By Victor, with contributions by Vince Giuliano
Satellites in Muscle Part I
It is well established that the maintenance of skeletal muscle mass contributes significantly to disease prevention and avoidance of falls and many other problems that degrade quality of life(ref). “Sarcopenia can be defined as the age-related loss of muscle mass, strength and function (Waters, Baumgartner & Garry 2000; Vandervoort & Symons 2001). Although there is no specific level of lean body mass or muscle mass at which one can say sarcopenia is present (Roubenoff 2001), any loss of muscle mass is of importance because there is a strong relationship between muscle mass and strength (Roth, Ferrell & Hurley 2000). Sarcopenia appears to begin in the fourth decade of life and accelerates after the age of approximately 75 years (Waters, Baumgartner & Garry 2000).”(ref) Sarcopenia is a major factor in age-related frailty and susceptibility to fall-related injuries. This blog entry is mainly intended to introduce readers to the major pathways and factors involved in muscle growth and regeneration. In a future Part II blog entry, I intend to discuss therapeutic approaches targeting many of these factors, including a more detailed discussion of myostatin, mTOR, Notch, MAPK signalling, and the role of inflammatory cytokines. There or in a Part III entry, we will discuss whether the processes of sarcopenia can be slowed or avoided, or even reversed. And we will discuss what is known about nutritional exercise, and possible epigenetic interventions for sarcopenia.
In young adults muscle tissue makes up roughly 50% of total body mass. This percentage gradually decreases with age reaching, on average, 25% by age 75, often being replaced by fat tissue.(ref) Muscular function diminishes more rapidly than does muscle mass, indicating an age-related decline in muscle “quality”, as well as mass. This loss of muscle mass and quality occurs in individuals who are physically active and exercise regularly, so it is not merely the result of physical inactivity. Sarcopenia will affect virtually all individuals that reach middle-age. Its debilitating effects rapidly increase after age 70. Not only does the loss of muscle tissue impair mobility and quality of life, it is an important risk factor for many other serious health conditions. Furthermore, therapeutic treatments for sarcopenia have the potential of treating a wide range of other muscle wasting disorders, including muscular dystrophy, AIDS, kidney and heart disease, and cancer. 50% of all cancer patients and 80% of all terminal cancer patients, for example, suffer from muscle wasting, also known as cachexia. The majority of these actually die from complications of the muscle wasting syndrome. Given the vital importance of muscle tissue for physical activity and overall health, understanding the mechanisms involved in the formation, maintenance and regeneration of myogenic tissue has great importance and has been the focus of considerable investigation.
Satellites in Muscle
Muscle tissue has a very remarkable ability to regenerate following damage. This ability was dramatically demonstrated in 1964 when it was shown that entire muscles could regenerate when replaced, after being removed and minced.(ref) What is the reason for such a remarkable regenerative capacity? The key to such regenerative ability is the presence of myogenic stem cells, called “satellite cells”. Satellite cells were first discovered in 1961 by electron microscopic examination of myofibers.(ref) They were named “satellite cells” due to their peripheral location between the sarcolemma and the basal lamina.
Quiescence, Self-renewal, and Differentiation
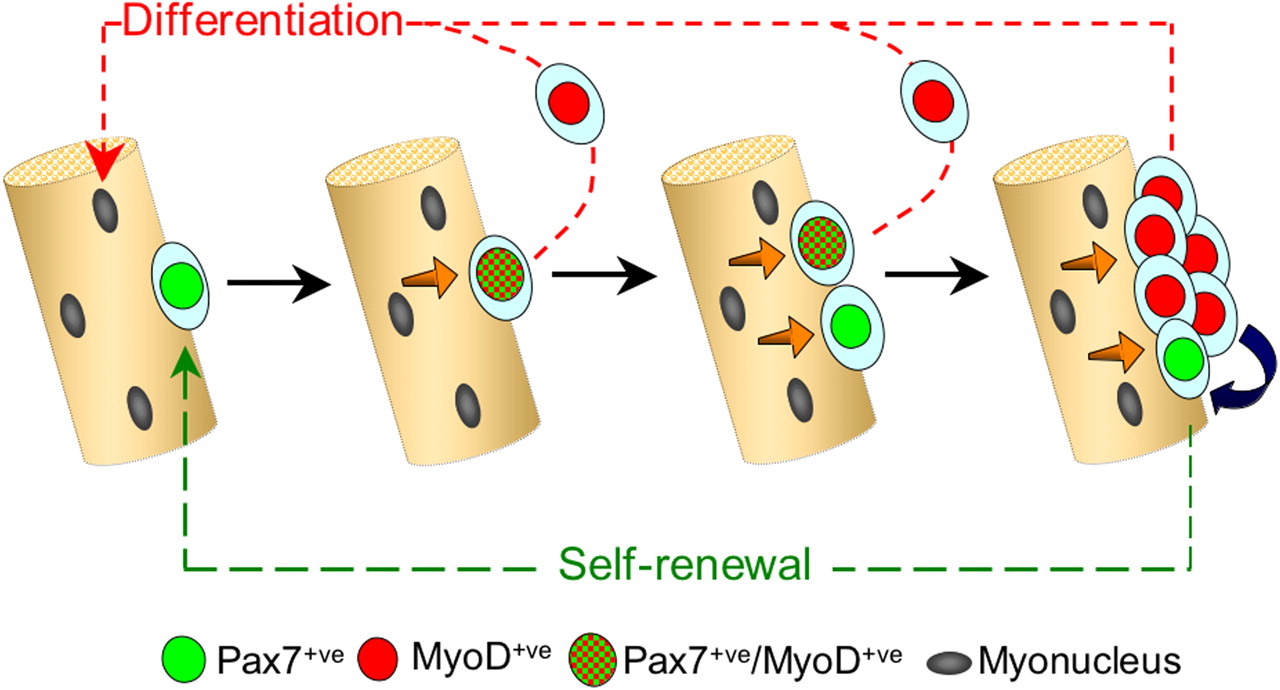
In response to signaling factors in their microenvironment (niche), satellite cells can do one of three different things. They can remain in a state of inactive quiescence until needed. They can proliferate, producing more satellite cells in order to maintain the supply pool; or they can differentiate, becoming muscle tissue precursor cells (myoblasts). Myoblasts proliferate, then fuse together to form myofibers; or as is most often the case, they fuse with existing myofibers increasing the number of nuclei. Interestingly, through a process known as “asymmetric division” satellite cells can divide producing both a daughter satellite cell and a differentiated myogenic precursor cell, in effect combining self-renewal and differentiation in one process.
