- By Vince Giuliano and James P Watson
This blog entry generalizes on the concept of hormesis, discusses the multiple pathways through which hormesis takes place, and suggests a myriad of ways that ordinary people can take advantage of hormesis to maintain their health and possibly extend their lifespans. Some of the ideas laid out here emerged from a series of e-mail and phone exchanges between Jim Watson and myself although I (Vince) am the primary writer of this blog. We believe that hormesis is a fundamental process of human biology widely applicable in both a theoretical and practical sense across a very wide variety of health and aging issues. It is time that this concept, so long in the closet, be accorded front-stage status.
Background
Hormesis is a process through which moderate stress induces a body response that is protective against insults, confers health and possibly even longevity benefits. It is a process much mentioned in previous entries in this blog. I introduced the concept of hormesis without naming it in a July 2009 entry Stress and longevity. “Longevity is correlated with having and meeting a healthy level of challenge – not too little and not too much stress.” A later 2009 blog entry Hormesis and age retardation started out by saying “An important approach to retarding aging that I have not discussed explicitly so far is hormesis, challenging cells and body systems by mild stress resulting in them becoming stronger and resistant to aging(ref). The stress can be physical, chemical and even possibly psychological.” The definition applies to the maintenance of health as well as to slowing aging. That entry was the first to discuss the roles of heat shock and chaperone proteins in hormesis. One important heat shock protein is discussed further in the blog entry HSP70 to the rescue The relatively recent blog entries Mitohormesis and Radiation hormesis define hormesis in more detail and discuss it as it relates to mitochondrial oxidative stress and stress induced by radiation. Unlike the conventional wisdom, there is much evidence that small doses of X or gamma rays are health producing. Jim Watson’s recent post The Hormetic Wild Animal “Zoo” and Their “Zookeepers” discusses some familiar gasses like CO, NO, H2S and non- gases like HCN, O2-, and H2O2. These are Dr. Jekyll and Mr. Hyde substances. They serve as signaling molecules that act as 2nd messengers at very low doses triggering healthful body reactions in animals. Yet, at higher doses they are very toxic and even deadly.
Two sides of a familiar hormetic character
On hormesis and homeostasis
The basic purpose of hormesis is maintaining a health condition of homeostasis or better in a cell, organ or entire organism. That is, it is to keep conditions wihthin a normal functioning range and also improve that functioning. “All organisms live in a world of changing conditions. But, to remain alive, the conditions inside of every organism need to remain fairly constant. An organism must have ways to keep the conditions inside of itself from changing (too much, that is) as its external environment changes. One of the most important characteristics of all living things is the ability to maintain a (reasonably) constant internal environment. This ability is known as homeostasis(ref).” Homeostasis is not a static state; rather it is a dynamic process of constant changes and adjustments. A hormetic response to a stress not only maintains a functional internal environment but also improves it. It can be thought of as a tune-up on homeostasis.
The basic hormesis response curve
Fundamental to understanding hormesis is a dose response-curve which I have discussed in previous blog entries and included yet-again here. Forgive me, I would rather err here by redundancy rather than by incompleteness.
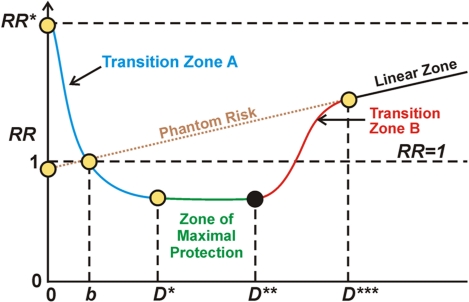
Understanding the typical dose-response curve associated with hormesis is critical for interpreting seemingly contradictory research. I explain the curve somewhat more generally here than in previous blog entries. In my interpretation, the horizontal axis depicts level of applied stress, say as driven by ROS load in a cell. On a more macroscopic level it could represent the amount of whole-body radiation received due to an exposure event or to the amount of exposure to carbon monoxide or to stress due to being at high altitude (hypoxia). The vertical axis represents relative risk, level of probable pathological organism response where normal level is 1. Below 1, there is a “health reserve,” such as an enhanced ability to do exercise or enhanced resistance to disease. Above 1 there is a less than normal “health reserve,” such as a lessened ability to resist disease or to do additional exercise. To the left of the first axis crossing in the diagram (point b), positive body reactions to the stress situation is progressively kicking but not sufficiently so as to overcome the direct negative effects of the stress. In the case discussed in other blog entries of ROS stress in cells, the Keap1-Nrf2 pathway is progressively becoming activated but in Transition zone A to the left of point b there is under-expression of the ARE genes and a negative health reserve. Between stress levels b and D*** there is hormetic protection compared to what would be expected given a linear model of negative response to stress. In the case of oxidative stress in cells, this is due at least in part to activation of Nrf2 and the ARE genes. The zone of maximum protection is between D* and D**. Starting at D** to D*** the stress load begins to overwhelm the hormetic defensive activities and the hormetic protection becomes less and less until at point D*** it vanishes. In the case of oxidative stress in cells, at that point the hormetic response associated with ARE gene activation becomes negligible. In the case of radiation damage at least, beyond point D*** the damage according to conventional wisdom is in linear proportion to the stressor, the amount of radiation. Phantom risk is theoretical risk for low stress levels that would apply if the linear model were extrapolated for low stress dosages.
We conjecture that the hormesis curve applies to all forms of stress, physical or psychological. However the scaling will vary widely from individual to individual and from one stress event to another for a given individual. We explore how some of those variances work in this blog entry.
History of hormesis – The establishment of hormesis as a sound biological principle has been a slow process and until recently many scientists did not recognize its legitimacy.
From the 2010 publication Cellular Stress Responses, The Hormesis Paradigm, and Vitagenes: Novel Targets for Therapeutic Intervention in Neurodegenerative Disorders: “Hormesis is a dose–response phenomenon characterized by a low-dose stimulation and a highdose inhibition (Fig. 1). It may be graphically represented by either an inverted U-shaped dose response or by a J- or U-shaped dose response. The term hormesis was first presented in the published literature in 1943 by Southam and Ehrlich, who reported that low doses of extracts from the red cider tree enhanced the proliferation of fungi with the overall shape of the dose response being biphasic. However, credit for experimentally demonstrating the occurrence of hormesis goes to Hugo Schulz (396), who reported biphasic dose responses in yeast after exposure to a large number of toxic agents. The work of Schulz inspired a large number of investigators in diverse fields to assess whether such low-dose effects may be a general feature of biological systems. In fact, similar types of dose–response observations were subsequently reported by numerous researchers assessing chemicals (49) and radiation (41, 50–53, 246, 307, 313, 367, 381, 397, 431,432) with investigators adopting different names such as the Arndt-Schulz Law, Huppe’s Rule, and other terms to describe these similar dose–response phenomena (368). Despite the rather substantial historical literature concerning hormetic dose responses, this concept had a difficult time being incorporated into routine safety assessment and pharmacological investigations, principally because it (a) required more rigorous evaluation in the low-dose zone, (b) failure of investigators to understand its clinical significance, (c) failure to appreciate the quantitative features of the hormetic dose response, (d) failure to understand the limitations of its implications for commercial applications in agriculture as well as medicine, (e) because of the predominant interest in responses at relatively high doses during most of the 20th century, and (f ) the continuing, yet inappropriate, tendency to associate the concept of hormesis with the medical practice of homeopathy (64, 89, 91). However, from the late 1970s (423, 433) there has been a growing interest in hormetic-like biphasic dose responses across the broad spectrum of biomedical sciences. This resurgence of interest resulted from a variety of factors, including the capacity to measure progressively lower doses of drugs and chemicals, the adoption of cell culture methods, which has permitted more efficient testing of numerous doses and the need to reexamine the validity of linearity at low-dose modeling of cancer risks due to their enormous cost implications for regulations (379), as well the astute observations of independent investigators and their capacity to generalize their findings across biological systems (267, 423).”
