By James P Watson
I believe the the message of the previous blog entry Health through stressing fruits and vegetables – the Xenohormetic Live Food Hypothesis by Vince and Melody is worthy of acclaim. As so frequently happens, it fueled my “why gene mutation” and set me thinking and further researching. Following are some observations that expand on the actions and mechanisms of plant polyphenols such as are in fruits and vegetables. I also cite research on additional stressors not covered in the original blog entry that can affect fruits and vegetables including physical wounding, ethylene gas, root hypoxia, osmotic stress, cold stress, drought and pathogen attack. I believe these can apply both pre and post harvesting. I discuss how polyphenols are involved in causing leaves to become colored and die in the Fall – a form of apoptosis. I also mention some personal reactions – the interest generated in me by some of these points.
A third blog entry Further extensions to and implications of the Xenohormetic live food hypothesis goes on to revisit xenohormesis in a little more theoretical depth and covers additional topics such as pre-harvest plant stressing, and how pre or post-harvest stressing may be used to induce additional benefits such as crop mold resistance and reduced food spoilage. It cites a number of other findings from the literature related to stressing plants. And it describes a simple and practical approach that ordinary consumers could use to further preserve and enhance nutritional values of fruits and vegetables while they sit in the refrigerator.
1. Polyphenol Synergism: 1 + 1 = more than 2
Plant polyphenols have synergistic effects, a fact worth emphasizing. There are many practical implications of this. The most important is that for health we need multiple polyphenols and not just one. This is why a variety of fruits and vegetables are important. Here is some evidence for synergism:
a. Flavonoid mixtures are more effective than single flavonoids in their antibacterial effects. For instance:
- quercetin + quercitrin are better than either flavonoid alone
- quercetin + morin are better than either flavonoid alone
- quercetin + rutin are much better than either flavonoid alone
- Rutin alone has NO antibacterial effect, but when combined with quercetin and morin, there is a greater antibacterial effect.
What does this synergy mean? My inference with respect to antibacterial action is that quorum sensing inhibition by polyphenols is synergistic. (i.e. 1 + 1 is greater than 2). See the recent blog entries Quorum sensing Part 1: quorum sensing inhibition via phytochemicals – a new approach against infectious diseases and Quorum Sensing Part 2 – Intra and inter-species molecular communications
Reference: Rutin-enhanced antibacterial activities of flavonoids against Bacillus cereus and Salmonella enteritidis.
b. Flavonoid mixtures are more effective than single flavonols in their anticancer effects.
For instance, quercetin + kaempferol reduces the risk of cancer more than the predicted additive effects of the two polyphenols in in vitro testing of GI cancer cells and breast cancer cells.
2. Wounding lettuce induces polyphenol synthesis: further evidence that polyphenols are stress hormones
This is right in line with what was said in the previous blog with respect to garlic. The publication Effect of Intensity and Duration of Heat-shock Treatments on Wound-induced Phenolic Metabolism in Iceberg Lettuce seemed mainly focused on going over the effects of extreme heat on lettuce, but what interested me was that just simply excising the midrib section of lettuce leaves induces polyphenol synthesis. As expected, too much heat stopped this from occurring. (i.e. greater than 40 C). From 20-40 C, the increase in polyphenol synthesis from wounding was maintained, but above 40 C, the amount of polyphenol synthesis from wounding dramatically was reduced. Again, this suggests that polyphenol synthesis is a hermetic stress response for lettuce, the stress in this case being wounding via the removal of lettuce leaves.
3. Ethylene is the plant hormone that induces polyphenol synthesis in response to wounding
The ancient Egyptians used to gash figs in order to stimulate ripening. They had figured out that this wounding process would make them ripen faster. The Chinese would burn incense in closed rooms to enhance the ripening of pears. In 1901, a Russian scientist named Dimitry Neljubow showed that the compound responsible for this ripening was ethylene. However, it was not known until 1934 that plants actively synthesized ethylene and that this was not just a byproduct of plant metabolism (this sounds much like the free radical story, nitric oxide story,carbon monoxide story, and hydrogen sulfide story). Of interest is the fact that the ethylene is produced by plants in very small amounts (I do not know exactly how much, but I am sure it is much like ROS, RNS, CO, H2S, etc.)