Nuclei
The nucleus is the control center of a cell, regulating all cellular activities. Myofibers contain multiple nuclei which increase their functional capacity. In order for myofibers to regenerate or grow, more myonuclei are necessary. Supplying the required nuclei is the primary purpose of satellite cells, and accounts for their amazing regenerative capacity. A 2005 study showed that “as few as seven satellite cells associated with one transplanted myofiber can generate over 100 new myofibers containing thousands of myonuclei. Moreover, the transplanted satellite cells vigorously self-renew, expanding in number and repopulating the host muscle with new satellite cells. Following experimental injury, these cells proliferate extensively and regenerate large compact clusters of myofibers.”(ref) These researchers remarkably found that the regenerative ability lost by the destruction of all satellite cells in muscle tissue (by local irradiation) can be restored by grafting just one intact myofiber. The repaired muscle tissue resulting from seven satellite cells in a single myofiber was able to undergo further repetitive damage-repair cycles, illustrating the remarkable ability of a very limited number of satellite cells to both regenerate muscle tissue and to self-replenish in order to maintain future regenerative capacity. Although clinical engraftment of myogenic stem cells is one potential treatment approach, it is unlikely to have much success without the signaling factors necessary to regulate satellite cell activity. Furthermore, given the remarkable ability of existing satellite cells to self-renew, cell transplantation would seem to be unnecessary, once these regulatory mechanisms are effectively targeted.
Just as the number of myonuclei can increase in order to promote growth and repair, it can also decrease through a process known as myonuclear apoptosis leading to muscle atrophy. The loss of myonuclei through apoptosis is thought to play a key role in age-related muscle decline and sarcopenia.(ref) Clinical interventions designed to prevent myonuclear apoptosis are the focus of research designed to increase muslce mass, and improve muscle function. These approaches will be discussed later on.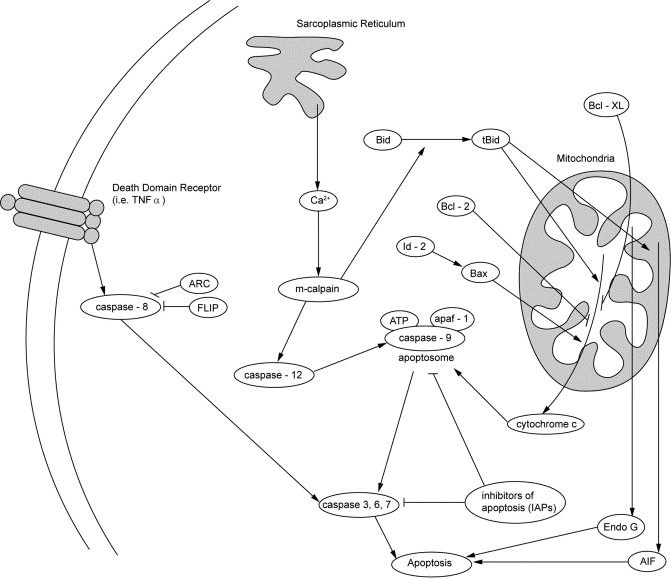
“Fig. 1. Pathways involved in apoptosis in skeletal muscle. Receptor, sarcoplasmic reticulum-, and mitochondrial-mediated pathways may play a role in the induction of apoptosis in skeletal muscle during disuse-induced atrophy and sarcopenia. Caspase-3 activation and subsequent apoptosis occurs through the release of cytochrome-c from the mitochondria in response to changes in the distribution of Bcl-2 family of proteins. Also, dysfunction of the sarcoplasmic reticulum activates caspase-9 and subsequently caspase-3 through calpain-mediated activation of caspase-12. Elevation of TNF-α with age triggers the receptor-mediated pathway, activating caspase-3 through the activation of caspase-8. In addition, caspase-independent pathways, such as the release of AIF and EndoG from the mitochondria may play an important role in disuse-induced apoptosis in muscle from old animals. Inhibitors of apoptosis (IAP, ARC, FLIP) may in skeletal muscle protect the cells from undergoing premature death or nuclear loss.”(ref)
Cardiac Muscle Tissue
Cardiac muscle tissue differs from skeletal muscle tissue in important ways. Unfortunately, cardiac muscle does not typically exhibit the same regenerative capacity as skeletal muscle. Recently, a great deal of research has been done on the use of stem cells to regenerate damaged cardiac tissue. This is currently an intense area of investigation; and has already yielded some very interesting results. Human cells have been reprogrammed to become pluripotent stem cells.(ref) Patches of cardiac tissue have been produced from stem cells.(ref) Various factors have been identified that regulate cardiac stem cell activity.(ref) Compounds have been engineered to effectively alter stem cell activity.(ref) Methods for enhancing stem cells to promote myocardial repair have been developed.(ref) Clearly, science is on the verge of some exciting breakthroughs in the field of myocardial progenitor cell research.