Hormesis is a remarkably general phenomenon, producing the same results across a wide variety of stimuli. Yet, it has taken a long time for its importance to be recognized.
“What has emerged from these research initiatives from highly diverse biomedical areas is the recognition that hormetic dose responses were common and highly generalizable, being independent of biological model, endpoints measured, and chemical class and/or physical agent studied (50–54, 68, 306, 448). This was an unexpected finding as hormetic responses were often considered by many in the so-called mainstream branches of toxicology and pharmacology to be paradoxical, not commonly expected and being of questionable reliability with a lack of capacity for replication. The casual dismissal of the hormesis concept during the mid decades of the last century is reflected in the general absence of the hormesis concept from the leading toxicological and biomedical textbooks. This situation has radically changed such that hormesis is now incorporated into all leading textbooks of toxicology (e.g., ref. 167) encyclopedias (89, 94) and other leading monographs. In fact, while the terms hormetic and hormesis were cited only about 160 times during the entire decade of the 1980s within the Web of Science database, in 2008 alone these terms were cited nearly 2300 times(ref).”
“Analysis of nearly 8,000 dose responses within the hormesis database indicates that quantitative features of phenotypic plasticity are highly generalizable, being independent of biological model, endpoint measured and chemical/physical stress inducing agent(ref).”
Hormesis played a key role in evolution.
The 2010 book The Fundamental Role of Hormesis in Evolution relates: “Hormesis can be considered a major mechanism underlying Darwin’s and Wallace’s theory of evolution by natural selection. The ability of organisms to respond adaptively to low levels of exposure to environmental hazards in a manner that increases their resistance to more severe similar or different hazards is fundamental to the evolutionary process. The organisms that survive and reproduce are those best able to tolerate or avoid environmental hazards while competing successfully for limited energy (food) resources. Therefore many of the genes selected for their survival value encode proteins that protect cells against stress (heat-shock proteins, antioxidant enzymes, antiapoptotic proteins, etc.) or that mediate behavioral responses to environmental stressors (neurotransmitters, hormones, muscle cell growth factors, etc.). Examples of environmental conditions that can, at subtoxic levels, activate hormetic responses and examples of the genes and cellular and molecular pathways that mediate such adaptive stress responses are provided to illustrate how hormesis mediates natural selection.”
So, from the viewpoint of evolution, we would expect that adaptive mechanisms would have evolved for all the important forms of stress that might be encountered by an organism. This appears to be the case. As organisms become more sophisticated the exact pathways used to produce a given stress response also evolve. For example, this diagram summarizes the hormetic effects of calorie restriction for yeast, nematodes, mice and us.
Calorie Restriction Pathways in Different Species Image source: Leonard Guarante, Mitochondria- A Nexus for Aging, Calorie Restriction, and Sirtuins?.
“In yeast, SIR2 has been implicated downstream of mitochondrial changes in response to calorie restriction (CR), whereas in mammals the SIR2 ortholog SIRT1 has been implicated upstream of mitochondrial changes. In C. elegans, sirtuins have not been implicated in dietary restriction to date. The pathway in mice shows that the increase in mitochondrial number and activity may work via the mitochondrial sirtuins SIRT3, 4, and 5 or by reducing reactive oxygen species (ROS). The drug Resveratrol and CR may increase SIRT1 activity, which is part of an autoregulatory feedback loop that includes the enzyme endothelial nitric oxide synthase (eNOS).” SIRT1 is the epicenter for linking the CR Mechanisms found in lower life forms to human CR.
A. The personal practice of hormesis
This short section of this blog entry relates to the personal practice of hormesis, Section B below will further discuss the science of hormesis.
What do you mean, “take it easy?”
Well-meaning friends often tell me that at my age I should take it easy and avoid stress. I tell them that they are 100% wrong. I thrive on stress because that is what keeps me young and going. I tell them that some stress is not only a good thing. It is absolutely necessary for health and longevity.
Hormesis is a commonly-occurring every-day phenomenon. Hormesis is also multi-factorial because multiple stimuli can activate multiple pathways producing multiple results with multiple time and response characteristics.
Let me illustrate these points with a down-home highly personal example, based on yesterday which is like most days:
A day of hormesis and me
- When I got out of bed, I woke up in a cold (60 degree F) bedroom and immediately experienced feeling cold. Though I turned up the heat, with no clothes on I was cold for 10-20 minutes while washing up and shaving until I warmly dressed. I was not cold enough to shiver or suffer but definitely cold enough to experience a body reaction. In doing this I triggered the cold shock hormetic pathway to start the morning.
- Before breakfast, I took some phytosubstance supplement pills including curcumin and ashwagandha which triggering the Nrf2 hormesis pathway. Breakfast included blueberries and walnuts, also substances triggering this pathway. The Nrf2 activation happens very quickly. It not only promotes the immediate activation of my ARE genes but also, in ways not completely known, creates a permanent recoding of parts of my epigenome.
- After breakfast coming up to the computer I found some critical software was crashed. I experienced a frustrating time wading through endless phone menus and trying to reason with a customer care representative in India whose mastery of US English was poor. Finally the problem was resolved. There was an amount of emotional/neurological stress. Heart pounding a bit, I probably triggered several hormetic stress pathways.
- Other events producing emotional/neurological stress during the day was learning that an uncle of my wife had died, a small fight with my wife about not emptying the dishwasher, and, especially, trying to get my mind around all the research involved in this hormesis issue (a repeated event). I like to think that each such event was a hormetic one.
- My daily 45 minutes on the treadmill produced exercise-related hormesis, probably via a combination of the Nrf2, heat shock and HIF pathways. I end up breathing hard and sweating. The exercise effect is not only short-term. It affects my daily circadian rhythm clock, my weight, metabolism, muscles, hunger response and critical components of epigenetic encoding.
- Chain-sawing some trees fallen by our last hurricane, I spilled a couple of drops of gasoline on my hand and breathed chainsaw fumes for a short time. This no-doubt activated the ARE detoxifying enzymes. Hopefully the toxic stresses where in the hormetic range.
- During the day I take additional phytosubstance supplement pills and drink a little green tea, again all Nrf2 hormesis activators.
- For lunch I opened a can of Italian tuna fish, a favorite of mine. This exposed me to both mercury and BHP, both toxins. Hopefully the Phase 2 detoxifying enzymes kicked in and the total effect was hormetic. To prevent chronic mercury exposure, I take some dietary supplements in my daily regimen which are heavy-metal chelators.
- I ate a couple of small packaged snacks during the day, ones probably containing traces of cancer-producing chemicals and pesticides. These may help too. Yes, at very low doses they too induce hormesis.
- Supper included salmon seasoned with olive oil, garlic, ginger, oregano and a touch of pepper sauce, broccoli and mixed greens – After supper, for desert I munched on 80% coco chocolate. All these are Nrf2 hormesis-promoting phytosubstances. I am also working on low-density lipoprotein-induced hormesis. No steak for several weeks now.
- In the process of going to bed I again exposed myself to a cold bedroom with no clothes on, like in the morning. Again, in doing this, I triggered the cold shock hormetic pathway.
These are events that I suspect produced hormetic results in me. There probably were many other hormetic events in me in the course of the day that I don’t know about.
Once hormesis was mainly regarded as a curious laboratory phenomenon that happened when you exposed pseudomonas aeruginosa to a polychlorinated dibenzodioxin in a Petri dish. Not just so! It can go off in us multiple times every day.
B. More on the science of hormesis
Mechanisms of hormesis
There are several different pathways through which hormesis can take place, including;
- The keap1/Nrf2 pathway. In simplistic terms, ROS stress in cells causes the protein keap1 to release Nrf2 which is resident in the cytoplasm whereupon Nrf2 translocates into the nucleus and activates at least 242 health-producing genes called antioxidant response elements (AREs)(ref). The same holds in the case of electrophilic stress. The basic operation and utility of this pathway is detailed in a trio of blog entries The pivotal role of Nrf2. Part 1 – a new view on the control of oxidative damage and generation of hormetic effects, The pivotal role of Nrf2. Part 2 – foods, phyto-substances and other substances that turn on Nrf2 and The pivotal role of Nrf2. Part 3– Is promotion of Nrf2 expression a viable strategy for human human healthspan and lifespan extension?.” This pathway is also discussed in the Mitohormesis blog entry. There is much more to what Nrf2 does than I have been able to cover so far. It appears for example that Nrf2 impacts on the Notch1 pathway.
