Microtubules, the intra-cellular transport system, health and longevity
By Vince Giuliano
Microtubules play important and ever-changing structural roles in cells, play key roles in embryo development and cell division, are the basis for the intra-cellular molecular transport system, and most likely play important roles in gene activation and epigenetic processes. I focus here on some essential things known about microtubules, and the cargo-carrying motor proteins that run along them. And I also review some facts about nuclear membranes within cells that must be crossed for intra-cellular signaling and transport. I discuss why microtubules are extremely important and review some of their known roles in aging and age-related disease processes. Some of the basic research described here dates back into the 1990s. And some important findings related to histone epigenetics, mitochondria, and the transport of P53 are absolutely current and could be highly relevant to cancer, to neurological diseases, and to aging.
This is a long and complex blog entry. As usual, I highlight the main “take home” points as I see them.
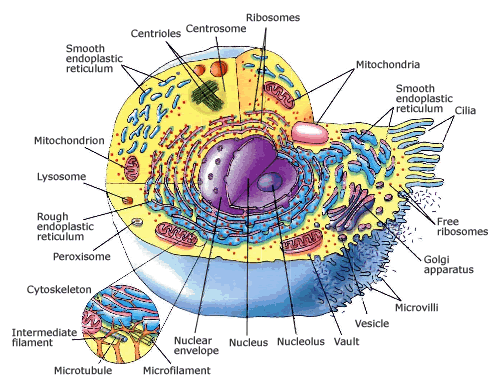
In very rough terms, microtubules are like support beams fanning out from the center of a cell through the cytoplasm to the periphery. A little like tinkertoy struts, these beams are arranged very differently at different times in the cell division cycle and can be moved and re-arranged. However, unlike tinkertoy struts they can be extended or shortened, bent, dissolved or replaced. These beams also serve as rail lines for moving substances through the cytoplasm. Motor proteins are like locomotives that carry cargo back and forth, cargo ranging from cell structure building materials, to signaling proteins, to entire mitochondria. Building a cell in the process of mitosis as well as keeping a cell in good operating shape requires a lot of hauling. And the structural and hauling systems of microtubules are constantly being torn down and rebuilt. Microtubules play a crucial role in the internal organization of cells and placement of key components. They act as scaffolds for many internal organelles including the endoplasmic reticulum, ribosomes and the golgi apparatus. The tinkertoy structures created by mitochondria are all temporary, created to meet the needs of a cell at a moment. They typically just last for a number of minutes. Yet, what these microtubule systems do may have profound effects on events in the nucleus and miotochondria in cells and impact on gene expression.
I have touched on microtubules in two previous blog entries One of these is Quantum biology, where I pointed out that there is a group of researchers who think microtubules also function as quantum computers. I will not go further into that interesting hypothesis in this blog entry. In another entry, Buckyballs, health and longevity – state of knowledge, I speculated that important biological impacts of C60 fullerenes could be mediated by the binding of C60 to microtubules.
1, BASIC FUNCTIONING AND BIOLOGY OF MICROTUBULES
For background, I first dive a bit into some key elements of microtubule biology. My impression is that much of the basic research on microtubules was done in the 1990s and early 2000s, though they were first identified back in 1903. So I will start in this Section with some of the basic and long-known properties of microtubules. The cell biology related to microtubules is very complex involving numerous specialized proteins and a highly technical literature. I cover only some of the main points here.
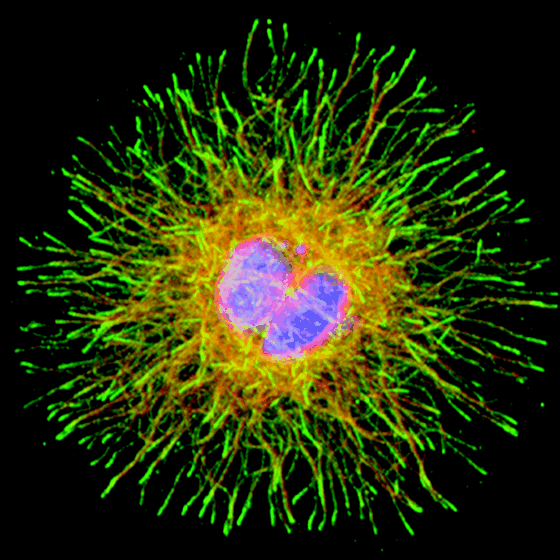
This description which highlights the beauty and wonderment of microtubules and what they do is from the 2000 publication The Molecular Perspective: Microtubules and the Taxanes: “Our cells are shaped and supported by a cytoskeleton of interlocking protein filaments. A beautiful star of microtubules, the largest of these filaments, radiates outward from the center of the cell to the cell surface. This “aster” of microtubules is the railway system of the cell. Many types of cargo are carried along these rails. The endoplasmic reticulum is pulled by molecular motor proteins along microtubules, spreading it evenly throughout the cell. Vesicles are delivered to their destinations along microtubules. And, when cells divide, the most valuable cargo of the cell is carried by microtubules. Paired copies of each of the chromosomes are attached to the ends of a doubled microtubule aster and carefully separated into the two daughter cells. The cytoskeleton, in contrast to our articulated skeleton of bones, is a dynamic structure. It is continually constructed and demolished according to the shifting needs of the cell. A typical microtubule lasts for only 10 minutes before it is disassembled and the parts used to build a new one. Microtubules are nucleated in the “microtubule-organizing center,” at the center of the cell, and then extended one piece at a time into the cytoplasm. Growth proceeds in fits and starts in a process known as “dynamic instability.” Tubules grow slowly and steadily, but are punctuated by periods of rapid disassembly, when large regions peel away from the ends. A small portion of the microtubule may break up, or the fragmentation may extend all the way back to the start, completely destroying the tubule. In special cases, such as the microtubules that support the long axons and dendrites of nerve cells, auxiliary proteins stabilize the microtubule for longer periods of time. But the dynamic structure of microtubules is essential for their essential for their everyday function in transport and cell division.”
The cytoskeleton
Microtubules are one of three major kinds of structures found in the cells cytoskeleton, the internal structural system of a cell. The other two kinds of structures are microfilaments made out of actin and intermediate filaments. These elements often cooperate with each other to achieve various structural tasks. Intermediate filaments “– averaging 10 nanometers in diameter, are more stable (strongly bound) than actin filaments, and heterogeneous constituents of the cytoskeleton. Like actin filaments, they function in the maintenance of cell-shape by bearing tension (microtubules, by contrast, resist compression. It may be useful to think of micro- and intermediate filaments as cables, and of microtubules as cellular support beams). Intermediate filaments organize the internal tridimensional structure of the cell, anchoring organelles and serving as structural components of the nuclear lamina and sarcomeres. They also participate in some cell-cell and cell-matrix junctions(ref).”
What are microtubules?