Regulatory Factors
Without a doubt, understanding the roles of the various factors that regulate the supply and fate of satellite cells is of fundamental importance. It was first discovered in 1986 that satellite cells could be activated in response to external signaling factors. Satellite cells were stimulated to divide in response to the presence of mitogen from surrounding crushed muscle tissue.(ref) Soon various “growth factors” were identified that influence satellite cell activity. For example, it was discovered by further in vitro experiments that transforming growth factor beta (TGF-β) prevents satellite cell differentiation, and that fibroblast growth factor (FGF) stimulates proliferation, while IGF1 increases both proliferation and differentiation.(ref) Hepatocyte growth factor (HGF) was found to regulate differentiation as well as proliferation and mobility of satellite cells.(ref)
Satellite cell research changed dramatically with the discovery of specific molecular markers, including two important families of transcription factors, the myogenic regulatory factors (MRFs) which include MyoD, Myf5, myogenin, and MRF4, and the paired box family of transcription factors (Pax1-9). Two important signaling pathways, Notch and Wnt were also found to play crucial roles in regulating satellite cell activity, and make promising targets for clinical interventions.
A Note on Stem Cells
Although they are commonly referred to as “stem cells”, satellite cells are technically more accurately referred to as “tissue-specific” stem cells. This is because, although they possess the properties of self-renewal and differentiation, current evidence indicates that they lack the property of pluripotency, which is the ability to produce cells of many different lineages (tissue types). Although many cell types can be artificially induced to switch lineages, satellite cells appear to be naturally committed to the myogenic lineage.(ref) It is possible that some studies which have reported the ability of satellite cells to produce progeny of other lineages (adipogenic, osteogenic, etc.) may have resulted from contamination of the cultures being used. In any event, such lineage changes are unlikely to occur under natural conditions.
Another persisting point of controversy is whether or not all satellite cells share the innate ability for self-renewal, or whether this ability is limited to a specific subpopulation. A definitive answer to this question is not yet available. Other cell types have been shown capable of producing myoblasts in cell cultures. However, these cells do not appear to be an important source of myoblasts, and do not contribute significantly to muscle regeneration or growth. Under natural, in vivo conditions, other cell types may not ever produce any myoblasts.
Before satellite cells were accepted as the source of the myonuclei necessary for growth and regeneration, the prevailing theory was “dedifferentiation”, which means that differentiated multinuclear cells revert to a precursor mononuclear status in order to facilitate the process of regeneration. Some fish and amphibians appear to use such a process for regeneration; and they are able to regenerate entire limbs when lost or injured.(ref) Under artificial conditions mammalian cells can be induced to dedifferentiate; however, there is no evidence that dedifferentiation is a significant source of mammalian regeneration under natural conditions.(ref) Mammals appear to be entirely dependent upon a viable pool of stem cells for tissue regeneration.
Markers
The role of satellite cells in the generation of myoblasts remained controversial for two decades after their initial discovery. One persistent problem was the dependence on electron microscopy for the identification of satellite cells. Very unique characteristics make them easy to identify based on morphology. Since quiescent cells have very low energy demands, satellite cells have very few mitochondria and practically no cytoplasm. In effect, they consist of a nucleus, a cell membrane, and the receptors necessary to respond appropriately to extracellular signals. In the 1990s molecular markers were discovered, which greatly facilitated the identification of satellite cells. Of these, Pax7 appears to be the most important for maintaining satellite cell supply, and is the most useful for identifying satellite cells, although its paralog, Pax3, is also expressed by satellite cells.
Paired Homeobox Genes (Pax3/7)
Pax7 and Pax3 appear to have both distinct and overlapping functions in maintaining satellite cell homeostasis and contributing to muscle growth and regeneration.(ref) Both are upstream regulators of the Muscle Regulatory Factors (MRFs), another family of muscle regulatory proteins. A great deal has been learned about the function of these muscle regulators by studying genetically altered mutant mice. In mice lacking Pax7, satellite cell numbers quickly diminish after birth. After fetal development, myogenesis ceases to take place. Muscle weakness along with smaller muscle fibers containing fewer nuclei are observed. Very little regeneration takes place after injury. Retarded growth usually results in death within weeks.(ref) It is interesting to note that Pax7 is not necessary for satellite cell function or myogenesis during the fetal period, only during the postnatal period. This illustrates the fact that regulatory factors play distinct roles during different developmental stages.
Similarly, the genetic ablation of Pax7 expressing cells in adult mice, either locally or systematically, by various methods results in muscle incapable of regeneration. Moreover, such ablation destroys any preexisting satellite cells, which require Pax7 for their survival. Other types of stem cells do not express Pax7 and so would be spared the effects of ablation; but they fail to produce muscle regeneration, underscoring the necessity of satellite cells for muscle repair and growth. Furthermore, myonuclei do not express Pax7, so they would be immune from the effects of ablation; yet the complete absence of regeneration indicates that myonuclear dedifferentiation does not occur under normal conditions. “In summary, these recent studies on the depletion or genetic ablation of satellite cells using complementary approaches clearly demonstrate that satellite cells are responsible for skeletal muscle regeneration after acute injury. Under such conditions, non-satellite cell populations are unable to substitute for the function of satellite cells, which are indispensable for muscle regeneration. The cell on the edge has now returned centre stage!” From Satellite cells are essential for skeletal muscle regeneration.