- The heat shock response pathway, involving the actions of heat shock and chaperone proteins. “The cellular response to heat shock includes the transcriptional up-regulation of genes encoding heat shock proteins (HSPs) as part of the cell‘s internal repair mechanism.[1] They are also called stress-proteins.[2] and respond to heat, cold and oxygen deprivation by activating several cascade pathways. HSPs are also present in cells under perfectly normal conditions.[2] Some HSPs, called chaperones, ensure that the cell’s proteins are in the right shape and in the right place at the right time.[1][2] For example, HSPs help new or misfolded proteins to fold into their correct three-dimensional conformations, which is essential for their function.[2] They also shuttle proteins from one compartment to another inside the cell, and target old or terminally misfolded proteins to proteases for degradation.[2] Heat shock proteins are also believed to play a role in the presentation of pieces of proteins (or peptides) on the cell surface to help the immune system recognize diseased cells.[3] — The up-regulation of HSPs during heat shock is generally controlled by a single transcription factor; in eukaryotes this regulation is performed by heat shock factor (HSF) –(ref)” The blog entry the HSP70 to the rescue offers an introductory discussion of HSPs, their role in the unfolded protein response (UPR), how HSPs play their roles in multiple species, and how HSP70 plays a role in the hormesis process. Induction of hormesis by small doses of SIRT1acting through activation of HSP70 which keeps HSF1 active via deacytelation is also discussed in the blog entry SIRT1, the hypoxic response, autophagy and hormesis
- The cold shock response pathway. Yes, cold shock such as from taking a cold shower can also induce a hormetic response. This pathway involves the cold-inducible RNA binding proteins: CIRP & RBM3. Although the molecular dynamics of the cold shock response has been less-understood than the heat shock response, the cold shock hormetic response has been recognized as something going on in lower organisms for some time(ref). It is now known to be evolutionarily conserved in humans and a considerable number of publications have been devoted to this subject(ref). From Cold-shock response and cold-shock proteins (1999) “Both prokaryotes and eukaryotes exhibit a cold-shock response upon an abrupt temperature downshift. Cold-shock proteins are synthesized to overcome the deleterious effects of cold shock. CspA, the major cold-shock protein of Escherichia coli, has recently been studied with respect to its structure, funtion and regulation at the level of transcription, translation and mRNA stability. Homologues of CspA are present in a number of bacteria. Widespread distribution, ancient origin, involvement in the protein translational machinery of the cell and the existence of multiple families in many organisms suggest that these proteins are indispensable for survival during cold-shock acclimation and that they are probably also important for growth under optimal conditions.”
- The hypoxic stress response training pathway. This is the pathway activated when there is insufficient oxygen, such as at high altitudes. I briefly discussed the hypoxic response and its relationship to hormesis in the 2010 blog entry SIRT1, the hypoxic response, autophagy and hormesis. The key stress-responsive transcription factor involved in the hypoxic stress response is HIF-1. From Wikipedia: “Hypoxia-inducible factors (HIFs) are transcription factors that respond to changes in available oxygen in the cellular environment, specifically, to decreases in oxygen, or hypoxia.[1] The HIF signaling cascade mediates the effects of hypoxia, the state of low oxygen concentration, on the cell. Hypoxia often keeps cells from differentiating. However, hypoxia promotes the formation of blood vessels, and is important for the formation of a vascular system in embryos, and cancer tumors. The hypoxia in wounds also promotes the migration of keratinocytes and the restoration of the epithelium.[9]— In general, HIFs are vital to development. In mammals, deletion of the HIF-1 genes results in perinatal death. HIF-1 has been shown to be vital to chondrocyte survival, allowing the cells to adapt to low-oxygen conditions within the growth plates of bones. HIF plays a central role in the regulation of human metabolism.[10](Wiki – HIF-1).”
- UPR hormesis pathways. Heat, ROS, RNS, RSS, XRT, UV, and aging alter proteostasis resulting in the accumulation of unfolded or misfolded proteins. These stressors can up-regulate stress coping mechanisms or induce cellular damage and apoptosis. Unfolded Protein Responses (UPRs) sense and deal with accumulation of unfolded proteins, protecting the cell and blocking apoptosis. One of the UPR pathways involved in hormesis is the heat shock response pathway mentioned above. There are two other UPR pathways to be considered: the mitochondrial UPR and the endoplasmic reticulum UPR. Diagrams of how these three UPR pathways work are thought to work can be found below. The UPR plays a role in a number of diseases of aging including cancer, heart disease, cerebrovascular disease, arthritis, osteoporosis, neurodegenerative disease like Alzheimer’s and Parkinson’s and Type II diabetes.
- Cell-membrane mediated hormesis. From the 2010 publication Cellular Stress Responses, The Hormesis Paradigm, and Vitagenes: Novel Targets for Therapeutic Intervention in Neurodegenerative Disorders “Evidence is emerging to support hormetic roles for low and transient increases in membrane oxidative stress. Levels of membrane lipid peroxidation are relatively low under most normal conditions. However, in some types of cells, lipid peroxidation increases considerably during periods of increased energy demand. For example, during vigorous physical exercise there is a marked increase in production of superoxide and hydrogen peroxide, hydroxyl radical, peroxynitrite, and lipid peroxidation (377). Evidence suggests that free radicals and products of lipid peroxidation generated during moderate exercise play important roles in hormetic effects of exercise on muscles, including changes in energy metabolism pathways, mitochondrial biogenesis, and upregulation of protein chaperones and antioxidant systems (377). Benefits of exercise on the cardiovascular system may also involve membrane oxidative stress-related mechanisms. Thus, it was reported that HNE activates nuclear factor erythroid 2-related factor 2 (Nrf2) and antioxidant gene expression in vascular cells (412). HNE may also activate other adaptive stress response pathways that promote the survival and plasticity of cells (349). Ceramide is also believed to mediate hormetic effects of moderate/transient increases in membrane-associated oxidative stress. For example, pretreatment of neurons with subtoxic concentrations of ceramide results in increased resistance of the neurons to subsequent high levels of oxidative stress (191). Other studies have provided evidence for a pivotal role for ceramide in the cardioprotective effect of preconditioning ischemia in animal models of myocardial infarction (16, 149). Preconditioning ischemia is a classic example of hormesis, wherein exposure of cells to a moderate transient stress protects them against more severe stresses. Changes in the PMRS in response to stress may also allow cells to adapt to potentially damaging conditions. A dramatic example comes from a study in which the mitochondria of cellswere rendered dysfunctional, and the cells were able to survive because of a compensatory upregulation of PMRS enzyme activities (212).”
Hormesis operates through multiple channels for cell survival, dependent on the stimulation.
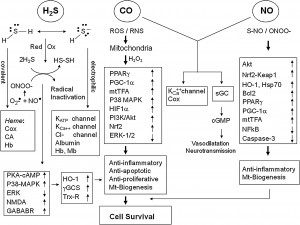
Illustrating a few of many possible situations, the following diagram illustrates the signaling operations for hormesis originated by three toxic gasses, hydrogen sulfide, carbon monoxide and nitric oxide:
Regarding these noxious gasses and hormesis, see Jim Watson’s recent blog entry The Hormetic Wild Animal “Zoo” and Their “Zookeepers.”
Hormesis, specifically the scaling of the hormesis curve, depends on several critical factors:
- Hormetic dose response is a critical consideration encountered in every situation of hormesis. The hormetic dose response curve is central to the process. Not enough or too much of a given stress can be harmful. A tiny whiff of carbon monoxide may be good for you but too much will kill you. Avoiding all stress in your life could be a good approach to early death.
