently produces 293 research publication citations. Many of these were published in the last two years and I note that these numbers go up daily..
Exposure to blue or ultraviolet-intensive light at the wrong time can also disrupt light-entrained rhythms and lead to pathologies, as pointed out in the 2012 blog entry Blue light, sleep, mental alertness and health.
It appears that our epigenetic clocks track if not to some extent regulate organismal aging.
There is a growing consensus that our circadian clocks have a lot to do with aging. A Pubmed search on “circadian rhythms aging” produces 2786 research publication citations. (9 more today than the same search produced yesterday). The September 2013 publication How does the body know how old it is? Introducing the epigenetic clock hypothesis reports “Animals and plants have biological clocks that help to regulate circadian cycles, seasonal rhythms, growth, development, and sexual maturity. It is reasonable to suspect that the timing of senescence is also influenced by one or more biological clocks. Evolutionary reasoning first articulated by G. Williams suggests that multiple, redundant clocks might influence organismal aging. Some aging clocks that have been proposed include the suprachiasmatic nucleus, the hypothalamus, involution of the thymus, and cellular senescence. Cellular senescence, mediated by telomere attrition, is in a class by itself, having recently been validated as a primary regulator of aging. Gene expression is known to change in characteristic ways with age, and in particular DNA methylation changes in age-related ways. Herein, I propose a new candidate for an aging clock, based on epigenetics and the state of chromosome methylation, particularly in stem cells. If validated, this mechanism would present a challenging target for medical intervention.”
Aging can lead to dysregulation of biological clock systems resulting in immune system impairment and increased disease susceptibilities.
The 2014 publication Chronobiology of the Neuroimmunoendocrine System and Aging reports: “The health maintenance depends on the preservation of the homeostatic systems, such as nervous, endocrine and immune system, and a proper communication between them. In this regard, the circadiansystem, which promotes a better functionality of physiological systems and thus well being, could be considered part of that homeostatic complex, since the neuroimmunoendocrine system possesses circadian patterns in most variables, as well as circannual or seasonal variations. With aging, an impairment of the homeostatic systems occurs and an alteration of circadian system regulation has been demonstrated. In the immune system, several function parameters, which are good markers of health and of the rate of aging, change not only with age (immunosenescence) but also throughout the day and year. Indeed, with advancing age there is a modification of immune cell circadian functionality especially in lymphocytes. Moreover, immune functions at early afternoon correspond to more aged values than at morning, especially in mature subjects (60-79 years of age). In addition, these mature men and women showed a significant impaired immune cell function, which is especially remarkable in the winter. It is noteworthy the role of immunomodulatory hormones, such as melatonin, in the regulation of biologicalrhythms and their involvement in the aging process. Furthermore, the evidence of a neuroimmune regulation of the circadian system and its disturbance with aging, highlights the importance of proinflammatory cytokines in this complex cross-talk. The biological rhythms disruption with age and some diseases (jet lag, cancer and seasonal affective disorder), could contribute increasing the immune system impairment and consequently the loss of health.”
The 2013 publication Noninvasive fractal biomarker of clock neurotransmitter disturbance in humans with dementia reports: “Human motor activity has a robust, intrinsic fractal structure with similar patterns from minutes to hours. The fractal activity patterns appear to be physiologically important because the patterns persist under different environmental conditions but are significantly altered/reduced with aging and Alzheimer’sdisease(AD). Here, we report that dementia patients, known to have disrupted circadian rhythmicity, also have disrupted fractal activity patterns and that the disruption is more pronounced in patients with more amyloid plaques (a marker of AD severity). Moreover, the degree of fractal activity disruption is strongly associated with vasopressinergic and neurotensinergic neurons (two major circadian neurotransmitters) in postmortem suprachiasmatic nucleus (SCN), and can better predict changes of the two neurotransmitters than traditional circadian measures. These findings suggest that the SCN impacts human activity regulation at multiple time scales and that disrupted fractal activity may serve as a non-invasive biomarker of SCN neurodegeneration in dementia.”