Muscle Regulatory Factors
Pax7 is a vitally important factor for maintaining satellite cells, and is a very useful marker for identifying them. However, in order to form muscle cells, satellite cells must become activated. What are the factors invovled in the process of activation and differentiation of precursor cells, leading to muscle cell formation? In 1987 genetic studies identified a factor, called MyoD, which had the ability to transform cells from other lineages into muscle cells.(ref) In the following years, other myogenic factors were identified, including Myf5, myogenin, and MURF4. They are collectively referred to as “Myogenic Regulatory Factors” (MRFs). MRFs are only expresssed in activated satellite cells and are associated with differentiation, and myoblast formation and fusion. Actually, some satellite cells appear to express Myf5, but these are a subpopulation already committed to differentiation, and incapable of self-renewal.(ref) There is no evidence that any MRFs are expressed in quiescent satellite cells. Remarkably, MyoD is such a potent regulatory factor that although it is normally expressed in cells of myogenic lineage, when activated in cells of other types, it can induce the differentiation of cells from other lineages into myoblasts. MyoD and Myf5 appear to have redundant functions, and act upstream of myogenin and MURF4. Various knockout and knockin studies have shown that MyoD and Myf5 specify terminal myoblast commitment and promote proliferation. They have overlapping functions and are called “redundant”, because the absence of one factor is, to some extent, compensated for by the other one. Once activated, myogenin and MURF4 play important roles in the differentiation process, and regulate the expression of genes involved in the processes of myocyte and myotube formation. One of the challenges to the therapeutic use of stem cells for muscle regeneration is the fact that satellite cells cannot be isolated and grown in vitro, because they immediately differentiate into myoblasts due to the spontaneous activation of the MRFs.(ref) MRFs are absolutely essential for muscle regeneration. Knockout mice lacking these factors have a complete loss of skeletal muscle formation.(ref)
“Model of satellite cell self-renewal. Quiescent satellite cells (green) activate to coexpress Pax7 and MyoD (green and red tartan), and then most proliferate, down-regulate Pax7, maintain MyoD (red), and differentiate (red pathway). However, activated Pax7+ve/MyoD+ve (green and red tartan) satellite cells can also divide to give rise to cells that adopt a different fate. These give rise to clusters of cells containing both Pax7−ve/MyoD+ve (red) progeny, whereas others down-regulate MyoD expression and cycle while maintaining only Pax7 (green). These clusters may grow by the further generation of cells with divergent fates. Pax7+ve/MyoD−ve cells (green) become quiescent, thus renewing the satellite cell pool (green pathway), whereas the MyoD+ve cells (red) differentiate to produce myonuclei (red pathway). Signaling from the myofiber (orange arrows) and/or between cells within the clusters (blue arrow) may dictate which fate the satellite cell adopts.” (ref)
Myogenin Regulates Exercise Capacity
In addition to its role in muscle development, recent research has revealed surprising new functions for myogenin. These new studies also cast serious doubt on the importance of myogenin for adult muscle maintenance and regeneration. Below is a summary of some of the results. For more informtion, please see:
Myogenin Regulates Exercise Capacity and Skeletal Muscle Metabolism
Myogenin Regulates Exercise Capacity but Is Dispensable for Skeletal Muscle Regeneration
- Myog deletion enhances exercise capacity.
- Myog is not required for adult survival or muscle regeneration.
- Myog deletion improves energy metabolism and increase lactate threshold.
Notch Signaling
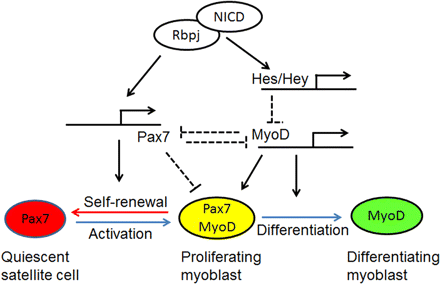
Notch signaling is a critical regulator of development and regeneration. This colorful name has an interesting origin. In 1914, a mutant fly with notched wings was described. In the 1980s the gene was cloned and found to encode a transmembrane receptor that mediates intercellular communication. When ligand proteins from adjacent cells bind to the extracellular domain, the intracellular domain is released and translocates to the nucleus where in concert with other transcription factors and coregulators the expression of specific genes is activated. On the one hand, Constitutive Notch Activation Upregulates Pax7 and Promotes the Self-Renewal of Skeletal Muscle Satellite Cells. On the other hand, a primary effect of Notch signalling is to prevent the differentiation of satellite cells. Why is this important for development and regeneration? Although differentiation of statellite cells is necessary for growth and repair, premature differentiation can rapidly deplete the satellite cell pool. The timing of each step in the process is strictly regulated in order to maintain satellite cell homeostasis. By preventing premature differentiation, Notch enables satellite cells and myogenic precursor cells to proliferate, ensuring and adequate supply before differentiation into myotubes for growth and tissue repair.
An earlier discussion of Notch signaling and the possibility of enhancing Notch signaling to help avert various disease conditions was offered in the October 2009 blog entry Niche, Notch and Nudge. Also, that blog entry points out how Notch signaling is also an important mechanism of cancer progression since it may enhance the survival and growth of cancer stem cells. And it discusses the role of MAPK in Notch signaling.
