By James P Watson with Preface and editorial assistance by Vince Giuliano
Preface by Vince Giuliano
According to the latest theories of cosmology, the universe has no center. However, anyplace in the universe, including where we are on Earth, can be a center from which systematic exploration can be launched to determine the nature of the universe. And different physical phenomena might be revealed departing on where you are looking out from. Similarly, according to the GUT concept of biology we are developing, biology has no center. There is no unique point of departure or view that can hope to explain the incredible complexity of biology or aging. And also similarly, different biological insights can be revealed depending on where you start to look out from. There are very many such places to look out from, anchor points of intellectual departure that can provide valuable insights: cell components like mitochondria, the nucleus, the nucleolus, chromatin in general, histones, lysosomes, cell membranes, hormones, key kinases, gene activating factors and cofactors, exosomes, microtubules, membrane transport proteins, etc. Also it is possible to look out from biological processes as centers, processes such as gene transcription, post-transcriptional modifications, inter and intra cell signaling, endocytosis, ribosylation, phosphorylation, methylation, acetylation, circadian rhythms, inter-species signaling and hormesis – just to name some of very many. It is possible to start with a class of substances such as sirtuins, hormones, acetylating or methylating substances, and expand out from there to examine what they say about biology and aging. You can even start with a single molecule such as NAD+ and even a very simple one like carbon monoxide. Or you can start with a substance like bitter melon that contains multiple plant polyphenols and other chemicals, each with complex biological actions. So, a corollary to the fact that biology has no center is that most anyplace can serve as a center to look out from.
The followers of this blog will recognize that to a large extent, representing such multiple viewpoints and examining biology and aging from different ones of them is what we have been attempting to do all along in this blog. Of course, we still have an incredibly long way to go. An important aspect of this multiple viewpoint approach is coming back again and again to the same topic, looking at it from different viewpoints to gain additional insights. For example take histone acetylation/de-acetylation. We have looked at these to a greater or lesser extent from the viewpoints of cancer, aging, multiple specific genes, sirtuins, chromatin structure, DNA Methylation, plant polyphenols, stem cell differentiation, metabolic processes, mitochondrial communications, biological stresses, and dementias. We believe that each time we cross such a subject again from a different perspective we can usually gain a little more insight, and that is our objective.
Telomere biology is a very familiar topic in the earlier history of this blog (2009-2012), and previously we have covered it from many viewpoints in over a dozen blog entries, often addressing central questions such as “Are telomere lengths the central clocks measuring aging?” No; “Do short telomere lengths drive cell senescence, or is it the other way around? Yes and Yes; “Is telomere shortening a fundamental upstream driver of aging?” No; “Can I extend my health and effective lifespans by taking a substance that purportedly extends my telomeres?” Probably not; If I extend my telomeres will I probably live longer?” Yes, but probably by other molecular alterations you make upstream of telomere lengthening not because of the longer telomeres themselves. “Are there practical interventions that can keep my telomeres long? Yes, many.
However, many questions remained open and telomere biology continues to represent an important point of view for examining aging. In that spirit, This three-part blog entry series offers The View from the Telomere end of the Chromsome. A lot of the material here is new and wanders into adjacent areas of histone biology. DNA adjacent to telomeres, telomere-mitochondria signaling, gene activation factors and cofactors and roles of key genes like P53. This is Part 1 of 3 in the series . Because of the length of the extended telomere story, we subdivided the discussion into three parts. this Part 1 relates to a number of more-practical and less-technical topics: the history of telomere biology, telomere length testing, telomerase Inhibitors for cancer, and supplements that activate telomerase and their possible roles for inhibiting or preventing cancers. Part 2 deals in finer detail with new discoveries related to the molecular biology of telomeres, Sections 1-9. Part 3 relates to additional newer discoveries, Sections 10-17. It also includes a concluding discussion of some of the implications for a Grand Unified Theory of biology of Aging and Biology, as viewed from the ends of a chromosome. And Jim tells me he is now drafting another blog entry which offers The View from the Centromere middle of the Chromsome.