The Complex (Epigenetic) Explanation for Sleep
The “Circadian Cistrome” (aka Epigenetic explanations for the necessity of sleep): Gene expression is regulated by epigenetic mechanisms by a molecular 24-hour clock
The epigenetic explanation for why sleep is so important is probably a much more comprehensive explanation than others, but is not easy to understand. It has to do with the epigenetic regulation of genes based on circadian rhythms. The details of this have recently been worked out by Dr. Joesph Takahashi, a researcher at UT Southwestern. Takahashi and his colleagues generated the above-mentioned paper Transcriptional Architecture and Chromatin Landscape of the Core Circadian Clock in Mammals. Sleep is important because it is important for healthy gene expression, and sustained lack of sleep can have long-lasting negative consequences relating to gene expression,
The chronobiological responses of men and women are somewhat different.
The 2014 publication Chronotype ontogeny related to gender reports: “Chronotype is an established concept designed to identify distinct phase relationships between the expression of circadian rhythms and external synchronizers in humans. Although it has been widely accepted that chronotype is subjected to ontogenetic modulation, there is no consensus on the interaction between age and gender. This study aimed to determine the relationship between age- and gender-related changes in the morningness-eveningness character in a large sample of people. A total of 14,650 volunteers were asked to complete the Brazilian version of the Horne and Östberg chronotype questionnaire. The data demonstrated that, on average, women were more morning-oriented than men until the age of 30 and there were no significant differences between men and women from 30 to 45 years of age. In contrast to the situation observed until the age of 30, women older than 45 years were more evening-oriented than men. These results suggest that the ontogenetic development of thecircadian timekeeping system is more plastic in men, as represented by the larger amplitude of chronotype changes throughout their aging process. The phase delay of adolescence and phase advance of the elderly seem to be phenomena that are more markedly present in men than in women. Thus, our data, for the first time, provide support that sharply opposes the view that there is a single path toward morningness as a function of age, regardless of gender.”
Sleep deprivation is one well-studied intervention that prevents circadian entrainment and that can lead to multiple pathologies.
In mouse pancreatic cells at least, sleep deprivation stress is associated with improper protein folding and induction of the unfolded protein response (UPR) in the endoplasmic reticulum and upregulation of apoptosis. One consequence in older animals can be hyperglycemia.
The November 2013 publication Aging and sleep deprivation induce the unfolded protein response in the pancreas: implications for metabolism reports: “Sleep disruption has detrimental effects on glucose metabolism through pathways that remain poorly defined. Although numerous studies have examined the consequences of sleep deprivation (SD) in the brain, few have directly tested its effects on peripheral organs. We examined several tissues in mice for induction of the unfolded protein response (UPR) following acute SD. In young animals, we found a robust induction of BiP in the pancreas, indicating an active UPR. At baseline, pancreata from aged animals exhibited a marked increase in a pro-apoptotic transcription factor, CHOP, that was amplified by SD, whereas BiP induction was not observed, suggesting a maladaptive response to cellular stress with age. Acute SD increased plasma glucose levels in both young and old animals. However, this change was not overtly related to stress in the pancreatic beta cells, as plasma insulin levels were not lower following acute SD. Accordingly, animals subjected to acute SD remained tolerant to a glucose challenge. In a chronic SD experiment, young mice were found to be sensitized to insulin and have improved glycemic control, whereas aged animals became hyperglycemic and failed to maintain appropriate plasma insulin concentrations. Our results show that both age and SD cooperate to induce the UPR in pancreatic tissue. While changes in insulin secretion are unlikely to play a major role in the acute effects of SD, CHOP induction in pancreatic tissues suggests that chronic SD may contribute to the loss or dysfunction of endocrine cells and that these effects may be exacerbated by normal aging.”
As explained in a Penn University news release Sleep-Deprived Mice Show Connections among Lack of Shut-Eye, Diabetes, Age: : “During sleep deprivation cells upregulate the UPR — the unfolded protein response — a process where misfolded proteins get refolded or degraded.
Five years ago, researchers at the Perelman School of Medicine, University of Pennsylvania, showed that the UPR is an adaptive response to stress induced by sleep deprivation and is impaired in the brains of old mice. Those findings suggested that inadequate sleep in the elderly, who normally experience sleep disturbances, could exacerbate an already-impaired protective response to protein misfolding that happens in aging cells. Protein misfolding and clumping is associated with many diseases such as Alzheimer’s and Parkinson’s, noted Nirinjini Naidoo, Ph.D., research associate professor in the Division of Sleep Medicine in that study.