The precise mechanisms by which Notch prevents differentiation are still being investigated. However, research clearly shows that multiple redundant pathways are involved.(ref) Notch activity interacts with a variety of transcription factors including MRFs, PAX7 and FOXO.(ref) The genes targeted by Notch include the HES and HEY gene families. These genes encode proteins, which act as repressors for other transcription factors necessary for differentiatiion. Notch activity can be inhibited by various means. Numb is an antagonist that binds to the intracellular domain and prevents it from translocating to the nucleus. Numb also plays a role in regulating asymmetric division. Differentiated daughter cells express Numb, while non-differentiated daughter cells do not.(ref) In experimental studies, Notch inhibition through various methods invariably leads to uncontrolled differentiation of progenitor cells resulting in their depletion and in defective muscle regeneration. In contrast, Notch activation results in enhanced proliferation and renewal of progenitor cells.(ref)
“Model for Notch’s regulation of stem cell fate in muscle. Quiescent satellite cells (stem cells) express high levels of Pax7. The activated satellite cells (myoblast) coexpress Pax7 and MyoD and proliferate. The proliferating myoblast can either downregulate Pax7 to differentiate or downregulate MyoD to self-renew. Activated Notch (NICD) binds to RBP-Jκ to form a transcriptional activation complex that upregulates the transcription of Pax7 as well as canonical Notch targets (Hes and Hey family genes). Hes/Hey proteins inhibit MyoD gene transcription. Therefore, Notch activation upregulates Pax7 to promote satellite cell self-renewal, while inhibiting MyoD to block myogenic differentiation. Pax7 upregulation and MyoD downregulation may together lead to cell cycle withdrawal. The reciprocal inhibitory action between Pax7 and MyoD further amplifies the effect of Notch signaling.”(ref)
Since pools of myogenic precursor cells are known to decline with age, many researchers have speculated that this attrition is attributable to age-related decline in Notch activity, leading to the compromised regenerative ability of aged muscles. If so, then Notch activation would make a very promising target for age-related muscle deterioration. Since Notch plays a similar role in many other tissue types, including liver and brain tissues, targeting Notch activity would make a promising target, not just for preventing sarcopenia, but also for treating age-related declines in the function of many diverse organs and tissues. Several animal models have demonstrated that Notch inhibition results in impaired regeneration of young muscles and that forced Notch activation restores youthful regenerative capacity to aged muscles.(ref) Researchers studying liver function found that heterochronic parabiosis (This means that young and old animals share a common circulatory system.) was able to restore Notch activity as well as the proliferation and regenerative capacity of satellite cells. This study indicates that Notch activity responds to systemic factors in the circulatory system that change with age. See Systemic signals regulate ageing and rejuvenation of blood stem cell niches
In a 2009 study, Molecular aging and rejuvenation of human muscle stem cells, the results from these animals models were confirmed and expanded in human muscle tissue. It was found that both declines in Notch activity and increases in levels of TGFbeta prevent satellite cell activation and proliferation. (Myostatin is a member of the TGFbeta family and will be discussed later in greater detail.) This study also found that MAPK signalling plays an important role in Notch activation. The researchers conclude by saying, “This work uncovers the molecular culprits responsible for the lack of tissue maintenance and repair seen in old humans, and demonstrates that, as seen in mice, old human muscle stem cells are actually capable of productive regeneration, but are inhibited by their own muscle to do so. However, particular molecular cues or exposure to young human serum restores ‘youthful’ responses to muscle stem cells isolated from 70-year-old humans, rendering them similar to cells from 20-year olds. Interestingly, young human muscle stem cells are ‘aged’ instantly, by the aged tissues, and thus are unlikely to work upon transplantation into the old.” Another cause for decreased Notch activity with age is decreased levels of testosterone. Testosterone is known to increase Notch signaling.(ref) Animal studies have shown that testosterone supplementation is able to restore youthful Notch activity.(ref, ref)
Wnt Signaling
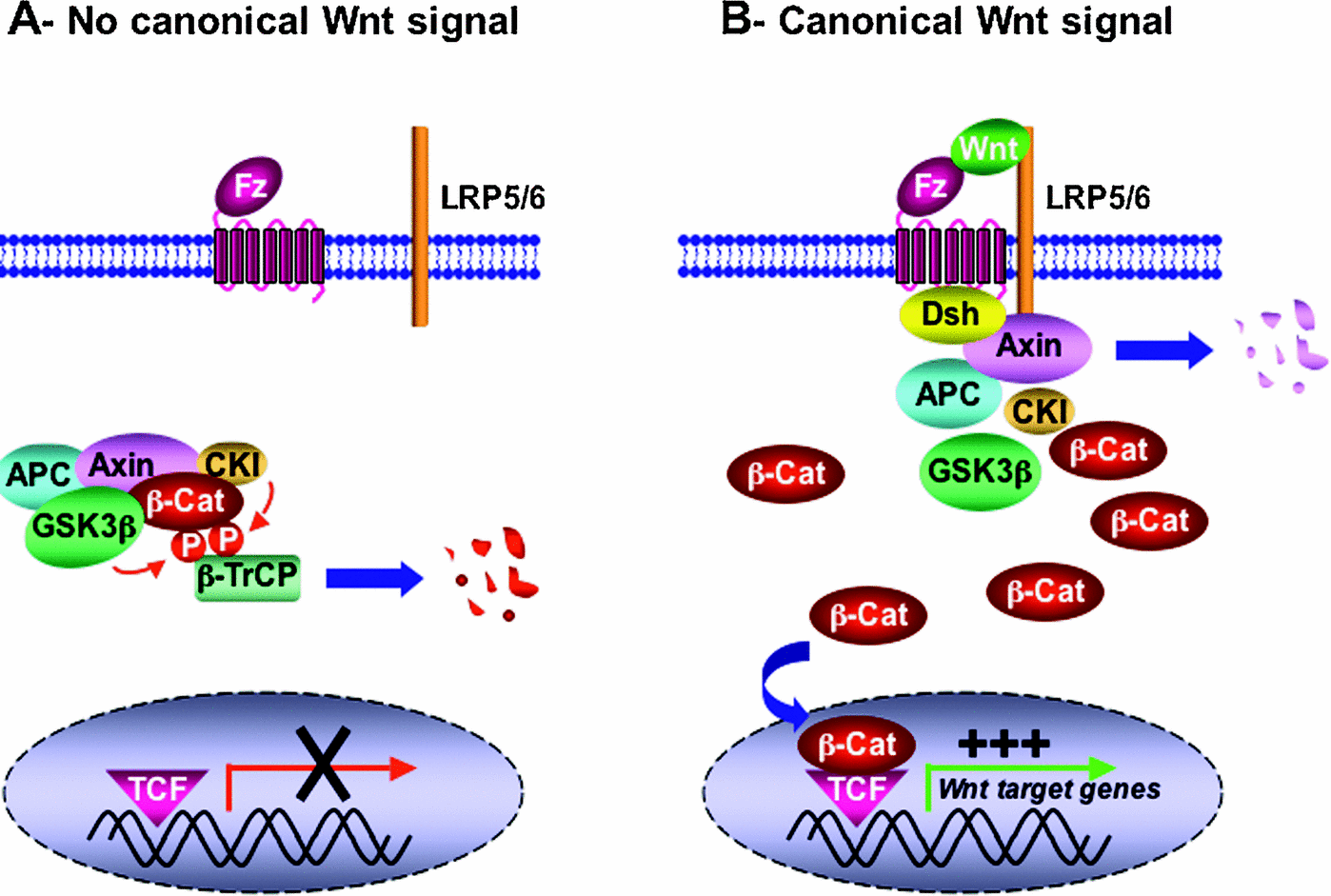