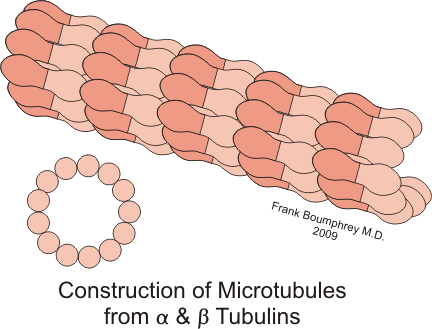
From the 2009 document On and around microtubules: an overview: “Microtubules are hollow tubes some 25 nm in diameter participating in the eukaryotic cytoskeleton. They are built from alphabeta-tubulin heterodimers that associate to form protofilaments running lengthwise along the microtubule wall with the beta-tubulin subunit facing the microtubule plus end conferring a structural polarity. The alpha- and beta-tubulins are highly conserved. A third member of the tubulin family, gamma-tubulin, plays a role in microtubule nucleation and assembly. Other members of the tubulin family appear to be involved in microtubule nucleation. Microtubule assembly is accompanied by hydrolysis of GTP associated with beta-tubulin so that microtubules consist principally of ‘GDP-tubulin’ stabilized at the plus end by a short ‘cap’. An important property of microtubules is dynamic instability characterized by growth randomly interrupted by pauses and shrinkage. Many proteins interact with microtubules within the cell and are involved in essential functions such as microtubule growth, stabilization, destabilization, and interactions with chromosomes during cell division. The motor proteins kinesin and dynein use microtubules as pathways for transport and are also involved in cell division. Crystallography and electron microscopy are providing a structural basis for understanding the interactions of microtubules with antimitotic drugs, with motor proteins and with plus end tracking proteins.”
Besides generating structures and serving as molecular rail lines, microtubules can generate forces, for example ones that deform the nuclear envelope.
From the 2007 document Microtubules: an overview: “Microtubules are found in all eukaryotes and are built from alphabeta-tubulin heterodimers. The alpha-tubulins and beta-tubulins are among the most highly conserved eukaryotic proteins. Other members of the tubulin family have come to light recently and, like gamma-tubulin, appear to play roles in microtubule nucleation and assembly. Microtubule assembly is accompanied by hydrolysis of GTP associated with beta-tubulin so that microtubules consist principally of “GDP-tubulin” stabilized by a short “GTP cap.” Microtubules are polar, cylindrical structures some 25 nm in diameter. Protofilaments made from tubulin heterodimers run lengthwise along the microtubule wall with the beta-tubulin subunit at the microtubule plus end. The crystallographic structures of tubulins are essential to understand in detail microtubule architecture and interactions with stabilizing and destabilizing drugs and proteins.”
From Wikipedia: “Microtubules are a component of the cytoskeleton. These cylindrical polymers of tubulin can grow as long as 25 micrometers and are highly dynamic. The outer diameter of microtubule is about 25 nm. Microtubules are important for maintaining cell structure, providing platforms for intracellular transport, forming the mitotic spindle, as well as other cellular processes.[1] There are many proteins that bind to microtubules, including motor proteins such as kinesin and dynein, severing proteins like katanin, and other proteins important for regulating microtubule dynamics.[2][3]”—“ Microtubules have a distinct polarity which is important for their biological function. Tubulin polymerizes end to end with the α subunit of one tubulin dimer contacting the β subunit of the next. Therefore, in a protofilament, one end will have the α subunit exposed while the other end will have the β subunit exposed. These ends are designated the (−) and (+) ends, respectively. The protofilaments bundle parallel to one another, so, in a microtubule, there is one end, the (+) end, with only β subunits exposed, while the other end, the (−) end, has only α subunits exposed. Elongation of microtubules typically only occurs from the (+) end.” –“Microtubules are part of a structural network (the cytoskeleton) within the cell’s cytoplasm. The primary role of the microtubule cytoskeleton is mechanical. However, in addition to structural support, microtubules also take part in many other processes. A microtubule is capable of growing and shrinking in order to generate force, and there are also motor proteins that allow organelles and other cellular factors to be carried along a microtubule. This combination of roles makes microtubules important for organizing cell layout.” – “Microtubule nucleation: Microtubules are typically nucleated and organized by dedicated organelles called microtubule-organizing centers (MTOCs). MTOCs associated with the base of a eukaryotic cillium or flagellum are typically termed basal bodies, otherwise they are called centrioles. In many cell types microtubules are primarily nucleated at MTOCs however there are also many exceptions to this rule.”
Microtubules are polarized and are semiconductors. They generally orient themselves so that their positively charged ends are oriented towards the cell’s periphery, the negatively charged ends are oriented towards the cell’s center.
For transporting cargo along microtubules, there are two classes of motor proteins kinesins which move cargo by “walking” them towards the positive ends (away from the center of the cell towards the periphery) and dyneins which move cargo by walking it towards the negative ends (towards the cell’s center). A fascinating video which illustrates how kinesins walk and carry large cargos can be seen here.
“A kinesin is a protein belonging to a class of motor proteins found in eukaryotic cells. Kinesins move along microtubule filaments, and are powered by the hydrolysis of ATP (thus kinesins are ATPases). The active movement of kinesins supports several cellular functions including mitosis, meiosis and transport of cellular cargo, such as in axonal transport. Most kinesins walk towards the plus end of a microtubule, which, in most cells, entails transporting cargo from the centre of the cell towards the periphery. This form of transport is known as anterograde transport.” — “Kinesins were discovered as microtubule (MT)-based anterograde intracellular transport motors.[1] — Molecular genetic and genomic approaches have led to the recognition that the kinesins form a diverse superfamily of motors that are responsible for multiple intracellular motility events in eukaryotic cells.[5][6][7][8] For example, the genomes of mammals encode more than 40 kinesin proteins,[9] organized into at least 14 families named kinesin-1 through kinesin-14.[10] (ref).”
Regarding dyneins “Cytoplasmic dynein probably helps to position the Golgi complex and other organelles in the cell.[1] It also helps transport cargo needed for cell function such as vesicles made by the endoplasmic reticulum, endosomes, and lysosomes (Karp, 2005). Dynein is involved in the movement of chromosomes and positioning the mitotic spindles for cell division.[2] Dynein carries organelles and microtubule fragments along the axons of neurons in a process called axoplasmic transport.[1] (ref).”
Microtubules are extremely versatile and play different roles during the various phases of a cell cycle and, under control of and choreographed by a number of microtubule-related proteins, can be assembled into various structures, broken down and re-assembled.
Because of their versatility and because they spool off of central hubs, microtubules are like improved spokes in tinkertoys, spokes which can be spooled off in any controlled directions, spokes which can bend and be hardened and softened, and spokes whose lengths can be dynamically regulated.
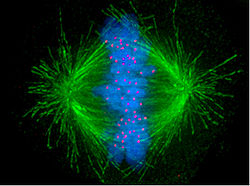
Microtubules play essential dynamic roles during cell division including construction of spindle poles and centrosomes.
“In cell biology, spindle apparatus refers to the subcellular structure that segregates chromosomes between daughter cells during cell division. It is also referred to as the mitotic spindle during mitosis or the meiotic spindle during meiosis. — While the spindle apparatus is composed of hundreds upon hundreds of proteins,[1] the fundamental machinery are the spindle microtubules. Attachment of microtubules to chromosomes is mediated by kinetochores, which actively monitor spindle formation and prevent premature anaphase onset. Microtubule polymerization and depolymerization dynamics drive chromosome congression. Depolymerization of microtubules generates tension at kinetochores;[2] bipolar attachment of sister kinetochores to microtubules emanating from opposite cell poles couples opposing tension forces, aligning chromosomes at the cell equator and poising them for segregation to daughter cells. Once every chromosome is bi-oriented, anaphase commences and cohesin, which couples sister chromatids, is severed, permitting the transit of the sister chromatids to opposite poles(ref).”