Naidoo is also senior author of a follow-up study in Aging Cell this month that shows, for the first time, an effect of sleep deprivation on the UPR in peripheral tissue, in this case, the pancreas. They showed that stress in pancreatic cells due to sleep deprivation may contribute to the loss or dysfunction of these cells important to maintaining proper blood sugar levels, and that these functions may be exacerbated by normal aging. — “The combined effect of aging and sleep deprivation resulted in a loss of control of blood sugar reminiscent of pre-diabetes in mice,” says Naidoo. “We hypothesize that older humans might be especially susceptible to the effects of sleep deprivation on the disruption of glucose homeostasis via cell stress.” — Working with Penn colleague Joe Baur, Ph.D., assistant professor of Physiology, Naidoo started a collaboration to look at the relationship of sleep deprivation, the UPR, and metabolic response with age. Other researchers had suggested that the death of beta cells associated with type 2 diabetes may be due to stress in a cell compartment called the endoplasmic reticulum (ER). The UPR is one part of the quality control system in the ER, where some proteins are made. Knowing this, Naidoo and Baur asked if sleep deprivation (SD) causes ER stress in the pancreas, via an increase in protein misfolding, and in turn, how this relates to aging. The team examined tissues in mice for cellular stress following acute SD, and they also looked for cellular stress in aging mice. Their results show that both age and SD combine to induce cellular stress in the pancreas.
Older mice fared markedly worse when subjected to sleep deprivation. Pancreas tissue from older mice or from young animals subjected to sleep deprivation exhibited signs of protein misfolding, yet both were able to maintain insulin secretion and control blood sugar levels. Pancreas tissue from acutely sleep-deprived aged animals exhibited a marked increase in CHOP, a protein associated with cell death, suggesting a maladaptive response to cellular stress with age that was amplified by sleep deprivation. Acute sleep deprivation caused increased plasma glucose levels in both young and old animals. However, this change was not overtly related to stress in beta cells, since plasma insulin levels were not lower following acute lack of sleep. — Accordingly, young animals subjected to acute sleep deprivation remained tolerant to a glucose challenge. In a chronic sleep deprivation experiment, young mice were sensitized to insulin and had improved control of their blood sugar, whereas aged animals became hyperglycemic and failed to maintain appropriate plasma insulin concentrations.
While changes in insulin secretion are unlikely to play a major role in the acute effects of SD, cellular stress in pancreatic tissue suggests that chronic SD may contribute to the loss or dysfunction of endocrine cells, and that these effects may be exacerbated by normal aging, say the researchers.”
Newer techniques of genetic and epigenetic profile and computer mathematical systems modeling are being applied to study the impacts of circadian rhythms on gene expression
The 2013 publication Circadian rhythms investigated on the cellular and molecular levels reports “Investigations on circadian rhythms have markedly advanced our understanding of health and disease with the advent of high-throughput technologies like microarrays and epigenetic profiling. They elucidated the multi-level behaviour of interactive oscillations from molecules to neuronal networks and eventually to processes of learning and memory in an impressive manner. The small-world topology of synchronized firing through neuron-neuron and neuron-glia gap junctions is discussed as a mathematical approach to these intensively studied issues. It has become evident that, apart from some disorders caused by gene mutations, the majority of disorders originating from disturbances of rhythms arise from environmental influences and epigenetic changes. In this context, it was mandatory to think of and devise experiments on temporary scales, which exponentially increased the volumes of data obtained from time-series and rapidly became prohibitive of manual inspection. Therefore, more and more sophisticated mathematical algorithms have been developed to identify rhythmic expression of genes and to find coexpression by their clustering. It is expected that disturbed rhythmic behaviour in mental disorders is reflected in altered oscillatory behaviour of gene expression.”
The networks that relate clock gene, circadian rhythms and gene activation in organs and individual networks and within neurons themselves are very complex and are being actively studied using computer and mathematical modeling approaches.
These 2012 publications Modeling the emergence of circadian rhythms in a clock neuron network describes “a model for a network of interacting clock neurons to describe the emergence (or damping) of circadian rhythms in Drosophila fly, in the absence of zeitgebers.” The 2012 publication Effect of network architecture on synchronization and entrainment properties of the circadian oscillations in the suprachiasmatic nucleus describes a mathematical model that accounts for the heterogeneous organization of the SCN is presented and used to study the implication of the SCN network topology on synchronization and entrainment properties.