References:
- Neljubov D. (1901). “Uber die horizontale Nutation der Stengel von Pisum sativum und einiger anderen Pflanzen”. Beih Bot Zentralbl 10: 128–139.
- Doubt, Sarah L. (1917). “The Response of Plants to Illuminating Gas”. Botanical Gazette 63 (3): 209–224.
- Gane R. (1934). “Production of ethylene by some fruits”. Nature
- Yang, S. F., and Hoffman N. E. (1984). “Ethylene biosynthesis and its regulation in higher plants“.Ann. Rev. Plant Physiol. 35: 155–89
Vince told me an interesting story related to banana boats. Bananas release ethylene gas while ripening, and the gas itself promotes ripening. There was a time when entire boatloads of bananas being shipped from Central America arrived in New York overripe or mushy due to the ethylene So, refrigeration was introduced early in banana boats and. Also, maintainance of fresh air circulation in banana boats is now recognized as important. Control of ethylene is what makes possible our access to bananas. “Without refrigerated transport, shiploads of fruit frequently arrived at best overripened, at worst in a downright rotten state, making the mass marketing of bananas impossible. With the preservative power of refrigeration and the speed of steam-powered engines, however, bananas could be shipped in enormous volumes. […] In a matter of decades, refrigerated vessels had helped turn what in the 1890s was an exotic curiosity into a mass-market product, paving the way for a massive and highly lucrative trans-American trade(from Moveable Feasts).”
Ethylene production can also significantly reduce the shelf life of fresh flowers, potted plants and many fruits and vegetables. This plant hormone has many effects and appears to play a major role in maturation.
4. Many Environmental stressors induce ethylene synthesis by plants
Once I found out that ethylene was actively synthesized by plants, that ethylene synthesis was stimulated byplant wounding, and that ethylene induced plant polyphenol synthesis, I thought I had everything figured out, but this just raised more questions that it answered. Specifically, most plants are not wounded. Why would a pathway for ethylene synthesis occur in nature? There had to be more than just a “wounding response” that would induce selection pressure for such a biochemical pathway to evolve. Well, here is what I found out:
Many environmental cues induce ethylene synthesis in plants:
- flooding
- drought
- chilling
- wounding
- pathogen attack
reference: Wikipedia article on ethylene (the page did not list the reference)
Now this is getting interesting! What induces ethylene synthesis? What is the gene activation pathway? Here is the answer, again from Wikipedia:
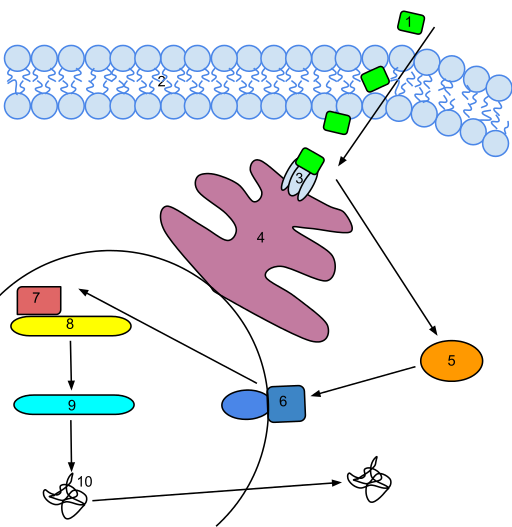
“An ethylene signal transduction pathway. Ethylene permeates the membrane and binds to a receptor on the endoplasmic reticulum. The receptor releases the repressed EIN2. This then activates a signal transduction pathway which activates a regulatory genes that eventually trigger an Ethylene response. The activated DNA is transcribed into mRNA which is then translated into a functional enzyme that is used for ethylene biosynthesis(ref)”
5. Plants respond to cold stress as well as heat stress by upgrading polyphenol production
The 2000 publication Resistance to cold and heat stress: accumulation of phenolic compounds in tomato and watermelon plants reports: “Tomato plants, Lycopersicon esculentum L. cv. Tmknvf2, and watermelon plants, Citrullus lanatus [Thomb.] Mansf. cv. Dulce maravilla, were grown for 30 days at different temperatures (15, 25 and 35°C). We analysed soluble phenolics, enzymatic activities(phenylalanine ammonia-lyase, polyphenol oxidase and peroxidase), and dry weight. The impact of the three temperatures was different in tomato and watermelon. Our results indicate that heat stress in tomato plants occurred at 35°C, while chilling stress occurred in watermelon plants at 15°C. Thermal stress in both plants caused: (1) decreased shoot weight; (2) accumulation of soluble phenolics; (3) highest phenylalanine ammonia-lyase activity; and (4) lowest peroxidase and polyphenol oxidase activity. These results indicate that thermal stress induces the accumulation of phenolics in the plant by activating their biosynthesis as well as inhibiting their oxidation. This could be considered an acclimation mechanism of the plant against thermal stress.”