Introduction and History of Telomere Biology – The Tale of Chromosome Tails
Chromosomes have “tails” called telomeres. The tails have “caps” called “Shelterin proteins”. The discovery of these tails, the caps, and how the tails shorten is a fascinating story that I have titled “The Tale of Chromosome Tails”. It is an exciting story that started out with erroneous conclusions, but ended with an explanation of important triggers of aging and cancer. Most recently, this same “tale” has been linked to the reason why mitochondria don’t work in aging and cancer, to the DNA damage response, to the P53 gene, and to other important matters. . To properly tell this “tale”, I start the story as a “once upon a time, 102 years ago, when people thought that cells were immortal”. I will start here with Alexis Carrel, a surgeon from New Orleans.
The Origin and Death of the Concept of Somatic Cell Immortality
In 1912, Alexis Carrel published the now infamous paper titled “On the permanent life of tissues outside the organism”. In this paper, he described how he was able to keep chick heart cells alive indefinitely, provided that the culture medium was frequently changed with fresh nutrients (Witkowski, 1980). What Alexis Carrel did not know was that the chick embryo extract which he was using as nutrition for the chick heart cells actually had chick cells in the embryo extract. This oversight let to the erroneous dogma that cells were immortal. For this reason, it was a shock to the scientific community when Leonard Hayflick cultured human diploid fibroblasts and noted that they consistently stopped dividing after 40-60 cell divisions (Hayflick and Moorhead, 1961). This gave rise to the term Hayflick limit (Shay, 2000), which referred to the maximal number of times cells could divide. Not all cells stop dividing after 40-60 divisions, however. In 2009, Elizabeth Blackburn, Carol Greider, and Jack Szostak were awarded the Nobel Prize in Physiology or Medicine for discovering the enzyme telomerase, which allows cells to divide much more than 40-60 times. (Press Release, 2009). For instance, testicular spermatogonia robustly express telomerase which allows them to continuously divide throughout life. By age 50, males produce sperm that have undergone approximately 840 cell divisions (Goriely, 2013). Cancer cells also express telomerase in 85% of cases, which explains their immortality. In the remaining 15%, an alternative method for telomere lengthening is activated (called the ALT pathway). However, in cells not expressing either telomerase or ALT pathway genes, telomere shortening was thought to be an “aging clock” that could measure physiologic age and predict death or disease.
About telomere lengths
The Life and Death of the Telomere Clock Theory of Aging
Unfortunately, the “telomere clock” did not keep time very well….it often ran too fast or could run in reverse with intervention or without intervention) (Hovatta, 2012). Part of this could be explained in terms of telomerase activation consequential to lifestyle modifications such as exercise, diet, or meditation. (Ornish, 2008) (Hoge, 2013). On an experimental level, major flaws in the “telomere clock theory” were found. A major one was the discovery that mice had long telomeres (50-150 kb) relative to human telomeres (15 kb), yet humans live as much as 50 times longer than mice (2 years vs 90-100 years) (Greider, 1996). If the “telomere clock theory” was correct, mice should outlive humans. The second flaw in the “telomere clock concept” was the discovery that mice expressed the enzyme telomerase, which should lengthen lifespan in mice to more than humans (Prowse, 1995). Instead, telomerase activation in mice appeared to contribute to their short lifespan since this increased the tendency of mice to form tumors (Due in part to the expression of telomerase, 90% of mice develop tumors during their lifespan). This lead to the theory that telomere shortening actually functioned as a tumor-suppressor mechanism which promoted organismal survival, but at the expense of getting old (Campisi, 2001).
The 3rd major flaw in the “telomere clock concept” was the discovery that telomere shortening occurred with other cellular phenomena besides mitosis (i.e. cell division). For instance, radiation was found to shorten telomeres (Fritz, 2000). In addition, it was found that radiation could induce cell cycle arrest even if telomeres did not shorten (Suzuki, 2001). Ultraviolet light was found to shorten telomeres, with or without cell division (Oikawa, 2001). The same was found with chemotherapy and toxins (Engelhardt,1998) (Muller, 2006). The 4th major flow in the “telomere clock concept” occurred when it was discovered that oxidative stress accelerated telomere shortening and that telomere shortening could occur even in cells that were not dividing (Zglinicki, 2002). Since oxidative stress is a universal feature of aging, this meant that telomere shortening was an “effect” of aging, rather than the “upstream cause” of aging. As a result of these 4 major flaws in the telomere clock theory, as well as the incongruous evidence from the telomere lengths in mice and men, the theory that telomeres are accurate measures of biological age has largely been put to rest. You can see the 2010 blog entries Telomere lengths, Part 3: Selected current research on telomere-related signaling, telomere lengths, cancers and disease processes, Part2: lifestyle, dietary, and other factors associated with telomere shortening and lengthening, and Part1: telomere lengths, cancers and disease processes.