The interactions among clocks is complex and not hierarchical
It has been thought that body clocks function in a somewhat hierarchical manner with one or a few “master” clocks controlling the other ones. That model is probably not correct with different clocks being able to come to the fore and control others depending on state, state dynamics, and circumstances. At least this appears to be the case in the case of clock neurons in the fruit fly. The 2014 publication The Drosophila Circadian Clock Is a Variably Coupled Network of Multiple Peptidergic Units reports “Daily rhythms in behavior emerge from networks of neurons that express molecular clocks. Drosophila’s clock neuron network consists of a diversity of cell types, yet is modeled as two hierarchically organized groups, one of which serves as a master pacemaker. Here, we establish that the fly’s clock neuron network consists of multiple units of independent neuronal oscillators, each unified by its neuropeptide transmitter and mode of coupling to other units. Our work reveals that the circadian clock neuron network is not orchestrated by a small group of master pacemakers but rather consists of multiple independent oscillators, each of which drives rhythms in activity.” The Editor’s Summary states “Circadian rhythms in the fruit fly Drosophilia are driven by neurons in the brain. Yao and Shafer (p. 1516) analyzed different sets of neurons that can drive circadian rhythms. Manipulating the period of each set of neurons separately revealed that when the various clock signals were fairly consistent, the fly showed a robust circadian rhythm. But when the various clock signals were seriously out of sync with one another, the fly was oblivious to the day-night cycle.”
The same research as reported in a University of Michigan 2014 press release is commented on as follows: “— Circadian clock like an orchestra with many conductors — Now, new findings from Shafer and U-M doctoral student Zepeng Yao challenge the prevailing wisdom about how our body clocks are organized, and suggest that interactions among neurons that govern circadian rhythms are more complex than originally thought. — Yao and Shafer looked at the circadian clock neuron network in fruit flies, which is functionally similar to that of mammals, but at only 150 clock neurons is much simpler. Previously, scientists thought that a master group of eight clock neurons acted as pacemaker for the remaining 142 clock neurons — think of a conductor leading an orchestra — thus imposing the rhythm for the fruit fly circadian clock. It is thought that the same principle applies to mammals. — Interactions among clock neurons determine the strength and speed of circadian rhythms, Yao says. So, when researchers genetically changed the clock speeds of only the group of eight master pacemakers they could examine how well the conductor alone governed the orchestra. They found that without the environmental cues, the orchestra didn’t follow the conductor as closely as previously thought. — Some of the fruit flies completely lost sense of time, and others simultaneously demonstrated two different sleep cycles, one following the group of eight neurons and the other following some other set of neurons. “The finding shows that instead of the entire orchestra following a single conductor, part of the orchestra is following a different conductor or not listening at all,” Shafer said. The findings suggest that instead of a group of master pacemaker neurons, the clock network consists of many independent clocks, each of which drives rhythms in activity. Shafer and Yao suspect that a similar organization will be found in mammals, as well. — “
Our old friend/enemy IGF-1 is implicated in sleep disruption, at least in the fruit fly. Further, inhibition of IGF-1 signaling and TOR may be an intervention that enhances sleep
Followers of the aging sciences know that IGF-1 signaling is essential in early animal development and yet plays a strong pro-aging and pro-disease role later in life. Indeed in many animals, inhibition of IGF-1 signaling in mature animals via TOR inhibition is a proven strategy for modest life extension That is how rapamycin extends life. Here is another angle: inhibition of IGF-1 signaling can lead to better sleep – at least in the fruit fly.