Because microtubules play such a central role in cell division, they are targeted by some approaches to cancer chemotherapy.
Microtubules play key roles in construction work in cells in the process of mitosis.
“The process of mitosis is facilitated by a subgroup of microtubules known as astral microtubules, defined as a microtubule originating from the centrosome that does not connect to a kinetochore. Astral microtubules develop in the actin skeleton and interact with the cell cortex to aid in spindle orientation. They are organized into radial arrays around the centrosomes. The turn-over rate of this population of microtubules is higher than that of any other population. Astral microtubules function in concert with specialized dynein motors, which are oriented with the light chain portion attached to the cell membrane and the dynamic portion attached to the microtubule. This allows for dynein contraction to pull the centrosome toward the cell membrane, thus assisting in cytokinesis(ref).”
Image source “Micrograph showing condensed chromosomes in blue, kinetochores in pink, and microtubules in green during metaphase of mitosis” Better than words, this image illustrates one of the key physical tasks accomplished by microtubules.
“In cell biology, the centrosome is an organelle that serves as the main microtubule organizing center (MTOC) of the animal cell as well as a regulator of cell-cycle progression. It was discovered by Edouard Van Beneden in 1883 [1] and was described and named in 1888 by Theodor Boveri.[2] In the theory of evolution the centrosome is thought to have evolved only in the metazoan lineage of eukaryotic cells.[3] Fungi and plants use other MTOC structures to organize their microtubules.[4][5] Although the centrosome has a key role in efficient mitosis in animal cells, it is not essential.[6][7][8] — Centrosomes are composed of two orthogonally arranged centrioles surrounded by an amorphous mass of protein termed the pericentriolar material (PCM). The PCM contains proteins responsible for microtubule nucleation and anchoring[9] including γ-tubulin, pericentrin and ninein. In general, each centriole of the centrosome is based on a nine triplet microtubule assembled in a cartwheel structure, and contains centrin, cenexin and tektin.[10] (ref)”
Microtubules are highly dynamic and can grow and shrink.
“Dynamic instability refers to the coexistence of assembly and disassembly at the (+) end of a microtubule. The microtubule can switch between the growing and shrinking phases dynamically at this region.[4] — Since tubulin adds onto the end of the microtubule only in the GTP-bound state, there is a cap of GTP-bound tubulin at the tip of the microtubule, protecting it from disassembly. When hydrolysis catches up to the tip of the microtubule, it begins a rapid depolymerization and shrinkage. This switch from growth to shrinking is called a catastrophe. GTP-bound tubulin can begin adding to the tip of the microtubule again, providing a new cap and protecting the microtubule from shrinking. This is referred to as “rescue”[5](ref). ”
Microtubules are used to transport proteins in order to build microtubule spindles and centrosomes. It is like using existing rail lines to transport materials to build extended or new rail lines.
The 2000 publication Construction of centrosomes and spindle poles by molecular motor-driven assembly of protein particles described the situation: “Centrosomes and other microtubule organizing centers are the largest non-membranous organelles in most cells. This morphologically diverse class of organelles shares a common ability to nucleate and organize microtubules in interphase and participates in the formation of mitotic spindles during cell division. This review summarizes recent evidence suggesting that assembly of centrosomes and mitotic spindle poles require transport of large protein particles along microtubules by the molecular motor cytoplasmic dynein.”
Although a great many proteins are involved in the dynamic organization and re-organization of microtubule structures in cells, dynein and dynactin seem to play particularly important roles.
The 2oo6 publication [Dynein and dynactin as organizers of the system of cell microtubules] reports: “A review of the role of the microtubule motor dynein and its cofactor dynactin in the formation of a radial system of microtubules in the interphase cells and of mitotic spindle. Deciphering of the structure, functions, and regulation of activity of dynein and dynactin promoted the understanding of mechanisms of cell and tissue morphogenesis, since it turned out that these cells help the cell in finding its center and organize microtubule-determined anisotropy of intracellular space. The structure of dynein and dynactin molecules has been considered, as well as possible pathways of regulation of the dynein activity and the role of dynein in transport of cell components along the microtubules. Attention has also been paid to the functions of dynein and dynactin not related directly to transport: their involvement in the formation of an interphase radial system of microtubules. This system can be formed by self-organization of microtubules and dynein-containing organelles or via organization of microtubules by the centrosome, whose functioning requires dynein. In addition, dynein and dynactin are responsible for cell polarization during its movement, as well as for the position of nucleus, centrosomes, and mitotic spindle in the cell.”
The 2007 publication Microtubule binding by dynactin is required for microtubule organization but not cargo transport is another relating to dynactin and microtubules. “Dynactin links cytoplasmic dynein and other motors to cargo and is involved in organizing radial microtubule arrays. The largest subunit of dynactin, p150(glued), binds the dynein intermediate chain and has an N-terminal microtubule-binding domain. To examine the role of microtubule binding by p150(glued), we replaced the wild-type p150(glued) in Drosophila melanogaster S2 cells with mutant DeltaN-p150 lacking residues 1-200, which is unable to bind microtubules. Cells treated with cytochalasin D were used for analysis of cargo movement along microtubules. Strikingly, although the movement of both membranous organelles and messenger ribonucleoprotein complexes by dynein and kinesin-1 requires dynactin, the substitution of full-length p150(glued) with DeltaN-p150(glued) has no effect on the rate, processivity, or step size of transport. However, truncation of the microtubule-binding domain of p150(glued) has a dramatic effect on cell division, resulting in the generation of multipolar spindles and free microtubule-organizing centers. Thus, dynactin binding to microtubules is required for organizing spindle microtubule arrays but not cargo motility in vivo.”
Neurons utilize microtubule transport mechanisms for their central signaling functions, transporting molecules over relatively very large distances. The proper functioning of microtubulin networks is particularly important in neurons.
A nerve cell axon is packed full of neural filaments and microtubules. Although electrical signals go outwards from the cell body to the synapse, molecular transport in the axon along microtubules is intense and two-way. The cell body makes proteins used at the extreme synapse ends of axons and materials are recycled back to the cell body. On a cell scale, immense distances may be involved. An axon can be more than three feet long.
“Axoplasmic transport, also called axonal transport, is a cellular process responsible for movement of mitochondria, lipids, synaptic vesicles, proteins, and other cell parts (i.e. organelles) to and from a neuron’s cell body, through the cytoplasm of its axon (the axoplasm). Axons, which can be 1,000 or 10,000 times the length of the cell body, were originally thought to contain no ribosomes or means of producing proteins, and so were thought to rely on axoplasmic transport for all their protein needs.[1][2] However, more recently translation of mRNA has been demonstrated in axons.[3][4] Axonal transport is also responsible for moving molecules destined for degradation from the axon back to the cell body, where they are broken down by lysosomes.[5] The vast majority of axonal proteins are synthesized in the neuronal cell body and transported along axons. Axonal transport occurs throughout the life of a neuron and is essential to its growth and survival.” Microtubules (made of tubulin) run along the length of the axon and provide the main cytoskeletal “tracks” for transportation. The motor proteins kinesin and dynein are mechanochemical enzymes that move cargoes in the anterograde (towards the axon tip) and retrograde (towards the cell body) directions, respectively. Motor proteins bind and transport several different cargoes including organelles such as mitochondria, cytoskeletal polymers, and vesicles containing neurotransmitters[1] (ref).”