- The time duration of the hormesis-generating stress is critical. If a hormetic level of stress is maintained too long, the result could be non-hormetic and dangerous. An example could be exercising to utter exhaustion; another would be breathing a very low level of carbon monoxide for too long.
- To be effective, the stress must be pulsed; it cannot be chronic. Chronic exposure to even very low levels of carcinogens, like second-hand tobacco smoke, could result in negative effects outside the hormesis zone. Another example is that radiation hormesis cannot be expected from a chronic stress source like radiation from Strontium70 absorbed in bones. You might be able to experience hormesis from cold shock by taking a cold shower, but if you stay in the shower too long your immune system might not be able to protect you from viral infections like common colds.
- Multiple stressors that address a certain pathway response may produce no better hormetic benefits than single ones. From Hormesis provides a generalized quantitative estimate of biological plasticity: “These quantitative features of the hormetic dose response have important medical implications. Most significantly, the hormetic dose response imposes constraints upon the magnitude of a drug to induce a desired effect. For example, if a drug increased cognitive performance in an elderly patient by approximately 25%–30%, the hormetic model suggests that this level of performance could not be further increased using a new drug combination. This concept has been supported in a variety of studies on hormesis and drug interaction. Flood (173–176) has demonstrated that the hormetic response for memory was bounded by the 30%–60% increase even when several drugs were used in combination that were designed to maximize memory outcome. This response magnitude constraint has been reported for immune stimulation, bacterial growth, increases in hair growth, plant growth, decrease in anxiety, decreases in tumor incidence, and numerous other endpoints (73).” The limits of hormetic responses are set by the nature of the hormetic response machinery that is involved. For example, consuming a phytosubstance like green tea may cause release of Nrf2 into the nucleus of cells activating ARE genes and creating a healthful hormetic response. But there is only so much Nrf2 sitting around in the cytoplasm of cells at any given time. So consuming green tea, curcumin, resveratrol, and ginger pills in the same gulp of pills may release no more Nrf2 or produce no better results – and might even exceed the hormetic dose.
- Periods of rest are required between stress impulses. For example, we all know that periods of rest are required between bouts of exercise. We conjecture that the cells require time to replenish stocks of Nrf2 in the cytoplasm after it is suddenly released into the nucleus. After oxidative stress the body requires time to clear out oxidation byproducts and otherwise re-establish homeostasis.
- Hormesis is most effective when synchronized with circadian time frame windows. For example, getting up from sleep and exercising at 3AM is not a good idea. There are likely to be a number of other windows-of-best-opportunity for other circadian clocks. This is an area requiring further research.
- The body can increase its stress tolerance and affect the height and breadth of the hormetic response curve to a stressor by repeated hormetic exposures to the stressor.
- A familiar example is exercise endurance training, where a runner may gradually increase his running time and speed to levels unthinkable for an ordinary individual. Another example relates to pot, yes, marijuana. THC, the active ingredient in marijuana, appears to be a hormetic molecule capable of promoting neurogenesis at low doses (probably smaller doses than ones from puffing on a marijuana cigarette)(ref). I know a couple of individuals who are regular marijuana smokers and appear consistently to be functional and mentally present. They tell me they smoke the substance ever day or so and that it leads them to clearer thinking. I conjecture that they have increased their stress tolerance to THC. I also know, based on personal experimentation during my hippy days 45 years ago or so, that just a few puffs of the same weed these friends are smoking would stop me from thinking clearly for a week or more. Another good example of increasing stress tolerance through repetition discussed below is myocardial ischemic preconditioning.
Jim Watson summarized these hormesis factors to me in a phone conversation. Though they are “of course” common sense observations, I have never seen them written down in one place.
The impacts of simple hormetic events on health and longevity might be profound and long-lasting.
Take for example, simple everyday events which activate Nrf2. I am talking about eating some broccoli, drinking some green tea, swallowing a curcumin or resveratrol capsule, and many other sucheveryday actions. We know Nrf2 activates the body’s endogenous antioxidant defense system. But there is solid research that says it does much more including.
- Affecting the differentiation and apoptosis of stem cell populations so as to affect organ regeneration and lifespan.
- Affecting the repair of damaged DNA and tissues. Many believe accumulated DNA damage is a fundamental driver of aging.
- Responding to electrophilic stress. It is possible that electrophilic stress is a more important cause of cellular aging than oxidative stress.
- Creating permanent changes to one’s epigenome.
Although we do not know the magnitude or relative importance of these responses, I briefly cite evidence for each one.
Hormetic expression of Nrf2 can affect the differentiation and apoptosis of stem cell populations so as to affect organ regeneration and lifespan.
The 2010 publication Regulation of Notch1 Signaling by Nrf2: Implications for Tissue Regeneration makes the case in point. “–Through transcriptional analyses in Keap1– or Nrf2-disrupted mice, we identified interactions with the Notch1 signaling pathway. We found a functional antioxidant response element (ARE) recognized by Nrf2 in the promoter of Notch1. Notch1 regulates processes such as proliferation and cell fate decisions. We report a functional role for this cross talk between the two pathways and show that disruption of Nrf2 impeded liver regeneration following partial hepatectomy and was rescued by re-establishment of Notch1 signaling. — The Notch family of transmembrane receptors participates in a signaling pathway controlling a broad spectrum of metazoan cell fates and developmental processes through local cell-cell interactions (21). Alteration of signaling through the Notch family of receptors can markedly affect differentiation, proliferation, and apoptotic events. Genetic ablation studies indicate that Notch1 is crucial for early development and re-growth of several tissues (22, 23). Activation of the Notch pathway inhibits differentiation in different developmental contexts and has been associated with the amplification of some somatic stem cells— not only the neural (24) and hematopoietic stem cells (25), but also hepatocyte (26, 27) and intestinal epithelial stem cells (28, 29). Considering the importance of the Notch1 signal cascade in developmental biology, the microarray observations indicated the possibility that Nrf2 could be a key molecule affecting both embryonic and adult tissue stem cell renewal as well as cell fates. This study characterizes the effects of Nrf2 genotype on the expression of Notch1 and its effector genes and the importance of Nrf2-Notch1 crosstalk in liver regeneration.”
Hormetic expression of Nrf2, among other impacts, can affect the repair of damaged DNA and tissues.
This point is illustrated in the following graphic from the publication When NRF2 Talks, Who’s Listening?

Possible means for regulation of cell survival and other cell-fate responses through interactions of NRF2 with additional cell-signaling pathways, including AhR, NF-κB, p53, and Notch1.
“Nonetheless, it is clear that the protective effects of upregulation of NRF2 signaling can take several forms. Protection can be immediate, reflecting induction of genes directly regulated through NRF2 binding to AREs in target genes (e.g., the innate immune response and elevated cytoprotective responses to blunt cytokine surges or detoxify reactive intermediates, respectively) (73). The protective effects can be secondary through induction of macromolecular damage repair/removal systems (proteasome, DNA repair) (84, 106). Last, the protective effects can be tertiary through activation of tissue repair/regeneration pathways. In these latter cases, involvement in cross talk with additional pathways affecting cell survival and other aspects of cell fate most certainly play important collaborating roles.”
Like oxidative stress, Nrf2 hormetically responds to electorphilic stress via the Keap1 pathway. This phenomenon may be highly relevant to aging.
Electrophilic stress is stress induced by the stealing of pairs of electrons from compounds in cells by electrophiles, creating new and sometimes radical molecular structures. “In general, electrophiles are positively charged species that are attracted to an electron rich centre. In chemistry, an electrophile (literally electron-lover) is a reagent attracted to electrons that participates in a chemical reaction by accepting an electron pair in order to bond to a nucleophile.(ref)” Electrophilic stress is often discussed along with oxidative stress for both appear to activate Nrf2 via a common process.