The 2014 publication Lowered Insulin Signalling Ameliorates Age-Related Sleep Fragmentation in Drosophila reports “Sleep fragmentation, particularly reduced and interrupted night sleep, impairs the quality of life of older people. Strikingly similar declines in sleep quality are seen during ageing in laboratory animals, including the fruit fly Drosophila. We investigated whether reduced activity of the nutrient- and stress-sensing insulin/insulin-like growth factor (IIS)/TOR signalling network, which ameliorates ageing in diverse organisms, could rescue the sleep fragmentation of ageing Drosophila. Lowered IIS/TOR network activity improved sleep quality, with increased night sleep and day activity and reduced sleep fragmentation. Reduced TOR activity, even when started for the first time late in life, improved sleep quality. The effects of reduced IIS/TOR network activity on day and night phenotypes were mediated through distinct mechanisms: Day activity was induced by adipokinetic hormone, dFOXO, and enhanced octopaminergic signalling. In contrast, night sleep duration and consolidation were dependent on reduced S6K and dopaminergic signalling. Our findings highlight the importance of different IIS/TOR components as potential therapeutic targets for pharmacological treatment of age-related sleep fragmentation in humans.” Further, “Sleep is essential for human health, but the quality of this fundamental physiological process declines with age and reduces quality of life. We therefore investigated the mechanisms by which ageing impairs sleep. We used the fruit fly Drosophila, whose sleep has many features in common with that of humans, including the age-related decline in quality. We examined the role of the insulin/IGF (IIS) and TOR signaling network, which has an evolutionarily conserved role in ageing. We found that flies with reduced IIS activity had improved sleep quality at night and higher activity levels by day. Importantly, day activity and night sleep were regulated through distinct mechanisms—day activity by the key IIS transcription factor dFOXO, adipokinetic hormone, and octopaminergic signalling—whereas night sleep was mediated through TOR and dopaminergic signalling. Surprisingly, acute inhibition of TOR, by rapamycin, even in old flies, improved sleep quality, suggesting that age-related sleep decline is reversible even after it has commenced. Given the high evolutionarily “conservation of IIS and TOR function, our results implicate potential therapeutic targets to improve sleep quality in humans.”
Natural expression of the melatonin hormone responds to the circadian cycle and exogenous hormone supplementation can be used to regulate the sleep cycle.
Melatonin is mentioned here because it has been much studied in relationship to circadian sleep rhythms (ref) and is widely used as a supplement. A Pubmed search on “melatonin circadian rhythm” today produces 5765 research article citations, more tomorrow. An entire blog entry could be devoted to its actions, and perhaps should be.
For the moment I mention only that here are many interesting sides to the melatonin story, for example, how circadian regulation or external taking of melatonin might affect dental hygiene and control dental biofilm and plaque formation and the formation of cavities. Relative to this, there is a 2014 review article: Melatonin in the oral cavity: physiological and pathological implications: “BACKGROUND AND OBJECTIVES: The purpose of this article was to summarize what is known about the function of melatonin in the oral cavity. MATERIAL AND METHODS: Databases were searched for the relevant published literature to 30 November, 2013. The following search items were used in various combinations: melatonin, gingiva, periodontium, inflammation, herpes, alveolar bone, periodontal ligament, dental implants, xerostomia, methacrylate, chlorhexidine, cancer. The literature uncovered is summarized herein. RESULTS: Salivary melatonin levels exhibit a circadian rhythm with highest values at night. Melatonin has both receptor-mediated and receptor-independent actions in cells of the oral cavity. Melatonin is released into the saliva by the acinar cells of the major salivary glands and via the gingival fluid. Functions of melatonin in the oral cavity are likely to relate primarily to its anti-inflammatory and antioxidant activities. These actions may suppress inflammation of the gingiva and periodontium, reduce alveolar bone loss, abrogate herpes lesions, enhance osteointegration of dental implants, limit oral cancer, and suppress disorders that have a free radical component. Sublingual melatonin tablets or oralmelatonin sprays and topica melatonin-containing gel, if used on a regular basis, may improve overall oral health and reduce mucosal lesions. CONCLUSION: Collectively, the results indicate that endogenously-produced and exogenously-appliedmelatonin are beneficial to the oral cavity.’