Microtubules are key components of cilia and play important roles in processes for which ciliary movement is essential, such as mammalian fertilization.
Microtubules are key elements of motile structures such as flagella. If you are interested in this aspect of microtubules, you can see the publications in this list and those in this list.
2. BEYOND BASIC BIOLOGY TO MORE CURRENT AND INTERESTING FINDINGS
There are clues suggesting that stabilizing the mechanical properties of microtublues could be very important for their functioning, the functioning of other cell systems, and for health in general.
For example, it appears that some cancer chempotherepeautic agents serve to stabilize microtubules. A November 2012 publication Mechanical and functional properties of epothilone-stabilized microtubules reports: “Using a suite of biophysical tools, we assess the mechanical, structural, and functional properties of microtubules stabilized by the chemotherapeutic compounds epothilone-A, epothilone-B, and taxolin vitro. We demonstrate that microtubules stabilized by epothilone-A or epothilone-B are competent to bind tau proteins, and support kinesin translocation. Kinesin speed is sensitive not only to the type of small molecule stabilizer used, but also to the presence of the essential microtubule-associated protein tau. Epothilone-stabilized microtubules are substantially less stiff than taxol-stabilized microtubules. The addition of tau proteins to microtubules stabilized by either epothilone compound or taxol further reduces stiffness. Taken together, these results suggest that small molecule stabilizers do not simply stabilize a ‘native’ microtubule structure, but rather they modulate the structure, function, and mechanics of the microtubules they bind. This may have important consequences to the therapeutic use of these agents in cancer chemotherapies.”
A number of other research publications have been exploring the roles of microtubules in cancer processes and possible microtubule-based anti-cancer interventions. These include:
(2011) Mechanism of microtubule-facilitated “fast track” nuclear import.
Acetylases and deacetylases known best for their impacts on histones can also acetylate/deacetylate microtubule protein. The alpha-tubulin in microtubules can be acetylated or deacetylated, with important consequences for microtubule function and stability. Specifically, HDAC6, known for its histone deacetylase properties, is also a tubulin deacetylase. So also is Sir2. Therefore, along with the epigenetic effects of histone acetylation/deacetylation, there may be additional important biological impacts associated with microtubule acetylation/deacetylation.
The 2002 publication HDAC6 is a microtubule-associated deacetylase reports: “Reversible acetylation of alpha-tubulin has been implicated in regulating microtubule stability and function. The distribution of acetylated alpha-tubulin is tightly controlled and stereotypic. Acetylated alpha-tubulin is most abundant in stable microtubules but is absent from dynamic cellular structures such as neuronal growth cones and the leading edges of fibroblasts. However, the enzymes responsible for regulating tubulin acetylation and deacetylation are not known. Here we report that a member of the histone deacetylase family, HDAC6, functions as a tubulin deacetylase. HDAC6 is localized exclusively in the cytoplasm, where it associates with microtubules and localizes with the microtubule motor complex containing p150(glued) (ref. 3). In vivo, the overexpression of HDAC6 leads to a global deacetylation of alpha-tubulin, whereas a decrease in HDAC6 increases alpha-tubulin acetylation. In vitro, purified HDAC6 potently deacetylates alpha-tubulin in assembled microtubules. Furthermore, overexpression of HDAC6 promotes chemotactic cell movement, supporting the idea that HDAC6-mediated deacetylation regulates microtubule-dependent cell motility. Our results show that HDAC6 is the tubulin deacetylase, and provide evidence that reversible acetylation regulates important biological processes beyond histone metabolism and gene transcription.”
Since this publication in 2002 there have been several other studies related to microtubule acetylation and stability. The situation rapidly gets complicated and interesting. For example formin proteins are known to play active roles in shaping microtubule spindles. See The formins: active scaffolds that remodel the cytoskeleton. Formins seem also to play important roles in neuron development(ref). Formins induce microtubule acetylation and therefore enhance their stability. The October 2012 publication The ability to induce microtubule acetylation is a general feature of formin proteins makes this point: “Cytoplasmic microtubules exist as distinct dynamic and stable populations within the cell. Stable microtubules direct and maintain cell polarity and i t is thought that their stabilization is dependent on coordinative organization between the microtubule network and the actin cytoskeleton. A growing body of work suggests that some members of the formin family of actin remodeling proteins also regulate microtubule organization and stability. For example, we showed previously that expression of the novel formin INF1 is sufficient to induce microtubule stabilization and tubulin acetylation, but not tubulin detyrosination. An important issue with respect to the relationship between formins and microtubules is the determination of which formin domains mediate microtubule stabilization. INF1 has a distinct microtubule-binding domain at its C-terminus and the endogenous INF1 protein is associated with the microtubule network. Surprisingly, the INF1 microtubule-binding domain is not essential for INF1-induced microtubule acetylation. We show here that expression of the isolated FH1 + FH2 functional unit of INF1 is sufficient to induce microtubule acetylation independent of the INF1 microtubule-binding domain. It is not yet clear whether or not microtubule stabilization is a general property of all mammalian formins; therefore we expressed constitutively active derivatives of thirteen of the fifteen mammalian formin proteins in HeLa and NIH3T3 cells and measured their effects on stress fiber formation, MT organization and MT acetylation. We found that expression of the FH1 + FH2 unit of the majority of mammalian formins is sufficient to induce microtubule acetylation. Our results suggest that the regulation of microtubule acetylation is likely a general formin activity and that the FH2 should be thought of as a dual-function domain capable of regulating both actin and microtubule networks.”
Decreases in microtubule acetylation levels are associated with Neurological disorders.
The 2011 publication Microtubule dynamics in the peripheral nervous system: A matter of balance reports: “The special architecture of neurons in the peripheral nervous system, with axons extending for long distances, represents a major challenge for the intracellular transport system. Two recent studies show that mutations in the small heat shock protein HSPB1, which cause an axonal type of Charcot-Marie-Tooth (CMT) neuropathy, affect microtubule dynamics and impede axonal transport. Intriguingly, while at presymptomatic age the neurons in the mutant HSPB1 mouse show a hyperstable microtubule network, at postsymptomatic age, the microtubule network completely lost its stability as reflected by a marked decrease in tubulin acetylation levels. We here propose a model explaining the role of microtubule stabilization and tubulin acetylation in the pathogenesis of HSPB1 mutations.”Another publication relating tubulin acetylation to the functionality of cortical neurons is the September 2012 report MEC-17 deficiency leads to reduced α-tubulin acetylation and impaired migration of cortical neurons. “Neuronal migration is a fundamental process during the development of the cerebral cortex and is regulated by cytoskeletal components. Microtubule dynamics can be modulated by posttranslational modifications to tubulin subunits. Acetylation of α-tubulin at lysine 40 is important in regulating microtubule properties, and this process is controlled by acetyltransferase and deacetylase. MEC-17 is a newly discovered α-tubulin acetyltransferase that has been found to play a major role in the acetylation of α-tubulin in different species in vivo. However, the physiological function of MEC-17 during neural development is largely unknown. Here, we report that MEC-17 is critical for the migration of cortical neurons in the rat. MEC-17 was strongly expressed in the cerebral cortex during development. MEC-17 deficiency caused migratory defects in the cortical projection neurons and interneurons, and perturbed the transition of projection neurons from the multipolar stage to the unipolar/bipolar stage in the intermediate zone of the cortex. Furthermore, knockdown of α-tubulin deacetylase HDAC6 or overexpression of tubulin(K40Q) to mimic acetylated α-tubulin could reduce the migratory and morphological defects caused by MEC-17 deficiency in cortical projection neurons. Thus, MEC-17, which regulates the acetylation of α-tubulin, appears to control the migration and morphological transition of cortical neurons. This finding reveals the importance of MEC-17 and α-tubulin acetylation in cortical development.”