As stated in Relationship of electrophilic stress to aging: “In the present review, I will discuss the nature of electrophilic stress and its role in aging. I hope to present compelling evidence that electrophiles are, in fact, a long-neglected causal contributor to aging, and that electrophilic stress, while initiated by an oxidative event, is distinct, and can be functionally decoupled, from oxidative stress. — “This review begins with the premise that an organism’s life span is determined by the balance between two countervailing forces: (i) the sum of destabilizing effects and (ii) the sum of protective longevity-assurance processes. Against this backdrop, the role of electrophiles is discussed, both as destabilizing factors and as signals that induce protective responses. Because most biological macromolecules contain nucleophilic centers, electrophiles are particularly reactive and toxic in a biological context. The majority of cellular electrophiles are generated from polyunsaturated fatty acids by a peroxidation chain reaction that is readily triggered by oxygen-centered radicals, but propagates without further input of reactive oxygen species (ROS). Thus, the formation of lipid-derived electrophiles such as 4-hydroxynon-2-enal (4-HNE) is proposed to be relatively insensitive to the level of initiating ROS, but to depend mainly on the availability of peroxidation-susceptible fatty acids. This is consistent with numerous observations that life span is inversely correlated to membrane peroxidizability, and with the hypothesis that 4-HNE may constitute the mechanistic link between high susceptibility of membrane lipids to peroxidation and shortened life span.” – “Where do biologically relevant electrophiles come from? There are two major sources of such compounds. The first is external. Xenobiotics can be present in food, especially of plant origin, can be inhaled, or can be administered on purpose, e.g., as pharmacological agents. Many xenobiotics are directly electrophilic or can be metabolically converted to electrophiles [activation of toxins or drugs; see ref. 8 for a review]. The other source of electrophiles is the cell’s own metabolism. Certain intermediary metabolites are electrophilic.” “The — paradigm I am proposing, (is) namely that electrophiles such as 4-HNE are relevant to aging, and that the formation of 4-HNE is largely decoupled from the initiating oxidative stress but is a function of membrane peroxidizability.”
Electrophile Response System for Cellular Stress Tolerance
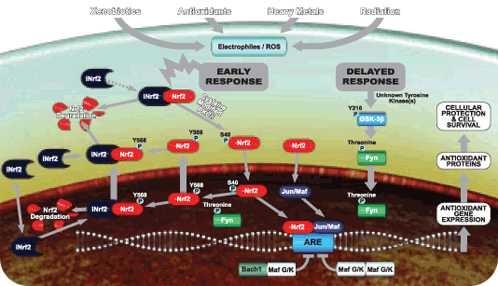 Image source Overview of Activation and Inactivation of Nrf2 Signaling by electrophiles or ROS.
Image source Overview of Activation and Inactivation of Nrf2 Signaling by electrophiles or ROS.
“Normally, INrf2 directs the degradation of Nrf2 by recruiting Cul3/Rbx1. After cells are exposed to stressors such as reactive oxygen species, INrf2 undergoes cysteine modification and Nrf2 is phosphorylated by PKC, resulting in dissociation of the INrf2:Nrf2 heterodimers. Phosphorylated Nrf2 translocates to the nucleus and binds the Antioxidant Response Element (ARE) with either small MAF or Jun. This leads to antioxidant gene expression that protects the cell, (A). The pathway is subsequently inactivated by two separate mechanisms. First, Maf homodimers and Bach1:Maf heterodimers compete with Nrf2 for ARE binding, resulting in diminished antioxidant gene expression. Second, GSK3b phosphorylates Fyn which leads to the Fyn translocating to the nucleus. Fyn subsequently phosphorylates Nrf2 at Tyr568. Nrf2 then is exported from the nucleus, binds to INrf2 and is subsequently degraded, (B).”
Hormetic expression of Nrf2 may create permanent health-inducing modifications to one’s epigenome.
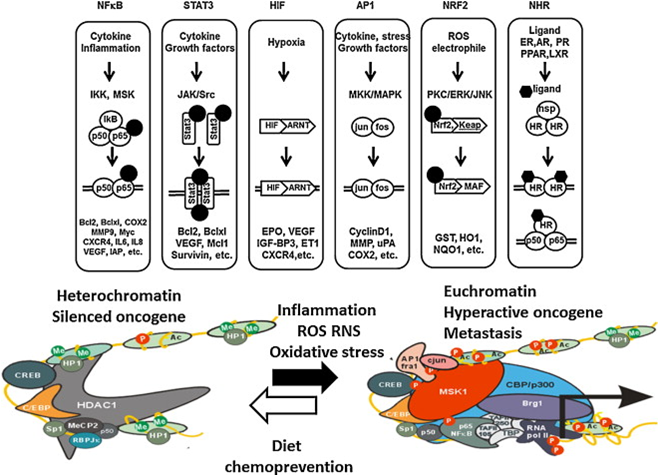
The idea of permanently changing one’s epigenome in a positive manner by eating a bit of broccoli or kale or exercising a little may not be completely preposterous. Consider the following three graphics from the publication Epigenetic impact of dietary polyphenols in cancer chemoprevention: Lifelong remodeling of our epigenomes
“Dietary modulation of transcription factor pathways which regulate chromatin “oscillation” dynamics between euchromatic and heterochromatic states at oncogenes and tumor suppressor genes.”
You can control your level of cellular stress and remodel your epigenome for health and longevity at the Dinner Table
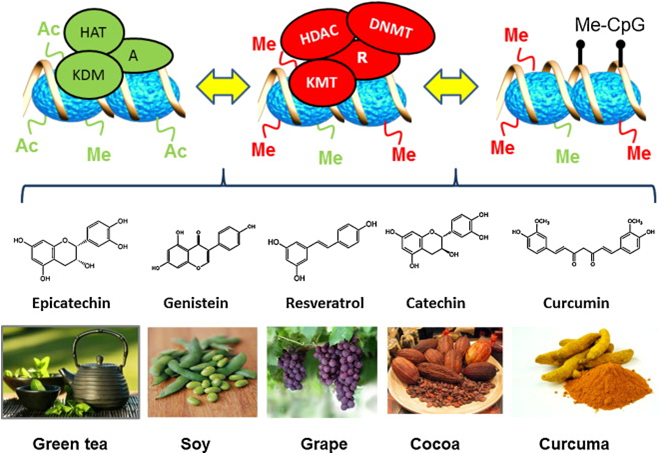
Epigenetic actions of common foods
“Development of functional foods or dietary supplements as nutrition based epigenetic modulators of chromatin writers, readers and erasers in cancer chemoprevention. HAT, histone acetyltransferase; HDAC, histone deacetylase; DNMT, DNA methyltransferase; KMT, lysine methyltransferase; KDM, lysine demethylase; Me-CpG, Methylcytosine; R, transcription repressor; A, Transcription activator; Ac, acety; Me, methyl.”
There appears to be no end to the list of toxic and carcinogenic substances that induce hormesis at very low doses. It is tempting to say that all toxic substances administered in pulsed intermittent doses qualify for hormesis.
The blog entry The Hormetic Wild Animal “Zoo” and Their “Zookeepers” describes some of these substances. Another of many examples is described in the 2006 publication Alpha-benzene hexachloride exerts hormesis in preneoplastic lesion formation of rat hepatocarcinogenesis with the possible role for hepatic detoxifying enzymes. “Recently there has been a shift in the prevailing paradigm regarding the dose dependence of carcinogen action with increasing acceptance of hormesis phenomenon, although underlying mechanisms remain to be established. To ascertain whether alpha-benzene hexachloride (alpha-BHC) might act by hormesis, rats were initiated with diethylnitrosamine and then alpha-BHC ranging from 0.01 to 500 ppm was administered in the diet for 10 weeks. The highest concentration of alpha-BHC significantly increased the number and area of glutathione S-transferase placental form (GST-P) positive foci, preneoplastic lesions in the liver, but its low dose, 0.05 ppm, caused significant reduction, showing a J-shape dose-response curve. The proliferating cell nuclear antigen positive index for GST-P positive foci in the low dose-treated group was significantly reduced. The dose response curves of CYP450 content, NADPH-P450 reductase activity and 8-hydroxydeoxyguanosine formation revealed the same pattern as GST-P positive foci data. The response curves of CYP2B1 and 3A2 in their activities, protein and mRNA expression showed a threshold but CYP2C11 activity exhibited an inverted J-shape. These results might suggest the possibility of hormesis of alpha-BHC at early stages of rat hepatocarcinogenesis. The possible mechanism involves induction of detoxifying enzymes at low dose, influencing free radical production and oxidative stress, and consequently pathological change in the liver.”