Another interesting side of the melatonin-circadian story relates to early-stage human development. The 2014 review article Melatonin and stable circadian rhythms optimize maternal, placental and fetal physiology reports: “BACKGROUND: Research within the last decade has shown melatonin to have previously-unsuspected beneficial actions on the peripheral reproductive organs. Likewise, numerous investigations have documented that stable circadian rhythms are also helpful in maintaining reproductive health. The relationship of melatonin and circadian rhythmicity to maternal and fetal health is summarized in this review. METHODS: Databases were searched for the related published English literature up to 15 May 2013. The search terms used in various combinations included melatonin, circadian rhythms, biological clock, suprachiasmatic nucleus, ovary, pregnancy, uterus, placenta, fetus, pre-eclampsia, intrauterine growth restriction, ischemia-reperfusion, chronodisruption, antioxidants, oxidative stress and free radicals. The results of the studies uncovered are summarized herein. RESULTS: Both melatonin and circadian rhythms impact reproduction, especially during pregnancy. Melatonin is a multifaceted molecule with direct free radical scavenging and indirect antioxidant activities. Melatonin is produced in both the ovary and in the placenta where it protects against molecular mutilation and cellular dysfunction arising from oxidative/nitrosative stress. The placenta, in particular, is often a site of excessive free radical generation due to less than optimal adhesion to the uterine wall, which leads to either persistent hypoxia or intermittent hypoxia and reoxygenation, processes that cause massive free radical generation and organ dysfunction. This may contribute to pre-eclampsia and other disorders which often complicate pregnancy. Melatonin has ameliorated free radical damage to the placenta and to the fetus in experiments using non-human mammals. Likewise, the maintenance of a regular maternal light/dark and sleep/wake cycle is important to stabilize circadianrhythms generated by the maternal central circadian pacemaker, the suprachiasmatic nuclei. Optimalcircadian rhythmicity in the mother is important since her circadian clock, either directly or indirectly via the melatonin rhythm, programs the developing master oscillator of the fetus. Experimental studies have shown that disturbed maternal circadian rhythms, referred to as chronodisruption, and perturbedmelatonin cycles have negative consequences for the maturing fetal oscillators, which may lead to psychological and behavioral problems in the newborn. To optimize regular circadian rhythms and prevent disturbances of the melatonin cycle during pregnancy, shift work and bright light exposure at night should be avoided, especially during the last trimester of pregnancy. Finally, melatonin synergizes with oxytocin to promote delivery of the fetus. Since blood melatonin levels are normally highest during the dark period, the propensity of childbirth to occur at night may relate to the high levels of melatonin at this time which work in concert with oxytocin to enhance the strength of uterine contractions. CONCLUSIONS: A number of conclusions naturally evolve from the data summarized in this review: (i)melatonin, of both pineal and placental origin, has essential functions in fetal maturation and placenta/uterine homeostasis; (ii) circadian clock genes, which are components of all cells including those in the peripheral reproductive organs, have important roles in reproductive and organismal (fetal and maternal) physiology; (iii) due to the potent antioxidant actions of melatonin, coupled with its virtual absence of toxicity, this indoleamine may have utility in the treatment of pre-eclampsia, intrauterine growth restriction, placental and fetal ischemia/reperfusion, etc. (iv) the propensity for parturition to occur at night may relate to the synergism between the nocturnal increase in melatonin and oxytocin.”
New molecular approaches are being discovered for resetting and regulating circadian clock functions.
For example, it appears that that the enzyme casein kinase 1epsilon (CK1epsilon) controls ease of entrainment, that is, how easily the body’s clockwork can be adjusted or reset by environmental cues such as light and temperature. The 2014 publication A Novel Mechanism Controlling Resetting Speed of the Circadian Clock to Environmental Stimuli relates to clock phase resetting and clock speeds. It reports: “Many aspects of mammalian physiology are driven through the coordinated action of internal circadian clocks. Clock speed (period) and phase (temporal alignment) are fundamental to an organism’s ability to synchronize with its environment. In humans, lifestyles that disturb these clocks, such as shift work, increase the incidence of diseases such as cancer and diabetes. Casein kinases 1δ and ε are closely related clock components implicated in period determination. However, CK1δ is so dominant in this regard that it remains unclear what function CK1ε normally serves. Here, we reveal that CK1ε dictates how rapidly the clock is reset by environmental stimuli. Genetic disruption of CK1ε in mice enhances phase resetting of behavioral rhythms to acute light pulses and shifts in light cycle. This impact of CK1ε targeting is recapitulated in isolated brain suprachiasmatic nucleus and peripheral (lung) clocks during NMDA- or temperature-induced phase shift in association with altered PERIOD (PER) protein dynamics. Importantly, accelerated re-entrainment of the circadian system in vivo and in vitro can be achieved in wild-type animals through pharmacological inhibition of CK1ε. These studies therefore reveal a role for CK1ε in stabilizing the circadian clock against phase shift and highlight it as a novel target for minimizing physiological disturbance in shift workers.”
A relatively new and only partially explored frontier of circadian-related research involves the roles of various species of RNAs – coding RNAs, micro RNAs, long non-coding RNAs, alternative-splice RNAs, lincRNAs, circular RNAs, etc. It appears both that there is both circadian control of expression of certain RNAs, and that certain RNAs can affect clock gene expression and circadian rhythms.
Initial research based on the fruit fly model suggests that there is significant circadian clock control of expression on non-coding RNAs, as well as control of alternative splicing and RNA editing.