One kinds of cargo carried into or away from the nucleus of cells along microtubules are viruses. The direction in which viruses are carried and the speed of transport is determined by dynactin or its inhibition. At stake is whether a cell is infected by a virus or not.
The 2011 publication The dynactin complex enhances the speed of microtubule-dependent motions of adenovirus both towards and away from the nucleus reports: “Unlike transport vesicles or organelles, human adenovirus (HAdV) directly binds to the microtubule minus end-directed motor dynein for transport to the nucleus. The dynein cofactor dynactin enhances nuclear transport of HAdV and boosts infection. To determine if dynactin has a specific role in cytoplasmic trafficking of incoming HAdV on microtubules, we used live cell spinning disc confocal microscopy at 25 Hz acquisition frequency and automated tracking of single virus particles at 20-50 nm spatial resolution. Computational dissection by machine-learning algorithms extracted specific motion patterns of viral trajectories. We found that unperturbed cells supported two kinds of microtubule-dependent motions, directed motions (DM) and fast drifts (FD). DM had speeds of 0.2 to 2 μm/s and run lengths of 0.4 up to 7 μm, while FD were slower and less extensive at 0.02 to 0.4 μm/s and 0.05 to 2.5 μm. Dynactin interference by overexpression of p50/dynamitin or a coiled-coil domain of p150/Glued reduced the speeds and amounts of both center- and periphery-directed DM but not FD, and inhibited infection. These results indicate that dynactin enhances adenovirus infection by increasing the speed and efficiency of dynein-mediated virus motion to the nucleus, and, surprisingly, also supports a hereto unknown motor activity for virus transport to the cell periphery.”
3. THE NUCLEAR ENVELOPE AND MICROTUBULES
Microtubules interact in various ways with the nuclear envelope, so it is important to grasp what this envelope is and the key roles it plays.
The 2005 publication Pushing the envelope: structure, function, and dynamics of the nuclear periphery relates: “The nuclear envelope (NE) is a highly specialized membrane that delineates the eukaryotic cell nucleus. It is composed of the inner and outer nuclear membranes, nuclear pore complexes (NPCs) and, in metazoa, the lamina. The NE not only regulates the trafficking of macromolecules between nucleoplasm and cytosol but also provides anchoring sites for chromatin and the cytoskeleton. Through these interactions, the NE helps position the nucleus within the cell and chromosomes within the nucleus, thereby regulating the expression of certain genes. The NE is not static, rather it is continuously remodeled during cell division. The most dramatic example of NE reorganization occurs during mitosis in metazoa when the NE undergoes a complete cycle of disassembly and reformation. Despite the importance of the NE for eukaryotic cell life, relatively little is known about its biogenesis or many of its functions. We thus are far from understanding the molecular etiology of a diverse group of NE-associated diseases.”
The 2008 publication Till disassembly do us part: a happy marriage of nuclear envelope and chromatin reports: “A characteristic feature of eukaryotic cells is the presence of nuclear envelope (NE) which separates genomic DNA from cytoplasm. NE is composed of inner nuclear membrane (INM), which interacts with chromatin, and outer nuclear membrane, which is connected to endoplasmic reticulum. Nuclear pore complexes are inserted into NE to form transport channels between nucleus and cytoplasm. In metazoan cells, an intermediate filament-based meshwork called as nuclear lamina exists between INM and chromatin. Sophisticated collaboration of these molecular machineries is necessary for the structure and functions of NE. Recent research advances have revealed that NE dynamically communicates with chromatin and cytoskeleton to control multiple nuclear functions. In this mini review, I briefly summarize the basic concepts and current topics of functional relationships between NE and chromatin.”
Complex dynamic interactions exist between the nuclear envelope and systems of microtubules, reflecting different rearrangements of cell architecture during different times in the cell cycle.
This has been known for some time. The 2001 publication Nuclear envelope dynamics relates: “The nuclear envelope (NE) provides a semi permeable barrier between the nucleus and cytoplasm and plays a central role in the regulation of macromolecular trafficking between these two compartments. In addition to this transport function, the NE is a key determinant of interphase nuclear architecture. Defects in NE proteins such as A-type lamins and the inner nuclear membrane protein, emerin, result in several human diseases that include cardiac and skeletal myopathies as well as lipodystrophy. Certain disease-linked A-type lamin defects cause profound changes in nuclear organization such as loss of peripheral heterochromatin and redistribution of other nuclear envelope components. While clearly essential in maintenance of nuclear integrity, the NE is a highly dynamic organelle. In interphase it is constantly remodeled to accommodate nuclear growth. During mitosis it must be completely dispersed so that the condensed chromosomes may gain access to the mitotic spindle. Upon completion of mitosis, dispersed NE components are reutilized in the assembly of nuclei within each daughter cell. These complex NE rearrangements are under precise temporal and spatial control and involve interactions with microtubules, chromatin, and a variety of cell-cycle regulatory molecules.”
Gene expression can be affected by what goes on in the nuclear envelope and what goes on in the nuclear envelope can be affected by actions of microtubules. Specifically, proteins originating in the nuclear envelope may affect epigenetic silencing of genes via histone deacetylation.
The 2007 publication Gene silencing at the nuclear periphery relates: “The nuclear envelope (NE) is composed of inner and outer nuclear membranes (INM and ONM, respectively), nuclear pore complexes and an underlying mesh like supportive structure–the lamina. It has long been known that heterochromatin clusters at the nuclear periphery adjacent to the nuclear lamina, hinting that proteins of the lamina may participate in regulation of gene expression. — Recent studies on the molecular mechanisms involved show that proteins of the nuclear envelope participate in regulation of transcription on several levels, from direct binding to transcription factors to induction of epigenetic histone modifications. Three INM proteins; lamin B receptor, lamina-associated polypeptide 2beta and emerin, were shown to bind chromatin modifiers and/or transcriptional repressors inducing, at least in one case, histone deacetylation. Emerin and another INM protein, MAN1, have been linked to down-regulation of specific signaling pathways, the retino blastoma 1/E2F MyoD and transforming growth factor beta/bone morphogenic protein, respectively. Therefore, cumulative data suggests that proteins of the nuclear lamina regulate transcription by recruiting chromatin modifiers and transcription factors to the nuclear periphery. In this minireview we describe the recent literature concerning mechanisms of gene repression by proteins of the NE and suggest the hypothesis that the epigenetic “histone code”, dictating transcriptional repression, is “written” in part, at the NE by its proteins. Finally, as aberrant gene expression is one of the mechanisms speculated to underlie the newly discovered group of genetic diseases termed nuclear envelopathies/laminopathies, elucidating the repressive role of NE proteins is a major challenge to both researchers and clinicians.”