Today, telomere length is merely considered to be a good biomarker of oxidative stress and should be measured at two time points (before and after) in a prospective clinical trial to really make meaningful conclusions. Measuring telomere length in at a single point in time and extrapolating “biological age” from this one measurement is suspect and subject to fundamental statistical errors based on standard deviations from the mean.
Telomere Length Testing and How to Avoid the Pitfalls of “Reverse Extrapolation”
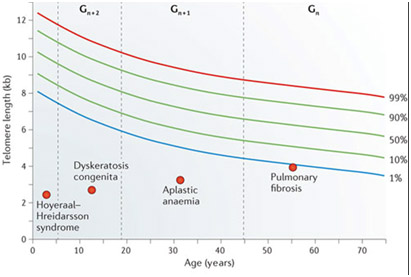
Measuring peripheral blood mononuclear cell telomere length has become a standardized, validated way of measuring oxidative stress. However, this is an “empiric science” and has many potential pitfalls. The following graph shows how telomere lengths normally decline with aging but that standard deviations from the norm exist (1% vs 99%).
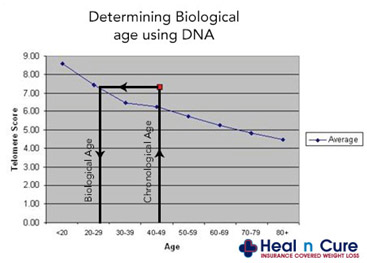
As you can see from the graph above, the standard deviation bars for telomere length for a given age can tempt a clinician to make a statistical error called “reverse extrapolation”. Here an individual with longer telomeres may fall within the normal standard deviations for his age, but in many telomere length test marketing brochures, a telomere test result is extrapolated back to the 50% mean for age. This results in a nice report for the paying customer, who gets a report like the one below:
In the advertisement above, no standard deviation error bars are depicted (unlike the first graph show above). As a result, the customer is told that “they are 29 years old based on their telomere length”. (in reality, the customer’s telomeres may be within one standard deviation from the mean for a 45 year-old, but the report will state that his “biological age” is 29). In this case, telomere length testing can be mis-used to make people “feel younger”. Here is another graph that hides the age-related scatter in lengths.
Here is an actual scatter plot off TRF length vs aging for different individuals, presenting quite a different picture:

As you can see, measured telomere length is a very poor predictor of biological age. In a scientific study, there is an easy way to eliminate this problem – always compare the subjects telomere length tests to his own baseline. This is why any study should measure telomere length before and after the study is completed and compare an individual’s telomere length with their own telomere length prior to the intervention or treatment.
Telomere Length and the Risk of Cardiovascular disease, Infectious Disease, and Cancer
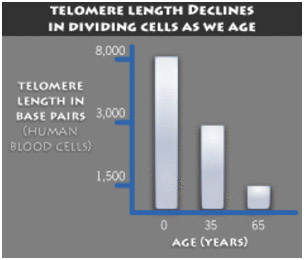
Unfortunately telomeres in general gradually shorten with aging, with a mean telomere length decline of 9% per decade (Brouilette, Lancet, 2007). This telomere shortening phenomena is highly reproducible and was originally thought to occur at a constant rate, due to cell division, and could thereby serve as an “aging clock”. (Vaziri, 1994). Hundreds of studies were done showing excellent correlation between peripheral blood leukocyte telomere length and objective physical measurements of health, such as pulse pressure (Jeancois, 2000), pulse wave velocity (Benetos, 2001), etc. As more and more studies were completed, however, it was clear that telomeres did not shorten at a constant rate. Instead, the rate of telomere shortening varied with disease. For instance, patients with shorter leukocyte telomeres had a 3-fold higher mortality rate from heart disease and a 8.5-fold higher mortality rate from infectious diseases (Cawthorne, Lancet, 2003). Another study showed that younger individuals with shorter telomeres had a 2.8-3.2 fold higher risk of premature myocardial infaction (Brouilette, 2003). Other studies showed the same cardiovascular risk effect in older individuals (Starr, 2007). Finally, a study showed that peripheral blood leukocyte telomere length was shown to have predictive value as a test for advanced cardiovascular disease (Willeit, 2010). More recently, telomere length was shown to be an independent risk factor for cancer (Willeit, 2010). In a 10-year Italian study, those with the longest telomeres had the lowest risk of cancer (5.1 cases per 1,000 person-years) and those with the shortest telomeres had the highest risk of cancer (22.5 cases pre 1,000 person-years). In Poland, a study was done which showed that short peripheral blood leukocyte telomeres doubled the risk of gastic cancer (Hou, 2009). In summary, there is a clear link between short telomeres and disease risk. The question is what is the mechanism for this link? The answer is cellular senescence.