6. ROOT HYPOXIA!
Now I am getting really excited! I can’t sleep I am so excited! This is my favorite stressor at the moment! Here is how it goes:
1. With flooding, roots suffer from a lack of oxygen (anoxia).
2. Hypoxia/anoxia induces the synthesis of 1-aminocyclopropane-1-carboxylic acid (ACC) in the flooded roots.
3. The roots synthesize ACC (1-Aminocyclopropane-1-carboxylic acid) from S-adenosyl-L-methionine (SAM), our old methyl donor friend.
4. The roots synthesize SAM from the amino acid methionine.
5. ACC is transported upwards in the plant and then oxidized in the leaves
6. An end result is production of ethylene.
Conclusion: Ethylene is the “upstream” stress hormone that induces polyphenol synthesis in plants. Its production is downstream of some stresses like root hypoxia. To use a human biochemical analogy, Ethylene is the plant “ACTH” for the induction of polyphenols, which are the plant’s “cortisol.” The Ethylene is “upstream” and the polyphenols are “downstream” compounds synthesized in response to stresses.
It seems fairly clear that if you pull a plant up by its roots, like a carrot or turnip or onion, root hypoxia can also happen.
Now this raises even more questions. For example, how is ethylene synthesized?
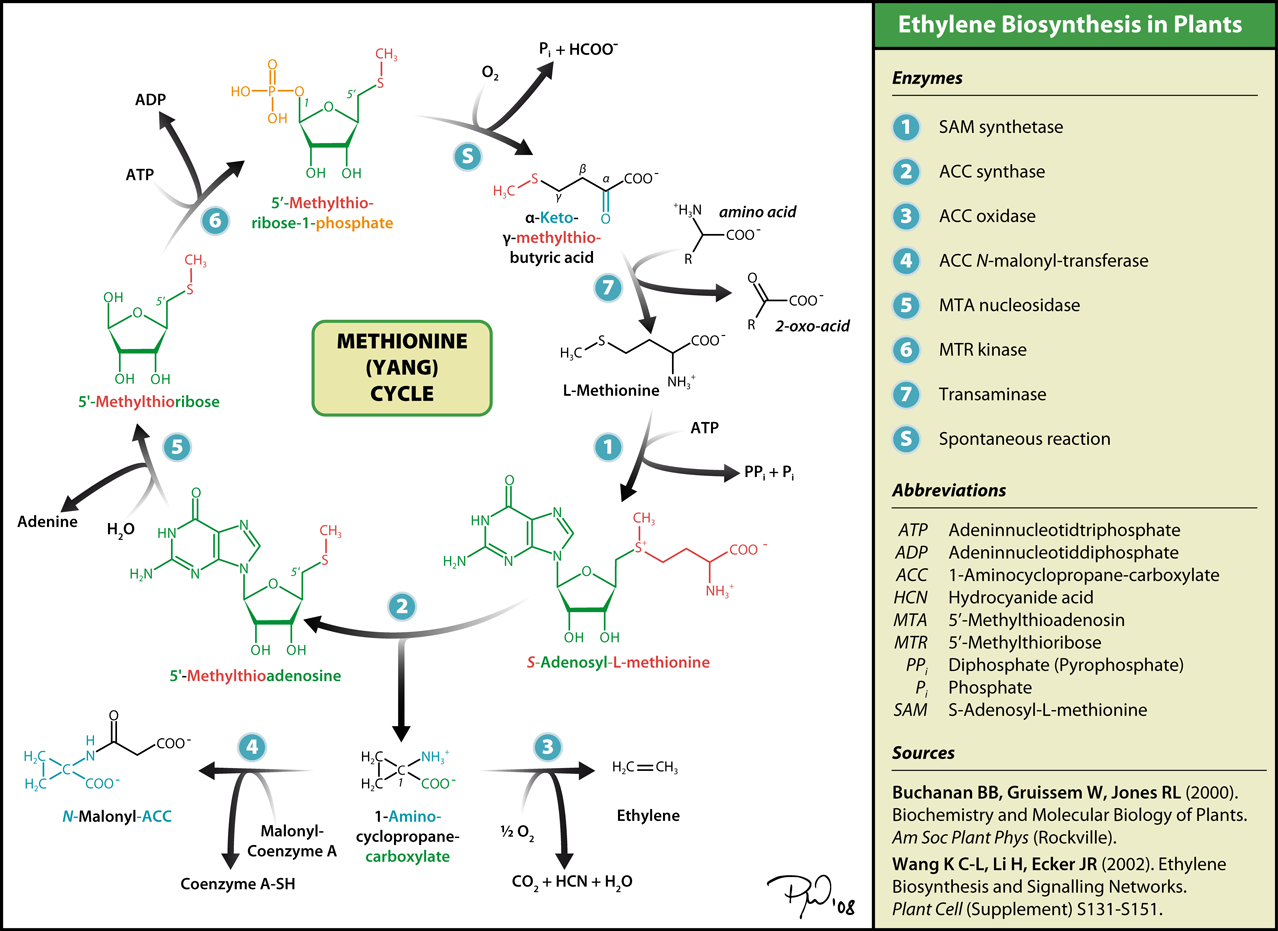
7. How Ethylene is Synthesized: The Yang Cycle
This is really getting interesting! The synthesis pathway for ethylene is called the Yang Cycle. After studying it, however, I think a better term would be the “SAM cycle” (see diagram below)