In cells, the shape of the nuclear envelope can be affected by forces generated by microtubules. Further, microtubule-induced deformations of the nuclear envelope might affect gene expression.
The 2011 publication Microtubule-induced nuclear envelope fluctuations control chromatin dynamics in Drosophila embryos relates: “Nuclear shape is different in stem cells and differentiated cells and reflects important changes in the mechanics of the nuclear envelope (NE). The current framework emphasizes the key role of the nuclear lamina in nuclear mechanics and its alterations in disease. Whether active stress controls nuclear deformations and how this stress interplays with properties of the NE to control NE dynamics is unclear. We address this in the early Drosophila embryo, in which profound changes in NE shape parallel the transcriptional activation of the zygotic genome. We show that microtubule (MT) polymerization events produce the elementary forces necessary for NE dynamics. Moreover, large-scale NE deformations associated with groove formation require concentration of MT polymerization in bundles organized by Dynein. However, MT bundles cannot produce grooves when the farnesylated inner nuclear membrane protein Kugelkern (Kuk) is absent. Although it increases stiffness of the NE, Kuk also stabilizes NE deformations emerging from the collective effect of MT polymerization forces concentrated in bundles. Finally, we report that MT-induced NE deformations control the dynamics of chromatin and its organization at steady state. Thus, the NE is a dynamic organelle, fluctuations of which increase chromatin dynamics. We propose that such mechanical regulation of chromatin dynamics by MTs might be important for gene regulation.”
Another publication relating microtubule dynamics to the nuclear membrane (and therefore likely relating to gene expression) is the November 2012 publication Microtubule dynamics alter the interphase nucleus: “Microtubules are known to drive chromosome movements and to induce nuclear envelope breakdown during mitosis and meiosis. Here we show that microtubules can enforce nuclear envelope folding and alter the levels of nuclear envelope-associated heterochromatin during interphase, when the nuclear envelope is intact. — Microtubule reassembly, after chemically induced depolymerization led to folding of the nuclear envelope and to a transient accumulation of condensed chromatin at the site nearest the microtubule organizing center (MTOC). This microtubule-dependent chromatin accumulation next to the MTOC is dependent on the composition of the nuclear lamina and the activity of the dynein motor protein. We suggest that forces originating from simultaneous polymerization of microtubule fibers deform the nuclear membrane and the underlying lamina. Whereas dynein motor complexes localized to the nuclear envelope that slide along the microtubules transfer forces and/or signals into the nucleus to induce chromatin reorganization and accumulation at the nuclear membrane folds. Thus, our study identified a molecular mechanism by which mechanical forces generated in the cytoplasm reshape the nuclear envelope, alter the intranuclear organization of chromatin, and affect the architecture of the interphase nucleus.”
4. MICROTUBULES AND DISEASE PROCESSES
p53 is transported from the cytoplasm into the nucleus by motor proteins running along microtubules. Interfering with the dynamics of the microtubulin network could be a way of enhancing nuclear P53 concentration and its downstream pro-apoptotic effects.
Of course, p53 is a very important tumor suppressor protein(ref). The 2000 publication p53 is associated with cellular microtubules and is transported to the nucleus by dynein reported “Here we show that p53 protein is physically associated with tubulin in vivo and in vitro, and that it localizes to cellular microtubules. Treatment with vincristine or paclitaxel before DNA-damage or before leptomycin B treatment reduces nuclear accumulation of p53 and expression of mdm2 and p21. Overexpression of dynamitin or microinjection of anti-dynein antibody before DNA damage abrogates nuclear accumulation of p53. Our results indicate that transport of p53 along microtubules is dynein-dependent. The first 25 amino acids of p53 contain the residues that are essential for binding to microtubules. We propose that functional microtubules and the dynein motor protein participate in transport of p53 and facilitate its accumulation in the nucleus after DNA damage.”
A number of subsequent publications relate to the transportation of p53 by microtubules. For example, the 2002 publication Enhanced microtubule-dependent trafficking and p53 nuclear accumulation by suppression of microtubule dynamics reports: “The tumor suppressor protein p53 localizes to microtubules (MT) and, in response to DNA damage, is transported to the nucleus via the MT minus-end-directed motor protein dynein. Dynein is also responsible for MT-mediated nuclear targeting of adenovirus type 2 (Ad2). Here we show that treatment with low concentrations of MT-targeting compounds (MTCs) that do not disrupt the MT network but are known to suppress MT dynamics enhanced p53 nuclear accumulation, and the activation of the p53-downstream target genes. p53 nuclear accumulation required binding of MTCs to MTs and enhanced the induction of p53-up-regulated modulator of apoptosis (PUMA) mRNA and apoptosis on challenging cells with the DNA-damaging drug adriamycin. Low concentrations of MTCs enhanced the rate of movement of fluorescent Ad2 to the nucleus and increased the nuclear targeting efficiency of Ad2. We propose that suppression of MT dynamics by low concentrations of MTCs enhances MT-dependent trafficking toward the minus ends of MTs and facilitates nuclear targeting.”
P50 and Cep135 plays a key role in the formation of microtubules.
The 2004 publication Interaction of Cep135 with a p50 dynactin subunit in mammalian centrosomes relates: “Cep135 is a 135-kDa, coiled-coil centrosome protein important for microtubule organization in mammalian cells [Ohta et al., 2002: J. Cell Biol. 156:87-99]. To identify Cep135-interacting molecules, we screened yeast two-hybrid libraries. One clone encoded dynamitin, a p50 dynactin subunit, which localized at the centrosome and has been shown to be involved in anchoring microtubules to centrosomes. The central domain of p50 binds to the C-terminal sequence of Cep135; this was further confirmed by immunoprecipitation and immunostaining of CHO cells co-expressing the binding domains for Cep135 and p50. Exogenous p50 lacking the Cep 135-binding domain failed to locate at the centrosome, suggesting that Cep135 is required for initial targeting of the centrosome. Altered levels of Cep135 and p50 by RNAi and protein overexpression caused the release of endogenous partner molecules from centrosomes. This also resulted in dislocation of other centrosomal molecules, such as gamma-tubulin and pericentrin, ultimately leading to disorganization of microtubule patterns. These results suggest that Cep135 and p50 play an important role in assembly and maintenance of functional microtubule-organizing centers.”
In recent years there has been a practical interest in microtubules because of the potentials they appear to offer for developing anti-cancer therapies. Vincristine, Paclitaxel and colchicine are among existing anti-cancer drugs that destabilize microtubules inhibiting cell cycle progression in cancer cells. Because these drugs also affect normal issues, they pose problems of toxicity and side effects
Vincristine is a microtubule destabilizing substance(ref), “Vincristine binds to tubulin dimers, inhibiting assembly of microtubule structures. Disruption of the microtubules arrests mitosis in metaphase. Therefore, the vinca alkaloids affect all rapidly dividing cell types including cancer cells(ref).” “Paclitaxel is one of several cytoskeletal drugs that target tubulin. Paclitaxel-treated cells have defects in mitotic spindle assembly, chromosome segregation, and cell division. Unlike other tubulin-targeting drugs such as colchicine that inhibit microtubule assembly, paclitaxel stabilizes the microtubule polymer and protects it from disassembly. Chromosomes are thus unable to achieve a metaphase spindle configuration. This blocks progression of mitosis, and prolonged activation of the mitotic checkpoint triggers apoptosis or reversion to the G-phase of the cell cycle without cell division.[29][30] (ref)”
Colchicine is another microtubule-inhibiting drug, often used to treat gout as well as used for adjunct treatment of some cancers and treatment of rare inflammatory diseases.