Telomerase Inhibitors for Cancer and Supplements that Activate Telomerase
Since 85% of cancers constitutively express telomerase, several institutions started developing telomerase inhibitor drugs for treating cancer. (Mergny,1999) (Norton,1996). Unfortunately, the telomerase inhibitor drugs failed in clinical trials for brain cancer and breast cancer (Geron, 2012). However, this same drug appears to be working for myelofibrosis, a rare type of bone marrow myeloproliferative disease. (Carrol, 2013). For aging, several supplement manufacturers have created supplements made of natural products such as the Chinese herb, astralagus (TA-65, Product B, etc.). Although these supplements have been shown to lengthen telomeres in mice, where the enzyme telomerase is expressed (de Jesus, 2013), they have failed to lengthen telomeres in human peripheral blood mononuclear cells (Harley, 2011). On the other hand, lifestyle modification with exercise, diet, and mediation have been proven to lengthen telomeres in humans. (Ornish, 2008) (Hoge, 2013) (Lin, 2012). Folic acid, Fish oil, Vitamin C, D, and E have been associated with longer telomeres, when measured at time points in large population studies, but no prospective, randomized clinical trial has been done to show that multivitamin supplementation will actually lengthen human telomeres. (Paul,2009) (Xu, 2009), (Farazaneh-Far, 2010) (Paul, 2010). Zinc has also been shown to activate telomerase in cancer cells in vitro, but no human clinical trials have shown that Zinc supplementation will lengthen telomeres (Nemoto, 2000).
This raises the question about whether folate, fish oil, Vitamins C, D, E, and Zinc are harmful, if a person has cancer and is being treated for cancer. There is evidence that in the setting of chemotherapy for cancer, exogenous anti-oxidants actually decrease the efficacy of cancer chemotherapy. The mechanism of telomerase activation could possibly explain part of this phenomena. (The phenomenon is also explainable by the impacts of such substances on reduction of chemotherapy-induced ROS which induces apoptosis in cancer cells.) In summary, activating telomerease is probably good for non-cancer cells but possibly harmful if one has cancer. On the other hand, telomerase inhibitor drugs failed to treat brain and breast cancer.
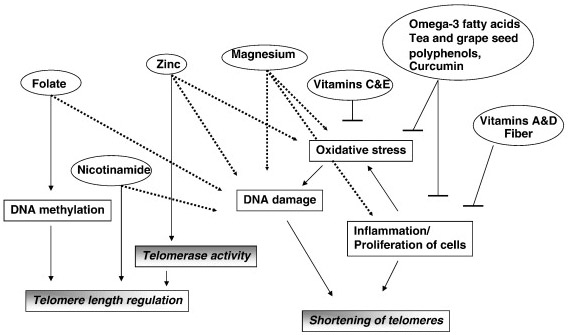
Many natural products known to have health benefits and anti-cancer properties. For this reason, several of these natural polyphenols with chemopreventative effects have been tested for their effects on the enzyme telomerase. To date, most of these have been shown to have inhibitory effects on telomerase, such as ECGC, hydroxytyrosol, resveratrol, curcumin, cinnamic acid, Bosweillic acid, (Yokoyama, 2004), (Naasani, 2003), (Rafehi,2012), (Fuggetta, 2006), (Cui, 2006) (Chakraborty, 2006), (Mishra, 2008), (Zang Ting,2001), (Zhenhua, 2000), (Khan, 2011). The mechanism by which curcumin inhibits telomere lengthening appears to be due to the prevention of the nuclear localization of telomerase (Lee, 2010). In summary, several plant polyphenols appear to inhibit the enzyme telomerase. This may be a major reason why they are unique and different than multivitamins in their anti-cancer value. The following diagram from Ligi Paul’s article does not adequately distinguish this effect of polyphenols on telomerase inhibition. However, it also shows how polyphenols can reduce oxidative stress and inflammation, thereby reducing the shortening of telomeres due to these factors which are independent from telomerase expression.