After adequate proliferation of precursor cells, the next step in the process of tissue regeneration is differentiation. Just as Notch is responsible for regulating the proliferation stage, Wnt signaling promotes differentiation.(ref) Wnt proteins are secreted signaling molecules that act through canonical and noncanonical pathways. The canonical pathway involves stabilization and nuclear translocation of beta-catenin.(ref) Interestingly, beta-catenin appears to act through multiple channels promoting differentiation (ref) and at other times self-renewal.(ref) The temporal balance between Notch and Wnt must be tightly regulated. Premature Wnt activation results in premature differentiation, while Wnt inhibition prevents differentiation. Crosstalk between these two myogenic regulators is mediated by GSK3beta, which inhibits Wnt signaling. As GSK3beta declines beta-catenin levels rise and differentiation takes place.(ref) Evidence indicates that muscle injury upregulates Wnt signaling and that it is necessary for muscle growth.(ref)
“Figure 4 Simplified overview of the canonical Wnt signalling pathway (adapted from He et al., 2003) (A) In the absence of Wnt ligand, b-catenin is sequestered in a multiprotein degradation complex containing the scaffold protein Axin, the tumour suppressor gene product APC, as well as the kinases CKI and GSK3b, among others. Upon sequential phosphorylation, b-catenin is ubiquitinated by the b-TrCP–E3-ligase complex and subsequently degraded by the proteasome machinery. There is no transcription of Wnt target genes. (B) Wnt ligand associates with Fz and LRP5/6 co-receptors. This in turn can lead to translocation of Axin (and perhaps the whole multiprotein complex) to the plasma membrane through direct interaction with LRP5/6 and Dsh/Fz. Translocation results in Axin degradation and/or dissociation of the multiprotein complex. GSK3b also might be displaced from this complex through Dsh action. b-catenin is then released from the multiprotein complex, accumulates in the cytoplasm in a non-phosphorylated form, and subsequently translocates into the nucleus where by association with TCF/LEF factors it promotes transcription of Wnt target genes.(ref)”
IGF1
Insulin-like Growth Factor-1 is a peptide hormone that binds to a transmembrane receptor found in satellite cells, muscle cells, and cells of many other tissue types. IGF1 is thought to induce muscle hypertrophy, by distinct mechanisms. The IGF1 receptor is a tyrosine-kinase receptor which induces cellular signal transduction chains by adding phosphate groups or “phosphorylating” specific proteins within the cell. Activation of the PI3K/AKT kinases cause phosphorylation of the FOXO transcription factors, which prevents them from entering the nucleus and promoting the expression of atrophic factors, like MuRF1. The AKT pathway (often called “PKB” instead of “AKT”) also inhibits the secretion of myostatin, thereby increasing both muscle cell differentiation, and protein synthesis.(ref) Myostatin inhibition results in a positive feedback cycle, since myostatin also inhibits the AKT pathway.(ref, ref) IGF1 also activates the mTOR pathway, which is well-known to play a central role in muscle growth. Apparently, PI3K activates mTOR by moving tuberous sclerosis complexes (mTOR inhibitors) from the membrane to the cytosol.(ref) (Independent of growth factors, amino acid availability, especially leucine, regulates mTOR activity, ref.) For a more detailed discussion of the AKT pathway, see: Akt: a nexus of growth factor and cytokine signaling in determining muscle mass For an overview of transcriptional regulation of muscle growth/atrophy pathways,see: Anabolic and catabolic pathways regulating skeletal muscle mass. However as pointed out below, more-recent research suggests the conventional wisdom may not be correct and that IGF1 pathway is not actually involved in exercise-driven muscle development in adults; it is involved only during the original muscle-development stages of the organism and in the repair of injuries.
Until recently the exact roles of these pathways and their relationships to IGF1, the effects of resistance exercise (mechanical load/stretch), and developmental stage have remained mysterious. However, current research involving transgenic models is quickly unraveling these mysteries.
Mechanical Stimuli Activate mTOR Independent of IGF1.
It was observed that mechanical stimulation induced local expression of IGF1 and other growth factors.(ref) These were accompanied by an increase in kinase phosphorylation signaling, and muscle growth. It was logical to conclude that IGF1 activated the pathways responsible for muscle growth. Subsequent research has cast serious doubts on this conventional theory. In 2004, it was shown that mechanical stimulation activates mTOR growth pathways, completely independent of IGF1 and the PI3K/AKT pathway. Pharmacologically inhibiting PI3K did not alter activation of mTOR. These results were confirmed with AKT-knockout mice (which lack the AKT gene).