Of course, multiple other stresses besides substances can induce hormetic responses: too much cold or heat, radiation, surgery, emotional stresses of many kinds, too bright lights, UV exposure, lack of sleep, lack of oxygen, physical injury, pain, many diseases, etc. etc. I conjecture that evolution has prepared a hormetic response for just about every kind of stress we commonly encounter.
The unfolded protein responses (UPRs) are important stress-response pathways subject to hormesis
As already mentioned, there are three Unfolded Protein Responses. All three of the UPR pathways are activated in response to protein stress.
Mitochondrial UPR
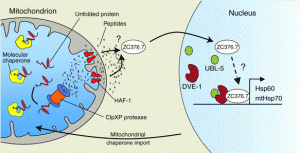
mtUPR in C. elegans
“A model for mitochondrial UPR signaling in C. elegans. It is assumed that signaling within the UPRmt initiates when the unfolded protein load in the matrix exceeds the capacity of the mitochondrial chaperones. The AAA+ protease ClpXP degrades unfolded or unassembled proteins to peptides, which are pumped across the inner membrane by the ABC-transporter HAF-1 and then cross the more porous outer membrane to the cytosol. The presence of peptides in the cytosol, the process of peptide efflux or some linked activity of HAF-1 leads to the activation and nuclear translocation of the bZip transcription factor ZC376.7; however, the underlying mechanism(s) have yet to be identified. Additionally, the homeobox protein DVE-1 and UBL-5 form a complex and bind to the hsp-60 promoter potentially remodeling chromatin structure to promote ZC376.7 binding and transcriptional activation. Transcriptional upregulation of mitochondrial chaperone genes leads to their subsequent import into mitochondria, thus relieving stress and re-establishing homeostasis.”
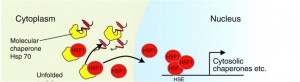
Cytoplasmic UPR (AKA Heat Shock response)
Endoplasmic reticulum UPR
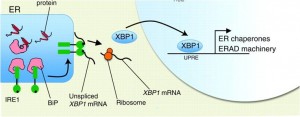
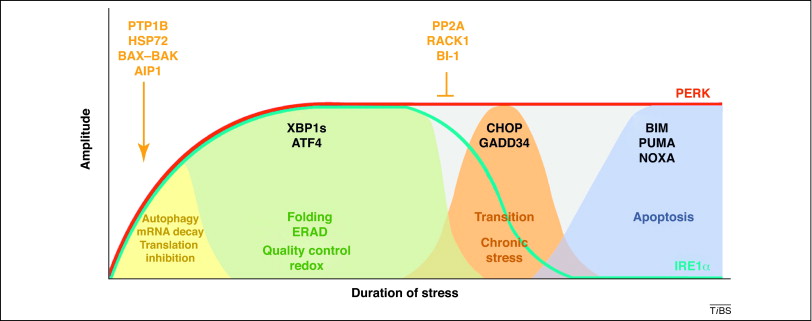
The relationship between duration and amplitude of stress to cell response in the endoplasmic reticulum UPR response is illustrated in the following graphic. If the stress is too great or lasts too long, the cell commits apoptosis.
Image source – “Figure 1. Kinetics of UPR signaling and cell fate decisions. The accumulation of abnormally folded proteins in the ER engages an adaptive stress response known as the UPR. Temporally distinct UPR-related events are observed in cells undergoing ER stress as a means to determine cell fate decisions. Two major ER stress sensors, IRE1α and PERK, transduce information about the folding status of the ER to the cytosol and nucleus to recover folding capacity. Whereas IRE1α is downregulated under conditions of chronic ER stress, PERK signaling is sustained, possibly sensitizing cells to apoptosis (blue and red lines, respectively). In a first, acute signaling response, IRE1α and PERK activity attenuates protein synthesis at the ER by decreasing protein translation and controlling the decay of mRNAs encoding ER proteins. Autophagy is also activated to remove aggregated proteins and damaged organelles. Then, downstream of PERK and IRE1α, the transcription factors XBP1s and ATF4 are expressed, leading to the upregulation of many genes related to folding, quality control, ERAD and redox metabolism. After prolonged ER stress, IRE1α is turned off, thereby downregulating XBP1s. Downstream of ATF4, expression of the transcription factor CHOP, and other related events, can regulate the transition from adaptation/survival events to a pro-apoptotic phase. This late fourth wave of signaling events is associated with the upregulation of many genes related to the BCL2 protein family including BIM, PUMA and NOXA, thereby activating the canonical mitochondrial apoptosis pathway. During the course of ER stress, a dynamic modulation of IRE1α signaling occurs. Several regulators assemble into the IRE1α scaffold to regulate its activity in terms of kinetic, amplitude and tissue specificity. This signaling platform is termed the UPRosome, and several interacting factors, including PTP1B, AIP1, HSP72, BAX and BAK, increase the amplitude of IRE1α signaling. Following prolonged ER stress, IRE1α returns to a latent state, a process modulated by an interaction with BI-1 and possibly the phosphatase PP2A in complex with RACK1.”
The heat shock response is relevant to aging and to possible life extension in humans.
UPR-regulated Heat Shock Proteins decline with age. Cells in young animals rapidly alter levels of HSPs. Older animals lose the ability to induce HSPs and other stress response proteins. This has been observed in yeast, worms, flies, and mouse and human cell lines.
Further, over-expression of Heat Shock Proteins extends lifepan at least in some lower species. This has been observed in Drosophila. The lifespan extension seems to be due to impacts on mitochondrial OXPHOS processes. See Gene expression profiling implicates OXPHOS complexes in lifespan extension of flies over-expressing a small mitochondrial chaperone, Hsp22. “Aging is a complex process accompanied by a decreased capacity to tolerate and respond to various stresses. Heat shock proteins as part of cell defense mechanisms are up-regulated following stress. In Drosophila, the mitochondrial Hsp22 is preferentially up-regulated in aged flies. Its over-expression results in an extension of lifespan and an increased resistance to stress. Hsp22 has chaperone-like activity in vitro, but the mechanism(s) by which it increases lifespan in flies are unknown. Genome-wide analysis was performed on long-lived Hsp22+ and control flies to unveil transcriptional changes brought by Hsp22. Transcriptomes obtained at 45 days, 90% and 50% survival were then compared between them to focus more on genes up- or down-regulated in presence of higher levels of hsp22 mRNA. Hsp22+ flies display an upregulation of genes mainly related to mitochondrial energy production and protein biosynthesis, two functions normally down-regulated during aging. Interestingly, among the 26 genes up-regulated in Hsp22+ flies, 7 genes encode for mitochondrial proteins, 5 of which being involved in OXPHOS complexes. Other genes that could influence aging such as CG5002, dGCC185 and GstS1 also displayed a regulation linked to Hsp22 expression. The up-regulation of genes of the OXPHOS system in Hsp22+ flies suggest that mitochondrial homeostasis is at the center of Hsp22 beneficial effects on lifespan.”
Some of the stressors that induce the HSP hormetic response are illustrated in this graphic:
“Triggering factors in heat-shock-protein-directed immunoregulation”
Image source: Heat-shock proteins induce T-cell regulation of chronic inflammation
The key transcription factor protein involved in the hypoxic stress response, HIF-1, modulates lifespan in lower species.