“Colchicine inhibits microtubule polymerization by binding to tubulin, one of the main constituents of microtubules. Availability of tubulin is essential to mitosis, and therefore colchicine effectively functions as a “mitotic poison” or spindle poison(ref).[11”
An interesting aspect of these three microtubule-destabilizing drugs is that each owes its origin as a plant-based folk medicine
Microtuble networks affect not only nuclear processes but also mitochondrial functioning. Specifically, p53 inhibition together with microtubule stabilization suppress mitochondrial dysfunction.
There are important implications related to possible treatments of neurological diseases.
The October 2012 publication Inhibition of p53 transactivation functionally interacts with microtubule stabilization to suppress excitotoxicity-induced axon degeneration reports: “Axon degeneration is a hallmark of many neurological disorders, including Alzheimer’s disease, motor neuron disease, and nerve trauma. Multiple factors trigger axon degeneration, and glutamate excitotoxicity is one of them. We have recently found that stabilization of microtubules and components of the dynein–dynactin complex modulate the process of excitotoxicity-induced axon degeneration. However, the molecular mechanisms involving these microtubule-based functions remain poorly understood. Here, we used hippocampal cultures and find that inhibition of p53 transactivation and microtubule stabilization function cooperatively to suppress excitotoxicity-induced mitochondrial dysfunction. Inhibition of p53 association with mitochondria has no effect on excitotoxicity-induced mitochondrial dysfunction, however, induces axon degeneration in normal condition. Association of p150Glued with mitochondria is significantly increased by simultaneously inhibiting p53 transactivation and microtubule stabilization under excitotoxic condition. Importantly, we find that inhibition of p53 transactivation and microtubule stabilization function cooperatively to suppress excitotoxicity-induced axon degeneration. Overexpression of p150Glued does not improve the effect by inhibition of p53 transactivation on axon degeneration suggesting that p150Glued and p53 function in a linear pathway in the process of axon degeneration.”
5. MICROTUBULES AND AGING
Declining functionality of microtubule networks is associated with aging and age-related diseases.
The 1992 publication Does Aging Affect Liver Microtubules? Reported: “Microtubules are essential for many cell processes, e.g., ligand-receptor endocytosis and the vectorial movement of endosomes. The cytoskeleton, particularly microtubules, may undergo age-related changes that are reflected in cell dysfunctions. For example, the translocation of 125I-IgA-containing vesicles from the sinusoidal surface to the pericanalicular cytoplasm is reduced (>40%) in old versus young rats. Electron microscopic analysis demonstrated that the concentration of microtubule profiles in young animals is within 10–20% of that in old rats. The relative concentration of polymerized tubulin declines >70% by 12 months of age, but the total tubulin content remains unchanged until later, i.e., declining 50% by 24 months. Concomitant increases occur in the free fractions of microtubule-associated proteins (MAP), i.e., MAP, and heat-stable MAPS. These fractions are not associated with polymerized tubulin. The declines in total and polymerized tubulin, together with the increases in the MAPS’free fractions, may be indicative of fewer and/or shorter microtubules. These data lend credence to the supposition that aging is accompanied by perturbations of microtubule functions that ultimately are expressed as biomarkers characteristic of aging.”
The theme of age-related microtubule instability is picked up in the 1999 article Centrosome and microtubule instability in aging Drosophila cells. “Several cytoskeletal changes are associated with aging which includes alterations in muscle structure leading to muscular atrophy, and weakening of the microtubule network which affects cellular secretion and maintenance of cell shape. Weakening of the microtubule network during meiosis in aging oocytes can result in aneuploidy or trisomic zygotes with increasing maternal age. Imbalances of cytoskeletal organization can lead to disease such as Alzheimer’s, muscular disorders, and cancer. Because many cytoskeletal diseases are related to age we investigated the effects of aging on microtubule organization in cell cultures of the Drosophila cell model system (Schneider S-1 and Kc23 cell lines). This cell model is increasingly being used as an alternative system to mammalian cell cultures. Drosophila cells are amenable to genetic manipulations and can be used to identify and manipulate genes which are involved in the aging processes. Immunofluorescence, scanning, and transmission electron microscopy were employed for the analysis of microtubule organizing centers (centrosomes) and microtubules at various times after subculturing cells in fresh medium. Our results reveal that centrosomes and the microtubule network becomes significantly affected in aging cells after 5 days of subculture. At 5–14 days of subculture, 1% abnormal out of 3% mitoses were noted which were clearly distinguishable from freshly subcultured control cells in which 3% of cells undergo normal mitosis with bipolar configurations. Microtubules are also affected in the midbody during cell division. The midbody in aging cells becomes up to 10 times longer when compared with midbodies in freshly subcultured cells. During interphase, microtubules are often disrupted and disorganized, which may indicate improper function related to transport of cell organelles along microtubules. These results are likely to help explain some cytoskeletal disorders and diseases related to aging.”
Microtubules and age-related neurological diseases
Microtublule destabilization seems to be implicated in Alzheimer’s disease, Parkinson’s disease and other serious age-related pathologies.
The 2011 publication The Sirtuin 2 microtubule deacetylase is an abundant neuronal protein that accumulates in the aging CNS is juicy because it brings us back to our old friends the sirtuins, because it relates to microtubule deacetylation which implies weakening, and because it relates to AD and PD, two key diseases of aging. “Sirtuin 2 (SIRT2) is one of seven known mammalian protein deacetylases homologous to the yeast master lifespan regulator Sir2. In recent years, the sirtuin protein deacetylases have emerged as candidate therapeutic targets for many human diseases, including metabolic and age-dependent neurological disorders. In non-neuronal cells, SIRT2 has been shown to function as a tubulin deacetylase and a key regulator of cell division and differentiation. However, the distribution and function of the SIRT2 microtubule (MT) deacetylase in differentiated, postmitotic neurons remain largely unknown. Here, we show abundant and preferential expression of specific isoforms of SIRT2 in the mammalian central nervous system and find that a previously uncharacterized form, SIRT2.3, exhibits age-dependent accumulation in the mouse brain and spinal cord. Further, our studies reveal that focal areas of endogenous SIRT2 expression correlate with reduced α-tubulin acetylation in primary mouse cortical neurons and suggest that the brain-enriched species of SIRT2 may function as the predominant MT deacetylases in mature neurons. Recent reports have demonstrated an association between impaired tubulin acetyltransferase activity and neurodegenerative disease; viewed in this light, our results showing age-dependent accumulation of the SIRT2 neuronal MT deacetylase in wild-type mice suggest a functional link between tubulin acetylation patterns and the aging brain.”