From Mechanical stimuli regulate rapamycin-sensitive signalling by a phosphoinositide 3-kinase-, protein kinase B- and growth factor-independent mechanism. “These surprising results indicate that mechanical stimuli are different from insulin-like growth factors in that mTOR-dependent signalling events are regulated via a PI3K/Akt1-independent mechanism. Furthermore, these results indicate that if mechanical stimuli regulate protein synthesis by the release of locally acting factors, then these factors must activate mTOR through a PI3K/ Akt1-independent mechanism. However, in both the co-incubation and conditioned-media experiments, the release of locally acting factors was not sufficient for the activation of mTOR-dependent signalling events, thus suggesting that mechanotransduction (e.g. mechanoreceptor) rather than ligand binding of autocrine/paracrine growth factors as the cause for the induction of the mTOR-dependent signalling events.”
These results were confirmed by a 2009 study, The role of PI3K in the regulation of mTOR following eccentric contractions: “In summary, the results from this study indicate that resistance exercise contractions, such as ECs (eccentric contractions), activate mTOR through a PI3K–AKT-independent mechanism.”
In 2007, another transgenic study using mice with a negative IGF1 receptor (one that binds IGF1, but doesn’t transduce signals) showed that the hypertrophic effects of mechanical load were NOT mediated by IGF1.(ref) “We demonstrate that IGF-I receptor-mediated signalling is not necessary for the induction of skeletal muscle hypertrophy in adult mice following a chronic increase in mechanical loading.”
The results of these studies have been further confirmed by a new transgenic study, in which researchers conclude, “Acute resistance exercise did not increase either IGF-1 receptor phosphorylation. . . [Furthermore] these data suggest that physiological loading does not lead to the enhanced activation of the PI3K/Akt/mTORC1 axis and that PI3K activation levels play no significant role in adult skeletal muscle growth.”(ref)
A number of studies have been done over the years to measure the purported anabolic effects of IGF1 in the elderly. To my knowledge, none have showed any significant effect. As an example, one study used GH to increase IGF1 in the elderly. It found that exercise had an anabolic benefit, but the addition of GH to increase IGF1 produced no additional benefit. See: Effect of growth hormone and resistance exercise on muscle growth and strength in older men
mTOR Causes Adult Muscle Growth, Not IGF1
Additional studies have confirmed that mTOR plays a central role in muscle growth; but they also confirm that this happens independent of the PI3K/AKT pathway.
A PI3K-independent Activation of mTOR Signaling Is Sufficient to Induce Skeletal Muscle Hypertrophy “In this study, we demonstrate that the overexpression of Rheb induces mTOR signaling through a PI3K/PKB-independent mechanism and that this event is sufficient to induce a robust and cell autonomous hypertrophic response. Furthermore, it was determined that the hypertrophic effects of Rheb occurred through a rapamycin-sensitive mechanism, that mTOR was the rapamycin-sensitive element in skeletal muscle that conferred the hypertrophic response, and that the kinase activity of mTOR was necessary for this event. Combined, these results strongly indicate that a PI3K/PKB-independent activation of mTOR signaling, in skeletal muscle, is sufficient to induce hypertrophy.” The researchers conclude by suggesting that muscle hypertrophy could be induced by the use of mTOR agonists.
IGF1 Only Increases Muscle Growth During Developmental Stages
It is clear from experimental data that the proliferative role of IGF1 is limited to developmental growth and to regenerative repair. IGF1 is necessary for proper development and repair following injury. Young, developing mammals not only need IGF1 for proper development, but overexpression leads to increased growth. The same does not happen in adults overexpressing IGF1. From a transgenic study published in 2010: “In conclusion, these data show that adult non-growing skeletal muscles are refractory to hypertrophy in response to the elevated IGF-1. By contrast, growing muscles respond by activating signalling downstream from the IGF-1 receptor (demonstrated by phosphorylation of Akt, p70S6K) to increase protein accretion by the myofibres. Thus, the IGF-1-mediated hypertrophy evident in adult transgenic muscles results from enhanced increase in muscle mass mainly during the postnatal growth phase. . . These data demonstrate that elevated IGF-1 has a hypertrophic effect on skeletal muscle only in growth situations.” (ref)
See, also: Regulation of skeletal muscle growth by the IGF1-Akt/PKB pathway.
Since IGF1 levels decline with age, many have thought that this decline is also responsible for the age-related decline in muscle mass. However, we now know that this is not the case. While IGF1 plays an important role in developmental muscle growth, it is not involved in adult muscle growth. This also explains why IGF1 treatments have uniformly failed to prevent muscle loss in the elderly. And we note that IGF1 expression is, if anything, negatively correlated with mammalian longevity(ref).
Myostatin
Myostatin (MSTN), also known as GDF8 (growth differentiation factor 8), is a potent negative regulator of muscle growth, which belongs to the TGF-beta superfamily. MSTN is produced in skeletal muscle tissue and circulates in the blood stream. It binds to transmembrane receptors of activin type 2. Receptor binding activates (phosphorylates) the Smad 2/3 transcription factors, which translocate to the nucleus in order to modulate the expression of genes, including the MRFs.(ref) MSTN prevents myogenesis by inhibiting transcription of the MRFs, including MyoD, which, as previously discussed, are necessary for satellite cell activation, proliferation and differentiation.(ref) In addition to its central role as a negative regulator of myogenesis, myostatin prevents muscle growth by both decreasing protein synthesis and increasing protein degradation. MSTN increases protein degradation by upregulating the ubiquitin-proteasome pathway; and it decreases new protein synthesis by inhibiting the mTOR pathway.(ref, ref).