The 2009 publication The HIF-1 Hypoxia-Inducible Factor Modulates Lifespan in C. elegans relates: “During normal development or during disease, animal cells experience hypoxic (low oxygen) conditions, and the hypoxia-inducible factor (HIF) transcription factors implement most of the critical changes in gene expression that enable animals to adapt to this stress. Here, we examine the roles of HIF-1 in post-mitotic aging. We examined the effects of HIF-1 over-expression and of hif-1 loss-of-function mutations on longevity in C. elegans, a powerful genetic system in which adult somatic cells are post-mitotic. We constructed transgenic lines that expressed varying levels of HIF-1 protein and discovered a positive correlation between HIF-1 expression levels and lifespan. The data further showed that HIF-1 acted in parallel to the SKN-1/NRF and DAF-16/FOXO transcription factors to promote longevity. HIF-1 over-expression also conferred increased resistance to heat and oxidative stress. We isolated and characterized additional hif-1 mutations, and we found that each of 3 loss-of-function mutations conferred increased longevity in normal lab culture conditions, but, unlike HIF-1 over-expression, a hif-1 deletion mutation did not extend the lifespan of daf-16 or skn-1 mutants. We conclude that HIF-1 over-expression and hif-1 loss-of-function mutations promote longevity by different pathways. These data establish HIF-1 as one of the key stress-responsive transcription factors that modulate longevity in C. elegans and advance our understanding of the regulatory networks that link oxygen homeostasis and aging.
During normal development or during disease, animal cells experience hypoxic (low oxygen) conditions, and the hypoxia-inducible factor (HIF) transcription factors implement most of the critical changes in gene expression that enable animals to adapt to this stress. Here, we examine the roles of HIF-1 in post-mitotic aging. We examined the effects of HIF-1 over-expression and of hif-1 loss-of-function mutations on longevity in C. elegans, a powerful genetic system in which adult somatic cells are post-mitotic. We constructed transgenic lines that expressed varying levels of HIF-1 protein and discovered a positive correlation between HIF-1 expression levels and lifespan. The data further showed that HIF-1 acted in parallel to the SKN-1/NRF and DAF-16/FOXO transcription factors to promote longevity. HIF-1 over-expression also conferred increased resistance to heat and oxidative stress. We isolated and characterized additional hif-1 mutations, and we found that each of 3 loss-of-function mutations conferred increased longevity in normal lab culture conditions, but, unlike HIF-1 over-expression, a hif-1 deletion mutation did not extend the lifespan of daf-16 or skn-1 mutants. We conclude that HIF-1 over-expression and hif-1 loss-of-function mutations promote longevity by different pathways. These data establish HIF-1 as one of the key stress-responsive transcription factors that modulate longevity in C. elegansand advance our understanding of the regulatory networks that link oxygen homeostasis and aging.”
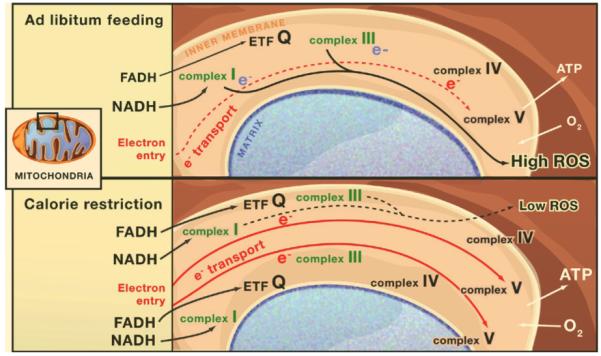
One of the consequences of calorie restriction and certain other hermetic stresses on mitochondria is mitochondrial biogenesis.
The impact described relates to electron transport chains, a topic introduced in the recent blog entry Mitochondria in health and aging, and possibilities for life prolongation – Part 1:basics. The situation is described in this diagram:
Image source: Leonard Guarante, Mitochondria- A Nexus for Aging, Calorie Restriction, and Sirtuins?. “Mitochondrial Biogenesis and Reactive Oxygen Species. Shown is mitochondrial biogenesis during calorie restriction versus ad libitum feeding in mice and its proposed effects on reactive oxygen species (ROS). In the ad libitum case, the number of electron transport chains is low, and if the rate of entry of electrons (red e–) exceeds the slowest step of flow through the chain, stalling of electrons at mitochondrial complexes I and III (blue e–) and production of ROS will be favored. During calorie restriction, mitochondrial biogenesis increases the number of electron transport chains, thereby reducing the rate of electron entry per electron transport chain. Calorie restriction may also increase the fraction of electrons that bypass complex I by entering the electron transport chain via the electron transfer flavoprotein dehydrogenase (ETF). These effects may reduce the production of ROS during calorie restriction and hence mitigate cellular damage, aging, and disease.”
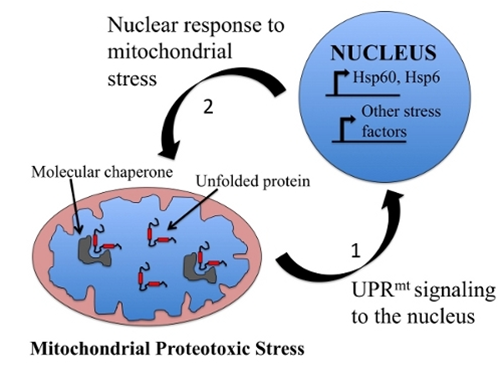
A response to mitochondrial stress appears to be up-regulation of heat shock proteins.
The diagram and this text are from Conservation of the Mitochondrial Unfolded Protein Response in Aging. “The mitochondrial unfolded protein response (UPRmt) has been shown to play an important role in aging of C. elegans by studies from the Dillin Lab. The inhibition of mitochondrial activity can extend lifespan in worms. This is not simply the result of lowered oxidative damage, but requires induction of a mitochondrial stress response known as the UPRmt. The UPRmt is thought to signal from the mitochondria to the nucleus to cause changes in gene expression to improve mitochondrial protein homeostasis in a cell autonomous and non-autonomous fashion. We are performing genetic screens to identify key genes involved in this response in order to better understand communication between the mitochondria and nucleus and its important role in aging. These screens are being performed in both yeast and C. elegans in order to understand which aspects of the UPRmt have been evolutionarily conserved and whether the importance of this pathway in aging is also conserved.”
Low –density lipoproteins induce hormesis, most likely via the Nrf2/Keap1 pathway.
The 0820 publication Characterization of oxidized low-density lipoprotein-induced hormesis-like effects in osteoblastic cells reports: “Epidemiological studies indicate that patients suffering from atherosclerosis are predisposed to develop osteoporosis. Atherogenic determinants such as oxidized low-density lipoprotein (oxLDL) particles have been shown both to stimulate the proliferation and promote apoptosis of bone-forming osteoblasts. Given such opposite responses, we characterized the oxLDL-induced hormesis-like effects in osteoblasts. Biphasic 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) reductive activity responses were induced by oxLDL where low concentrations (10-50 microg/ml) increased and high concentrations (from 150 microg/ml) reduced the MTT activity. Cell proliferation stimulation by oxLDL partially accounted for the increased MTT activity. No alteration of mitochondria mass was noticed, whereas low concentrations of oxLDL induced mitochondria hyperpolarization and increased the cellular levels of reactive oxygen species (ROS). The oxLDL-induced MTT activity was not related to intracellular ROS levels. OxLDL increased NAD(P)H-associated cellular fluorescence and flavoenzyme inhibitor diphenyleneiodonium reduced basal and oxLDL-induced MTT activity, suggesting an enhancement of NAD(P)H-dependent cellular reduction potential. Low concentrations of oxLDL reduced cellular thiol content and increased metallothionein expression, suggesting the induction of compensatory mechanisms for the maintenance of cell redox state. These concentrations of oxLDL reduced osteoblast alkaline phosphatase activity and cell migration. Our results indicate that oxLDL particles cause hormesis-like response with the stimulation of both proliferation and cellular NAD(P)H-dependent reduction potential by low concentrations, whereas high concentrations lead to reduction of MTT activity associated with the cell death. Given the effects of low concentrations of oxLDL on osteoblast functions, oxLDL may contribute to the impairment of bone remodeling equilibrium.”