As you can see, the Yang cycle is really just a “spin off” from methionine synthesis. However, to synthesize ethylene, you must create the intermediate S-adenosyl-L-methionine (SAM) and then ACC. (SAM is one of the best epigenetic drugs out there – a methyl donor for DNA cytosine Methylation) Then ACC is converted to ethylene in the presence of oxygen.
Why all this biochemistry? Well the diagram above misses the entire point!
THE ROOTS DIDN’T HAVE ENOUGH OXYGEN – THAT IS WHY THEY SYNTHESIZED ACC THE LEAVES HAD ENOUGH OXYGEN, SO THEY COULD COMPLETE THE ETHYLENE “HORMONE SYNTHESIS” PATHWAY.
Summary:
In summary, root hypoxia during flooding induces the conversion of methionine to SAM, which is then converted to ACC. ACC migrates from the roots to the leaves, which do have enough oxygen, where it is converted to ethylene in the presence of oxygen. Why do leaves have enough oxygen? THEY SYNTHESIZE OXYGEN, STUPID! In other words, because plant
leaves can synthesize oxygen from carbon dioxide in chlorophylls by photosynthesis, the methionine-SAM-ACC-ethylene pathway is a way for hypoxic roots to tell the entire plant that it is time to mature and then die! Ethylene is the stress hormone that does all of that.
8. Anthocyanins are the polyphenols responsible for the fall colors of leaves
Once I found out that root hypoxia causes ACC synthesis in the roots, which migrated to the leaves to induce ethylene synthesis, which in turn induced polyphenol synthesis, I asked myself “why”? (again my Why Gene has reared it’s ugly head). Here is what I started to think about:
In many countries, there is not only a spring rain, but an autumn rain. Plants exposed to flooding need to cope with this “water stress”. Plants synthesize anthocyanins in response to this stress.