The 2012 e- publication Deep sequencing the circadian and diurnal transcriptome of Drosophila brain reports: “Eukaryotic circadian clocks include transcriptional/translational feedback loops that drive 24-h rhythms of transcription. These transcriptional rhythms underlie oscillations of protein abundance, thereby mediating circadian rhythms of behavior, physiology, and metabolism. Numerous studies over the last decade have used microarrays to profile circadian transcriptional rhythms in various organisms and tissues. Here we use RNA sequencing (RNA-seq) to profile the circadian transcriptome of Drosophila melanogaster brain from wild-type and period-null clock-defective animals. We identify several hundred transcripts whose abundance oscillates with 24-h periods in either constant darkness or 12 h light/dark diurnal cycles, including several noncoding RNAs (ncRNAs) that were not identified in previous microarray studies. Of particular interest are U snoRNA host genes (Uhgs), a family of diurnal cycling noncoding RNAs that encode the precursors of more than 50 box-C/D small nucleolar RNAs, key regulators of ribosomal biogenesis. Transcriptional profiling at the level of individual exons reveals alternative splice isoforms for many genes whose relative abundances are regulated by either period or circadian time, although the effect of circadian time is muted in comparison to that of period. Interestingly, period loss of function significantly alters the frequency of RNA editing at several editing sites, suggesting an unexpected link between a key circadian gene and RNA editing. We also identify tens of thousands of novel splicing events beyond those previously annotated by the modENCODE Consortium, including several that affect key circadian genes. These studies demonstrate extensive circadian control of ncRNA expression, reveal the extent of clock control of alternative splicing and RNA editing, and provide a novel, genome-wide map of splicing in Drosophila brain.”
Micro RNAs play important roles in regulating circadian functions
The 2013 publication Micro-managing the circadian clock: The role of microRNAs in biological timekeeping reports: “Evolved under the selective pressures of a 24-h world, circadian timekeeping mechanisms are present in virtually all living organisms to coordinate daily rhythms in physiology and behavior. Until recently, the circadian clock was modeled as simple, interlocked transcription-translation feedback loops driving rhythms in gene expression of a handful of core clock genes. However, it has become evident that circadian clock regulation is immensely more complex than once thought and involves posttranscriptional, translational and posttranslational mechanisms. In particular, there has been a growing awareness of the vital role played by microRNAs (miRNAs) in regulating various aspects of circadian clock function. In this review, we will summarize our current knowledge of miRNA-dependent regulation of the circadian timing system in multiple organisms, including flies, mammals and higher plants. We will also discuss future perspectives for research on the role of miRNAs and noncoding RNAs in circadian regulation of health and disease.”
Another popular model besides the fruit fly for studying circadian rhythms with respect to gene expression is the zebra fish.
The 2014 publication The light-induced transcriptome of the zebrafish pineal gland reveals complex regulation of the circadian clockwork by light reports: “Light constitutes a primary signal whereby endogenous circadian clocks are synchronized (‘entrained’) with the day/night cycle. The molecular mechanisms underlying this vital process are known to require gene activation, yet are incompletely understood. Here, the light-induced transcriptome in the zebrafish central clock organ, the pineal gland, was characterized by messenger RNA (mRNA) sequencing (mRNA-seq) and microarray analyses, resulting in the identification of multiple light-induced mRNAs. Interestingly, a considerable portion of the molecular clock (14 genes) is light-induced in the pineal gland. Four of these genes, encoding the transcription factors dec1, reverbb1, e4bp4-5 and e4bp4-6, differentially affected clock- and light-regulated promoter activation, suggesting that light-input is conveyed to the core clock machinery via diverse mechanisms. Moreover, we show that dec1, as well as the core clock gene per2, is essential for light-entrainment of rhythmic locomotor activity in zebrafish larvae. Additionally, we used microRNA (miRNA) sequencing (miR-seq) and identified pineal-enhanced and light-induced miRNAs. One such miRNA, miR-183, is shown to downregulate e4bp4-6 mRNA through a 3’UTR target site, and importantly, to regulate the rhythmic mRNA levels of aanat2, the key enzyme in melatonin synthesis. Together, this genome-wide approach and functional characterization of light-induced factors indicate a multi-level regulation of the circadian clockwork by light.”