Myocardial ischemic preconditioning
Myocardial ischemic preconditioning (IPC) is an example of a hormetic process featuring the aspects of pulsing and increasing stress tolerance – even though the literature on IPC rarely if ever mentions the word “hormesis.” “IPC is an intrinsic process whereby repeated short episodes of ischaemia protect the myocardium against a subsequent ischaemic insult(ref).” “The myocardium possesses innate physiologic adaptive processes that render it more resistant to potentially lethal ischemic injury. A number of these adaptive mechanisms have been identified; one is the phenomenon of ischemic preconditioning which provides the myocardium with the most powerful means of delaying myocardial infarction that has been identified. Ischemic preconditioning refers to the protection conferred to ischemic myocardium by preceding brief periods of sublethal ischemia(ref)”The 2002 review article Ischemic preconditioning of myocardium related in summary “Preconditioning of the myocardium with short episodes of sublethal ischemia will delay the onset of necrosis during a subsequent lethal ischemic insult. Ischemic preconditioning seems to involve a variety of stress signals which include activation of membrane receptors and signaling molecules such as protein kinase C, mitogen-activated protein kinases, opening of ATP-sensitive potassium channel, and expression of many protective proteins.”IPS is a powerful tool of hormesis that increasingly is being utilized in medical practice, particularly in open cardiac surgery(ref)(ref). The 2007 publication Ischemic preconditioning: Protection against myocardial necrosis and apoptosis reported: “The phenomenon of ischemic preconditioning has been recognized as one of the most potent mechanisms to protect against myocardial ischemic injury. In experimental animals and humans, a brief period of ischemia has been shown to protect the heart from more prolonged episodes of ischemia, reducing infarct size, attenuating the incidence, and severity of reperfusion-induced arrhythmias, and preventing endothelial cell dysfunction. Although the exact mechanism of ischemic preconditioning remains obscure, several reports indicate that this phenomenon may be a form of receptor-mediated cardiac protection and that the underlying intracellular signal transduction pathways involve activation of a number of protein kinases, including protein kinase C, and mitochondrial KATPchannels. Apoptosis, a genetically programmed form of cell death, has been associated with cardiomyocyte cell loss in a variety of cardiac pathologies, including cardiac failure and those related to ischemia/reperfusion injury. While ischemic preconditioning significantly reduces DNA fragmentation and apoptotic myocyte death associated with ischemia-reperfusion, the potential mechanisms underlying this effect have not been fully clarified.”
Hormetic protectivity of polyphenols, as related to cellular stress and epigenetics
Again and again in these blog entries, I have highlighted the protective hormetic roles of plant-based polyphenol substances. I do so because I have become convinced that a dietary regimen that features consuming these substances when coupled with good lifestyle habits can produce significant health and longevity benefits right now. No need to wait for further research since enough is known already. No need to wait for new miracle drugs.
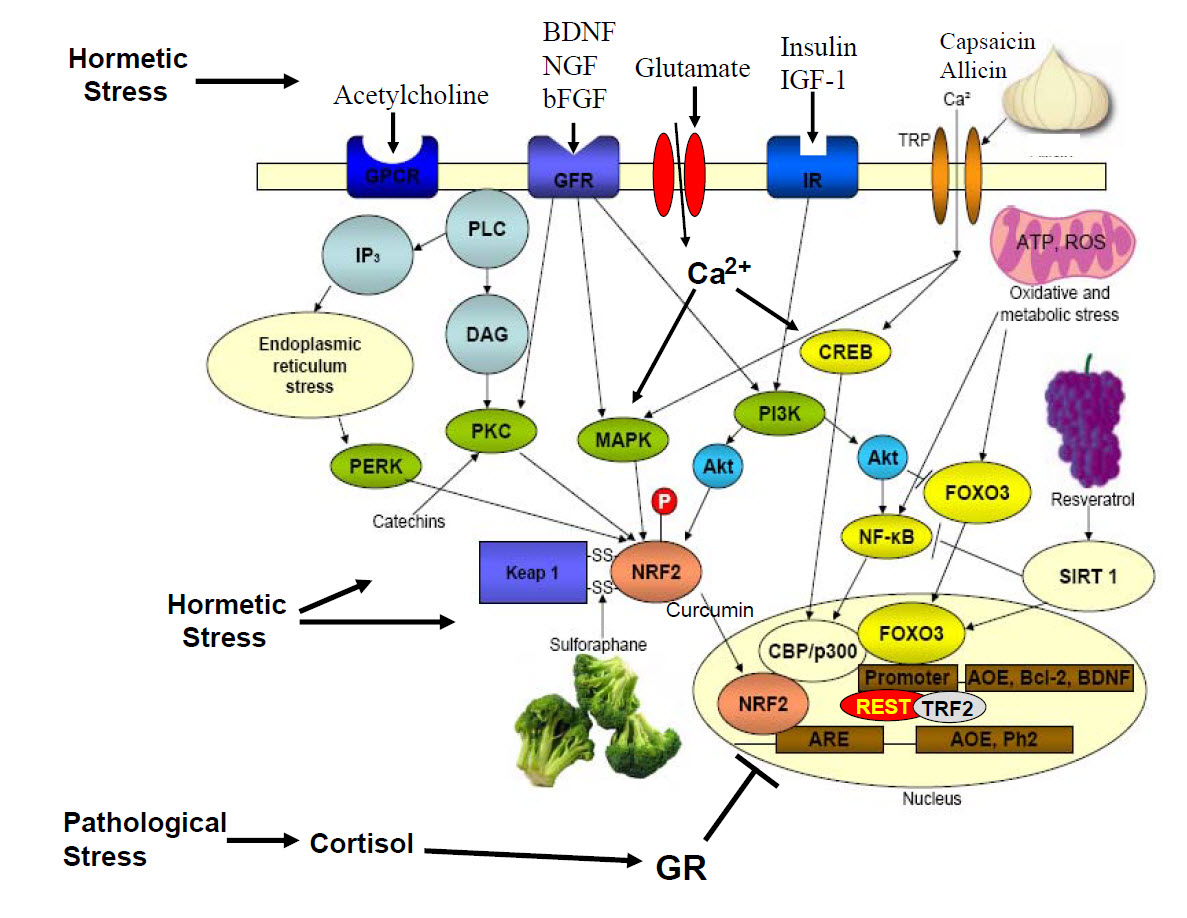
Image source: From the Hormesis project, the National Institute of AgingThe slide shows the impacts of broccoli, garlic, hot peppers, turmeric, grape skins and numerous other phyto-substances on the Nrf2, Sirt1, FOXO3 and NF-kappaB pathways to create positive reactions in the nucleus of cells – hormetic effects Note also that pathological stress and over-expression of the stress hormone cortisol can block the good benefits from happening.
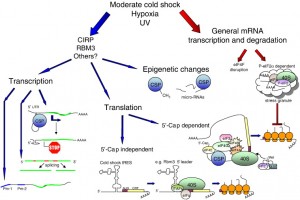
Molecular biology of the cold shock hormetic response
Here are a couple of graphics that illustrate how the cold shock response works. As you can see, the complexity is significant.
The Shock Response Cold-inducible RNA binding Proteins: CIRP & RBM3





Prof. Giuliano,
Would you care to comment on the relative merits between calorie-restriction and intermittent fasting (or endurance and high intensity exercises)? It seems the latter better meet your specification of non-chronic hormetic stress.
Recent study also casts doubt on the effectiveness of CR on longevity.
Pingback: Multifactorial hormesis II – Powerpoint presentation | AGING SCIENCES – Anti-Aging Firewalls
Pingback: Autophagy – the housekeeper in every cell that fights aging | AGING SCIENCES – Anti-Aging Firewalls