It appears that anthocyanins (once thought to just be UV sunscreens for plants) are osmotic regulators of plants. Specifically, anthocyanins can decrease leave osmotic potential.
There we go! Another “proof” that the polyphenol pathway is a stress response in plants.
But why turn into all of those fall colors and then die? Well it is now obvious – polyphenols also induce plant cell senescence.
Read on, unless you are scared!
9. Ethylene induces leaf senescence and xylem senesence (i.e. time to get rid of the leaves for the winter). Perhaps this is why polyphenols induce ROS
Xylem tissue is water and nutrient transporting tissue in vascular plants.
Here is a list of some of the things that ethylene does in plants (I eliminated the ones that were too confusing)
- Stimulates leaf and flower senescence
- Stimulates senescence of mature xylem cells in preparation for plant use
- Induces leaf abscission
- Induces seed germination
- Induces root hair growth — increasing the efficiency of water and mineral absorption
- Induces the growth of adventitious roots during flooding
- Stimulates fruit ripening
- Interference with auxin transport (with high auxin concentrations)
Reference: again, Wikepedia article on Ethylene
Summary: ethylene and the anthocyanin synthesis that the ethylene induces is a way of ending the life cycle for leaves. It is a way of saying….time to die.
What relevance does this have for us? A lot! Anthocyanins do NOT induce cellular senescence in humans, of course. But we know that high levels of many of these polyphenols induce ROS, and that this is the mechanism by which they kill cancer. Although I have not yet validated this in the plant literature (I ran out of time), my theory now is that high levels of anthocyanins produced in the Fall as a result of heat stress, water stress, etc. induce ROS production in plant leaves, which then make them die and fall off the tree. If so, this explains why polyphenols induce mammalian ARE genes and cell survival, but can also induce ROS and apoptosis in cancer. In other words, the polyphenols do the same thing to cancer cells as they do to leaves in the fall…..they kill them. I will likely research this further and create another blog entry on the topic
Conclusion: Polyphenols are pleiotropic compounds synthesized in plants in response to multiple plant stressors. Thus:
- Root flooding induces hypoxia, which induces ACC synthesis from methionine via SAM. ACC can migrate to the leaves where chlorophyl can synthesize oxygen to complete the process of producing ethylene. Ethylene is actively synthesized by plants to induce polyphenol synthesis.
- Polyphenols such as anthocyanins are also “osmotic stress hormones” that help them cope with with water stress.
- Polyphenols are implicated in many other stress coping pathways that can also be used to induce deciduous changes with the fall. Specifically, they can induce leaf senescence and make the leaves fall off. The bright colors seen in fall leaves are all due to anthocyanins, which induce leaf senescence and death.
- The mechanism by which they induce senescence is unclear, but I suspect it is a ROS-mediated induction of cellular senescence. This is why polyphenols can induce cancer cell death…..i.e. I strongly suspect it is the same as inducing leaf death.
10. Several transcription factors involved in plant polyphenol synthesis have been identified and studied
These include AtMYB12, VvMYBF12, VvMYBPA1, VvMYB5b, 2R3MBY and DkMyB4 With this knowledge, new means are being suggested for significantly increasing the polyphenol content in foods. These means are likely to be of interesting for post-harvesting interventions as well as pre-harvesting ones.
AtMYB12
The 2008 publication AtMYB12 regulates caffeoyl quinic acid and flavonol synthesis in tomato: expression in fruit results in very high levels of both types of polyphenol reports “Plant polyphenolics exhibit a broad spectrum of health-promoting effects when consumed as part of the diet, and there is considerable interest in enhancing the levels of these bioactive molecules in plants used as foods. AtMYB12 was originally identified as a flavonol-specific transcriptional activator in Arabidopsis thaliana, and this has been confirmed by ectopic expression in tobacco. AtMYB12 is able to induce the expression of additional target genes in tobacco, leading to the accumulation of very high levels of flavonols. When expressed in a tissue-specific manner in tomato, AtMYB12 activates the caffeoyl quinic acid biosynthetic pathway, in addition to the flavonol biosynthetic pathway, an activity which probably mirrors that of the orthologous MYB12-like protein in tomato. As a result of its broad specificity for transcriptional activation in tomato, AtMYB12 can be used to produce fruit with extremely high levels of multiple polyphenolic anti-oxidants. Our data indicate that transcription factors may have different specificities for target genes in different plants, which is of significance when designing strategies to improve metabolite accumulation and the anti-oxidant capacity of foods.”