My comment: There seems to be agreement that the complex regulatory interactions observed in the fruit fly and the zebrafish situations are very likely to be evolutionarily conserved in mammals and are also likely to be significantly more complex in us. But so far as far as I have looked, these are yet to be thoroughly investigated and understood as far as humans are concerned. We are only at an early stage of understanding how circadian rhythms affect gene expression in humans, how RNA species affect circadian patterns. And how circadian patterns impact on expression of RNA species..
This paper published last month summarizes the situation as far as long ncRNAs are concerned: Regulation of metabolism by long, non-coding RNAs. “Our understanding of genomic regulation was revolutionized by the discovery that the genome is pervasively transcribed, giving rise to thousands of mostly uncharacterized non-coding ribonucleic acids (ncRNAs). Long, ncRNAs (lncRNAs) have thus emerged as a novel class of functional RNAs that impinge on gene regulation by a broad spectrum of mechanisms such as the recruitment of epigenetic modifier proteins, control of mRNA decay and DNA sequestration of transcription factors. We review those lncRNAs that are implicated in differentiation and homeostasis of metabolic tissues and present novel concepts on how lncRNAs might act on energy and glucose homeostasis. Finally, the control of circadian rhythm by lncRNAs is an emerging principles of lncRNA-mediated gene regulation.”
This blog entry has been a sampler of a variety of recent research relating to circadian rhythms. Whole new blog entries could be devoted to some of the subtopics such as rhythms related to gene expression, ncRNAs, melatonin and relationships among clocks. And there is the whole field of plant circadian rhythms. I expect my colleagues and I will report further on some of these topics in the near future.


New findings on circadian rhythms and their impact appear almost daily. Here is one from a paper published just yesterday.
A Novel Protein, CHRONO, Functions as a Core Component of the Mammalian Circadian Clock
at http://www.plosbiology.org/article/info%3Adoi%2F10.1371%2Fjournal.pbio.1001839
listing the original info:
”Abstract: Circadian rhythms are controlled by a system of negative and positive genetic feedback loops composed of clock genes. Although many genes have been implicated in these feedback loops, it is unclear whether our current list of clock genes is exhaustive. We have recently identified Chrono as a robustly cycling transcript through genome-wide profiling of BMAL1 binding on the E-box. Here, we explore the role of Chrono in cellular timekeeping. Remarkably, endogenous CHRONO occupancy around E-boxes shows a circadian oscillation antiphasic to BMAL1. Overexpression of Chrono leads to suppression of BMAL1–CLOCK activity in a histone deacetylase (HDAC) –dependent manner. In vivo loss-of-function studies of Chrono includingAvp neuron-specific knockout (KO) mice display a longer circadian period of locomotor activity.Chrono KO also alters the expression of core clock genes and impairs the response of the circadian clock to stress. CHRONO forms a complex with the glucocorticoid receptor and mediates glucocorticoid response. Our comprehensive study spotlights a previously unrecognized clock component of an unsuspected negative circadian feedback loop that is independent of another negative regulator, Cry2, and that integrates behavioral stress and epigenetic control for efficient metabolic integration of the clock.
Author Summary:
The circadian clock has a fundamental role in regulating biological temporal rhythms in organisms, and it is tightly controlled by a molecular circuit consisting of positive and negative regulatory feedback loops. Although many of the clock genes comprising this circuit have been identified, there are still some critical components missing. Here, we characterize a circadian gene renamed Chrono (Gm129) and show that it functions as a transcriptional repressor of the negative feedback loop in the mammalian clock. Chrono binds to the regulatory region of clock genes and its occupancy oscillates in a circadian manner. Chrono knockout and Avp-neuron-specific knockout mice display longer circadian periods and altered expression of core clock genes. We show that Chrono-mediated repression involves the suppression of BMAL1–CLOCK activity via an epigenetic mechanism and that it regulates metabolic pathways triggered by behavioral stress. Our study suggests that Chrono functions as a clock repressor and reveals the molecular mechanisms underlying its function.”
Many experimental data support the hypothesis that the developmental pathways of cells and complex organisms are the results of conserved biological clocks based on metabolic hypercycles organized in fractal networks …
http://biomedphys.sgu.ru/Files/LIB/Springer/%20Fractals%20in%20Biology%20and%20Medicine/25.pdf
Pingback: FUNNY THINGS ARE HAPPENING TO ME ON THE WAY TO 100 - AGINGSCIENCES™ - Anti-Aging Firewalls™AGINGSCIENCES™ – Anti-Aging Firewalls™