The 2011 publication Microtubule Destabilization Is Shared by Genetic and Idiopathic Parkinson’s Disease Patient Fibroblasts reports: “Data from both toxin-based and gene-based models suggest that dysfunction of the microtubule system contributes to the pathogenesis of Parkinson’s disease, even if, at present, no evidence of alterations of microtubules in vivo or in patients is available. Here we analyze cytoskeleton organization in primary fibroblasts deriving from patients with idiopathic or genetic Parkinson’s disease, focusing on mutations in parkin and leucine-rich repeat kinase 2. Our analyses reveal that genetic and likely idiopathic pathology affects cytoskeletal organization and stability, without any activation of autophagy or apoptosis. All parkinsonian fibroblasts have a reduced microtubule mass, represented by a higher fraction of unpolymerized tubulin in respect to control cells, and display significant changes in microtubule stability-related signaling pathways. Furthermore, we show that the reduction of microtubule mass is so closely related to the alteration of cell morphology and behavior that both pharmacological treatment with microtubule-targeted drugs, and genetic approaches, by transfecting the wild type parkin or leucine-rich repeat kinase 2, restore the proper microtubule stability and are able to rescue cell architecture. Taken together, our results suggest that microtubule destabilization is a point of convergence of genetic and idiopathic forms of parkinsonism and highlight, for the first time, that microtubule dysfunction occurs in patients and not only in experimental models of Parkinson’s disease. Therefore, these data contribute to the knowledge on molecular and cellular events underlying Parkinson’s disease and, revealing that correction of microtubule defects restores control phenotype, may offer a new therapeutic target for the management of the disease.”
Going back to 2003, the publication Parkin binds to alpha/beta tubulin and increases their ubiquitination and degradation reports two interesting findings. First, toxins that cause Parkinson’s disease wreck microtubules. Second, Parkin, a microtubule-associated protein, can help protect neurons from the toxic effects of misfolded tubulins by getting rid of them via ubiquitination. “In addition to inhibiting the mitochondrial respiratory chain, toxins known to cause Parkinson’s disease (PD), such as 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine and rotenone, also strongly depolymerize microtubules and increase tubulin degradation. Microtubules are polymers of tubulin alpha/beta heterodimers, whose correct folding requires coordinated actions of cellular chaperonins and cofactors. Misfolded tubulin monomers are highly toxic and quickly degraded through a hitherto unknown mechanism. Here we report that parkin, a protein-ubiquitin E3 ligase linked to PD, was tightly bound to microtubules in taxol-mediated microtubule coassembly assays. In lysates from the rat brain or transfected human embryonic kidney (HEK) 293 cells, alpha-tubulin and beta-tubulin were strongly coimmunoprecipitated with parkin at 4 degrees C in the presence of colchicine, a condition in which tubulin exits as alpha/beta heterodimers. At the subcellular level, parkin exhibited punctate immunostaining along microtubules in rat brain sections, cultured primary neurons, glial cells, and cell lines. This pattern of subcellular localization was abolished in cells treated with the microtubule-depolymerizing drug colchicine. The binding between parkin and tubulin apparently led to increased ubiquitination and accelerated degradation of alpha- and beta-tubulins in HEK293 cells. Similarly ubiquitinated tubulins were also observed in rat brain lysates. Furthermore, parkin mutants found in PD patients did not ubiquitinate or degrade either tubulin. Taken together, our results show that parkin is a novel tubulin-binding protein, as well as a microtubule-associated protein. Its ability to enhance the ubiquitination and degradation of misfolded tubulins may play a significant role in protecting neurons from toxins that cause PD.”
Neural microtubles are reduced in aged individuals and in ones who have had Alzheimer’s disease.
The 2003 publication Microtubule reduction in Alzheimer’s disease and aging is independent of tau filament formation reports: “Biochemical studies show that phosphorylated tau, like that found in paired helical filaments (PHFs), does not promote microtubule assembly leading to the view that PHF formation leads to microtubule deficiency in Alzheimer’s disease (AD). However, although this issue is one of the most important aspects to further understanding the cell biology of AD, no quantitative examination of microtubule diminution in AD and its relationship with PHFs has been performed. To examine this issue directly, we undertook a morphometric study of brain biopsy specimens from AD and control cases. Ultrastructural analysis of neurons was performed to compare the microtubule assembly state in neurons of diseased and control cases and to examine the effect of PHF accumulation. We found that both number and total length of microtubules were significantly and selectively reduced in pyramidal neurons from AD in comparison to control cases (P = 0.000004) but that this decrement in microtubule density was surprisingly unrelated to PHFs (P = 0.8). Further, we found a significant age-dependent decrease in microtubule density with aging in the control cases (P = 0.016). These findings suggest that reduction in microtubule assembly is not dependent on tau abnormalities of AD and aging.”
Restoring microtubule acetylation levels by reducing levels of HDAC6 may offer an approach to restoring memory and learning in cases of Alzheimer’s disease.
Above, I have discussed the importance for nerve functioning of keeping microtubule proteins sufficiently acetylated. The November 2012 publication Reducing HDAC6 ameliorates cognitive deficits in a mouse model for Alzheimer’s disease reports: “Histone deacetylases (HDACs) are currently being discussed as promising therapeutic targets to treat neurodegenerative diseases. However, the role of specific HDACs in cognition and neurodegeneration remains poorly understood. Here, we investigate the function of HDAC6, a class II member of the HDAC superfamily, in the adult mouse brain. We report that mice lacking HDAC6 are cognitively normal but reducing endogenous HDAC6 levels restores learning and memory and α-tubulin acetylation in a mouse model for Alzheimer’s disease (AD). Our data suggest that this therapeutic effect is, at least in part, linked to the observation that loss of HDAC6 renders neurons resistant to amyloid-β-mediated impairment of mitochondrial trafficking. Thus, our study suggests that targeting HDAC6 could be a suitable strategy to ameliorate cognitive decline observed in AD.” My sense is that this finding, if confirmed in humans, could turn out to be very practically important.
Final comments
There is probably a lot more to be learned about microtubules, particularly as to how they relate to mitochondrial mtUPR and UPR signaling, to epigenetic effects and gene activation and to nuclear transport of key molecules such as p53. What is known, however, is that they play numerous absolutely essential roles in cell processes. There is much literature on what microtubules do and how they work. There is some literature on disrupting microtubule networks for cancer therapies. There is little explicit literature on interventions to keep microtubule networks healthy and fully functional. As usual, a number of intriguing questions are raised. For example, in addition to histone acetylation/deacetylation we need now also consider microtubule acetylation/deacetylation. What is the interplay between the two?




Dear Vincent,
Thank you for your updated (in details) information , very useful for integrative medicine clinicians.
Please, be so kind , adding the recent research on biophotons locus creation(micro-tubules) , according to A.Popp et al.
THANKS AGAIN, GEORGE
George
Thanks for the head up. Have you seen my blog entry on Quantum Biology at http://www.anti-agingfirewalls.com/2011/05/23/quantum-biology/ ?
I do discuss possible quantum effects in photosynthesis there and am familiar with some of the basic theories relating microtubules to quantum effects and speculation that they act as quantum computers – increasing the information processing power of animals by orders of magnitude beyond that explainable by the nervous system alone. It is clear to me, however, that there is a substantial body of theory and literature concerned with quantum consciousness, microtubules, photonic effects – that I know relatively little about. Prompted by you, I just came across the publication Consciousness and Quantum Information Processing: Uncovering the Foundation for a Medicine of Light (at http://online.liebertpub.com/doi/pdf/10.1089/107555304322848931) for example. My mind is currently wrapped up in other exciting topics in this blog, but at some time I will review the quantum biology area in greater depth.
Vince
Hey Vince,
I was wondering what you think of this video…
http://healthactivator.com/member-home/members-module-1-2-2/stephen-spindler/