3. VvMYBF1
There are some innovative scientists in Germany who have identified the transcription factor in grapes that controls polyphenol synthesis in grapes. This is part of the R2R3-MYB family and has a specific domain in it called the SG7 domain. It is unclear to me if this transcription factor is “upstream” or “downstream” from the two transcription factors listed below. I assume the transcribed polyphenols would include resveratrol, but this was not specifically studied.
4. VvMYBPA1 and VvMYB5b
A group of scientists in Australia and in France have identified the transcription factors found in grapevines that regulates the synthesis of proanthocyanidins. These are precursors in the flavanoid synthesis pathway. They have not tried over-expressing these transcription factors, but this is good news for those of us who like protoanthocyanidins. These are the molecules found in chocolate that are so good for us.
References: Publications related to the molecular mechanisms of the above and other modes of plant polyphenol production include:
- The grapevine transcription factor VvMYBPA1 regulates proanthocyanidin synthesis during fruit development.
- The transcription factor VvMYB5b contributes to the regulation of anthocyanin and proanthocyanidin biosynthesis in developing grape berries.
- Proanthocyanidin synthesis and expression of genes encoding leucoanthocyanidin reductase and anthocyanidin reductase in developing grape berries and grapevine leaves.
- The grapevine R2R3-MYB transcription factor VvMYBF1 regulates flavonol synthesis in developing grape berries.
- Ectopic expression of VvMybPA2 promotes proanthocyanidin biosynthesis in grapevine and suggests additional targets in the pathway.
- Berry skin development in Norton grape: distinct patterns of transcriptional regulation and flavonoid biosynthesis.
- The Arabidopsis TT2 gene encodes an R2R3 MYB domain protein that acts as a key determinant for proanthocyanidin accumulation in developing seed.
- DkMyb4 is a Myb transcription factor involved in proanthocyanidin biosynthesis in persimmon fruit.
- Identification of key amino acids for the evolution of promoter target specificity of anthocyanin and proanthocyanidin regulating MYB factors.
- Isolation of WDR and bHLH genes related to flavonoid synthesis in grapevine (Vitis vinifera L.).
- The basic helix-loop-helix transcription factor MYC1 is involved in the regulation of the flavonoid biosynthesis pathway in grapevine.
- Leucoanthocyanidin reductase and anthocyanidin reductase gene expression and activity in flowers, young berries and skins of Vitis vinifera L. cv. Cabernet-Sauvignon during development.
- Metabolic engineering of proanthocyanidins by ectopic expression of transcription factors in Arabidopsis thaliana.
- TT2, TT8, and TTG1 synergistically specify the expression of BANYULS and proanthocyanidin biosynthesis in Arabidopsis thaliana.
- A pomegranate (Punica granatum L.) WD40-repeat gene is a functional homologue of Arabidopsis TTG1 and is involved in the regulation of anthocyanin biosynthesis during pomegranate fruit development.
- The wound-, pathogen-, and ultraviolet B-responsive MYB134 gene encodes an R2R3 MYB transcription factor that regulates proanthocyanidin synthesis in poplar.
- MATE transporters facilitate vacuolar uptake of epicatechin 3′-O-glucoside for proanthocyanidin biosynthesis in Medicago truncatula and Arabidopsis.
- TRANSPARENT TESTA 19 is involved in the accumulation of both anthocyanins and proanthocyanidins in Arabidopsis.
- Impact of diurnal temperature variation on grape berry development, proanthocyanidin accumulation, and the expression of flavonoid pathway genes.