We also note that mTOR inhibition is one of the few well-documented approaches to increasing mammalian longevity(ref). This leads one to wonder if frequent exercise-induced mTOR activation might work against longevity, while at the same time contributing to muscle regeneration. All available evidence indicates that this is not the case. Regular exercise is associated with both increased longevity and improved muscle maintenance, probably because exercise activates mTOR very selectively, only in specific muscle tissues; it does not activate it globally. On the other hand, global mTOR inhibition is very likely to have adverse effects on muscle mass.
MSTN activity appears to be one important factor in the pathogenesis of sarcopenia. In order to investigate this, researchers have measured blood levels of myostatin in young and elderly sarcopenic men.(ref) Even though serum levels of myostatin do not appear to change much with age, when directly measured in muscle and bone tissue in rodents myostatin levels were found to increase very dramatically with age. The increased myostatin expression was accompanied by reduced proliferative capacity of bone and muscle progenitor cells.(ref) In a human study, post-exercise muscle biopsies revealed a 2-fold increase in myostatin and mRNA in the older subjects compared to young men. Stem cell myostatin levels between the two groups were identical at baseline, however at 24hr there was a 67% increase in myostatin-positive stem cells in the older group.(ref) This indicates that impaired myogenesis may result from a greater degree of myostatin colocalization with stem cells in aged individuals, which may not be reflected in serum levels.
The function of MSTN has been investigated with genetic studies. Mice with the MSTN gene “knocked out” have widespread increases in skeletal muscle mass, approximately twice the mass of control animals, as a result of both increased fiber number and fiber size. Similar results have been found in cattle, sheep and dogs.(ref, ref, ref)
Genetic mutations affecting MSTN are rare in humans, but when they occur a similar change in phenotype is observed. A German boy was recently found to be homozygous for an MSTN mutation, making him much stronger and more muscular than his peers. His mother has a mutation in one copy of the gene; and presumably his father does as well, though the father’s identity remains undisclosed.(ref) An American boy, Liam Hoekstra, has a similar phenotype resulting from a defect in the MSTN receptor. He appeared on the television show World’s Strongest Toddler.(ref)
Even though the role of MSTN in age-related muscle wasting is still not entirely clear, it makes a very promising target for therapeutic treatments. Both genetic treatments and pharmocological compounds designed to block MSTN have shown great promise in a variety of animals models, including primates. These will be discussed in greater detail in a follow-up blog entry.
Closely related topics yet to be covered
Discussion of interventions to avert, postpone or mitigate sarcopenia will be in the Part II blog entry. At this point, it is worth mentioning that there is a substantial body of publications related to nutritional interventions, and another substantial body of publications related to chronic and resistance exercising as an intervention. Also, we plan to explore the epigenetic pathways associated with sarcopenia. Just as a sampler, the 2012 publication Resistance Exercise Reverses Aging in Human Skeletal Muscle reports “Multiple lines of evidence suggest that mitochondrial dysfunction is a major contributor to sarcopenia. We evaluated whether healthy aging was associated with a transcriptional profile reflecting mitochondrial impairment and whether resistance exercise could reverse this signature to that approximating a younger physiological age. Skeletal muscle biopsies from healthy older (N = 25) and younger (N = 26) adult men and women were compared using gene expression profiling, and a subset of these were related to measurements of muscle strength. 14 of the older adults had muscle samples taken before and after a six-month resistance exercise-training program. Before exercise training, older adults were 59% weaker than younger, but after six months of training in older adults, strength improved significantly (P<0.001) such that they were only 38% lower than young adults. As a consequence of age, we found 596 genes differentially expressed using a false discovery rate cut-off of 5%. Prior to the exercise training, the transcriptome profile showed a dramatic enrichment of genes associated with mitochondrial function with age. However, following exercise training the transcriptional signature of aging was markedly reversed back to that of younger levels for most genes that were affected by both age and exercise. We conclude that healthy older adults show evidence of mitochondrial impairment and muscle weakness, but that this can be partially reversed at the phenotypic level, and substantially reversed at the transcriptome level, following six months of resistance exercise training.”


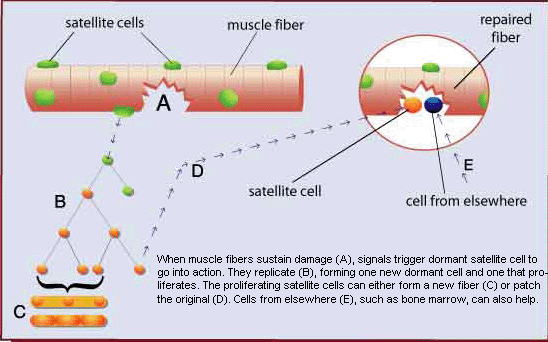
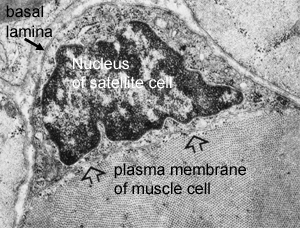


Excellent review of sarcopenia.
In the past a lack of dietary protein has sometimes been alluded to as part of the problem. In my opinion this is not supported by evidence. Alternate Day Feeding animals seem to thrive. Ketogenic diets do not in and of themselves induce sarcopenia.
What are the optimal protein intakes and timing of feeding? This is a question that in my opinion should be addressed and experimental observations made to gain insight into diet and health as we pursue healthy longer life.
Eric
I wonder if the cool hand glove developed at Stanford would help? Seems to be good for enhancing athletic training and performance.
http://news.stanford.edu/news/2012/august/cooling-glove-research-082912.html
It is also out of research and available to enhance workouts or performance for High intensity training.
The typo “differentiatiion” was located towards the middle of this page.
Where is part 2? 🙁