Image source “Potential mechanisms behind the influence of nutrients on telomere length. Nutrients influence telomere length by various mechanisms that reflect their role in cellular functions. Dashed line indicates effect of deficiency of a nutrient.”
Shift of focus to cellular senescence
As a result of all these discoveries, the focus has shifted from the concept of telomere shortening to the concept of cellular senescence. Specifically, cellular senescence means “cell cycle arrest” and can be the consequence of telomere shortening. But since cellular senescence can be induced WITHOUT telomere shortening, telomeres no longer are viewed as the focal point for interventions and instead, are now considered to be a GOOD BIOMARKER for oxidative and other stress, rather than a true “biological clock.” Studies showed that even psychological stress could shorten telomeres. Both perceived stress and true chronic stress could prematurely shorten telomeres (Epel, 2004). “Women with the highest levels of perceived stress have telomeres shorter on average by the equivalent of at least one decade of additional aging compared to low stress women.”
Partial picture of the telomere-aging-cancer elephant
A partial picture of the “telomere aging elephant” is given in the following graph. Here is the sequence of events.

Image and legend source “Telomere length is central to the process of aging and tumorigenesis in human cells. As result of the “end of replication problem”, telomeres shorten after each cell division. When telomeres reach a critical length, the recruitment of telomere binding proteins and telomere capping are insufficient. Uncapped chromosome end resembles a double stranded DNA break that is highly unstable and can give rise to chromosome rearrangements. Since telomere erosion limits the proliferative capacity of cells, premalignant transformed clones cease to expend when their telomeres shorten. Critically shortened telomeres elicit a potent DNA damage response and accumulate several DNA damage markers such as phosphorylated gH2AX, 53 BP1, CHK2 for instance (d’Adda di Fagagna et al., 2003; Takai et al., 2003). Many of these proteins localize directly to dysfunctional telomeres to form dysfunctional telomere-induced foci (TIFs). However, entry into senescence or apoptosis can be bypassed by dysfunctional cell cycle controls and lead to the accumulation of chromosomal rearrangement and a global instability of the genome. Telomeres are tightly linked to tumorigenesis and the reactivation of telomerase circumvents telomere shortening and enhances the proliferative capacity of a subset of cells.”
The image and explanation are over-simplified and do not convey that many causes besides cell replication can lead to telomere shortening, particularly stresses of various kinds, that telomere lengths can actually be lengthened, and the obvious points that genomic instability. cancer and cell deaths can lead to shorter lives (although apoptosis can be a good thing for longevity).
Summary of the Tale of Chromosome Tails
Today telomere shortening is considered a reversible event. In humans, telomeres generally shorten by 9% per decade, but this rate can be increased by oxidative stress, psychological stress, radiation, UV, genotoxic drugs, oncogenic stress, and many other factors. The beneficial effects of telomere shortening appears to be related to its tumor suppressor function. The harmful effects of telomere shortening is cell cycle arrest, which is called cellular senescence. Cellular senescence can occur even without telomere shortening, however, and appears to be reversible in some instances prior to the formation of senescence-associated heterochromatin formation (SAHF) in the nucleus of the cell. Today telomere length measurements are considered useful biomarkers of oxidative stress, rather than a “mitotic clock” for measuring biological age or life span. Lifestyle modification can increase telomere length by as much as 10%, but this does not reverse the physiological appearance (phenotype) of aging.


Pingback: NAD+ an emerging framework for life health and life extension — Part 2: Deeper into the NAD World, hopeful interventions | AGING SCIENCES – Anti-Aging Firewalls