- Identification and characterization of MYB-bHLH-WD40 regulatory complexes controlling proanthocyanidin biosynthesis in strawberry (Fragaria × ananassa) fruits.
11. Other Interesting Questions that May be Practical
A. Would Hot house maturation of live fruit and vegetable plants with stress allow for even greater stress responses to occur in the fruits and vegetables?
We live in the city and have practically no front yard. Our back yard is a steep bank that is untillable unless you want to roll down a 100 foot bank. For this reason, I am in the process of creating a rooftop garden. The major problem with our roof top is that it gets so darn hot up there. I initially thought this would be a real problem, but now after reading the Xenohormetic Live Food Hypothesis blog entry, I am thinking this may be a great “problem” to have! Here are some of my ideas:
1. Cultivating tomatoes with 35 C (95 F) conditions induces polyphenol synthesis and prevents polyphenol oxidation
Here is an article from Granada in 2000 that shows how growing tomatoes at 35 C induces more polyphenols than growng them at 15 C or 25 C. This was a two-fold effect in enzyme production of some of the metabolites of polyphenols. (i.e. a 100% increase). This is no surprise, since it appears that polyphenols are “stress coping molecules.” According to this article:
a) thermal stress induces polyphenol biosynthesis and
b) thermal stress inhibits the oxidation of polyphenols.
The plants did not grow very well at this temperature, however. This is why I would like to grow them at more tolerable temperatures, then “stress them” when the tomatoes have fully formed.
Referemce: Resistance to cold and heat stress: accumulation of phenolic compounds in tomato and watermelon plants.
Comments: My study of incubating blood under hyperthermic conditions and also under hypoxic conditions has led me to a greater understanding of the heat shock response elements and the hypoxic response element genes in humans.
Whereas 30 minutes of heat exposure (i.e. a long hot tub bath) does seem to activate existing heat shock proteins, meaningful changes in gene expression secondary to the nuclear translocation of the heat-induced timerization of HSF1, HSF2, HSF3, and HSF4 take several hours to occur. Peak increases in gene expression many times do not occur for 12-24 hours. For this reason, I would think that a similar effect might be seen when plants are exposed to 110-120 degree F temperatures. 30 minutes of steaming would be good, but 12-24 hours of “hot as Hell” conditions may be even better. I would like to experiment with this. The best place to do this would be on my roof, where we could easily induce these conditions in a solar reflector heated hot house (i.e. green house).
B. Would Inducing root hypoxia with root flooding be good for polyphenol content in vegetables?
This may be bad if done too much and continuously, but intermittent flooding would probably be a good thing.
C. Would cultivating tea plants with excess ethylene produce more EGCG?
I am not sure that we can make generalizations about all polyphenol synthesis pathways. I am very interested in producing some high ECGC tea leaves. This is just a thought, since I think that it takes several days to really induce synthesis via such pathways.
I expect to publish my next blog entry on this topic soon.


Pingback: Health through stressing fruits and vegetables – the Xenohormetic Live Food Hypothesis (updated) | AGING SCIENCES – Anti-Aging Firewalls
Vincent,
How might this effect the balance of gut flora? What metabolic paths are then effected downstream? What we eat and what drugs we take can effect our glucose and insulin levels and our healthy or unhealthy life spans. Eric
What seems important for aging is the ability to lower glucose and insulin. Can adjustments to the diet and drugs increase healthy lifespan? Can the research lead to other ways to increase healthy lifespan? Eric
These are truly wonderful questions and could and should set the agendas for new multi-year research programs. I firmly believe that really powerful questions move science forward. I expect that we will be producing new blog entries related to gut flora in the near future. And, thanks for the citation link.
Vince
IMAGE: Andrew Patterson, assistant professor of molecular toxicology, Penn State, is in his lab.
Click here for more information.
A drug that appears to target specific intestinal bacteria in the guts of mice may create a chain reaction that could eventually lead to new treatments for obesity and diabetes in humans, according to a team of researchers.
Pingback: Plant Communications | AGING SCIENCES – Anti-Aging Firewalls