By Vince Giuliano with important contributions by James P Watson
 Glucosamine is one of the most popular dietary supplements, used by millions who hope that it will lessen the ravages of osteoarthritis, arthritis, loss of cartilage and associated pain and discomfort. This natural shellfish-derived substance has been known since 1876 and its biological impacts were reported as far back as in 1919(ref). I have suggested taking Glucosamine since the 2008 version of the treatise Antiaging Firewalls – The Science and Technology of Longevity.
Glucosamine is one of the most popular dietary supplements, used by millions who hope that it will lessen the ravages of osteoarthritis, arthritis, loss of cartilage and associated pain and discomfort. This natural shellfish-derived substance has been known since 1876 and its biological impacts were reported as far back as in 1919(ref). I have suggested taking Glucosamine since the 2008 version of the treatise Antiaging Firewalls – The Science and Technology of Longevity.
Most of us have long thought of this substance as having a narrow purpose, focused on combating osteoarthritis and on cartilage regeneration . However, there has been mounting evidence that regular supplementation with glucosamine can significantly enhance longevity – in mammals probably as much as well studied interventions such as taking rapamycin. Here, I review studies that provide strong evidence for this prospect.
Research evidence for the new take on glucosamine is of several kinds: 1. biological mechanism of operation, 2. animal longevity impact studies and 3. human epidemiological studies . I start here with the first and second and then move to the third. Finally. I briefly review how the biological mechanisms of glucosamine compare to those of important alternative approaches to mitochondrial health.
If you are not familiar with metabolic processes in mitochondria and the concept of mitohormesis, you might want to review a few past blog entries as background to this one:
Mitochondria in health and aging, and possibilities for life prolongation – Part 1:basics
Biological activities of glucosamine and animal studies
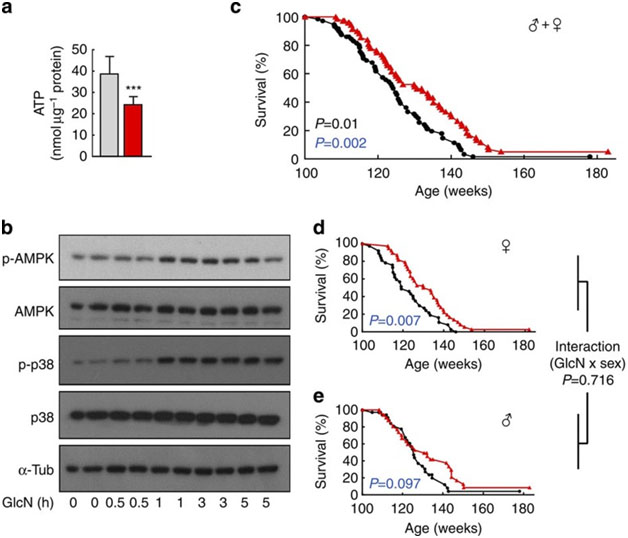
The most recent and most powerful study supporting you longevity impacts of glucosamine appeared in April 2014: D-Glucosamine supplementation extends life span of nematodes and of ageing mice: “d-Glucosamine (GlcN) is a freely available and commonly used dietary supplement potentially promoting cartilage health in humans, which also acts as an inhibitor of glycolysis. Here we show that GlcN, independent of the hexosamine pathway, extendsCaenorhabditis eleganslife span by impairing glucose metabolism that activates AMP-activated protein kinase (AMPK/AAK-2) and increases mitochondrial biogenesis. Consistent with the concept of mitohormesis, GlcN promotes increased formation of mitochondrial reactive oxygen species (ROS) culminating in increased expression of the nematodalamino acid-transporter 1(aat-1) gene. Ameliorating mitochondrial ROS formation or impairment ofaat-1-expression abolishes GlcN-mediated life span extension in an NRF2/SKN-1-dependent fashion. Unlike other calorie restriction mimetics, such as 2-deoxyglucose, GlcN extends life span of ageing C57BL/6 mice, which show an induction of mitochondrial biogenesis, lowered blood glucose levels, enhanced expression of several murine amino-acid transporters, as well as increased amino-acid catabolism. Taken together, we provide evidence that GlcN extends life span in evolutionary distinct species by mimicking a low-carbohydrate diet.”
Image source. It appears that average lifespan extension for mice is about 8 weeks or 6%
From the article: “Discussion: The current findings indicate that GlcN at pharmacologically relevant concentrations is capable of extending life span in C. elegans and ageing mice. This appears to be a result of decreased glycolysis and a compensatory increase of amino-acid turnover. Although it should be noted that GlcN may impact hexosamine metabolism34, we did not, however, detect any differences in downstream metabolites of GlcN-6-phosphate in mice, nor did the disruption of the hexosamine pathway prevent GlcN from extending life span in worms, together strongly suggesting that hexosamine metabolism has no bearing on the phenotypes observed. Mechanistically, the life span-extending phenotype is rather linked to an activation of SKN-1/NRF-2-dependent transcription, as the latter not only promotes longevity of C. elegans independently of GlcN16,32,38,39,40,41,42 but also following GlcN exposure (this study). In this regard, it is interesting to note that the aat-1 gene contains a proximally located SKN-1-binding site and that GlcN exposure induces aat-1 ranscription through SKN-1. This notion is supported by the observations that impairment of either skn-1 or aat-1 abrogates the effects of GlcN on life span and disruption of SKN-1 prohibits the induction of aat-1 expression in nematodes. Interestingly, this upregulation of amino-acid transporters, as well as increased L-leucine/L-isoleucine metabolism, is found in GlcN-exposed liver specimen of rodents, thereby suggesting that the glycolytic inhibitor GlcN induces a metabolic switch towards increased protein metabolism in both worms and mice, unambiguously culminating in extended life span. — Although the specific cause(s) of death of individual mice have not been determined in the current study, it will be interesting to see whether and how GlcN affects not only glucose metabolism, but also cancer growth in chronically supplemented mice, as a number of studies insinuate that GlcN effectively reduces cancer cell proliferation43,44,45,46,47,48. — Unlike for DOG and most other life span-extending compounds, extensive published evidence indicates that GlcN is safe for human use even at high doses, making it readily available for interventions to extend human healthspan particularly because, on an observational and uncontrolled basis, it has been repeatedly suggested that supplementation with GlcN may decrease overall mortality in humans49,50.”
Here are some technical highlights from the paper as summarized by Jim Watson:
1. Glc-N: a Glycolysis inhibitor – The mechanism by which D-Glucosamine inhibits glycolysis is by inhibiting hexokinase (or the liver analog, glucokinase). This impairs glucose metabolism and results in the cell “switching” over to amino acid metabolism to produce energy. Thus D-Glucosamine is a “metabolic switch” that converts “Warburg-type metabolism” in the cell (i.e. aerobic glycolysis in the presence of adequate oxygen) over to “protein metabolism”. 100 microM of Glc-N results in a 43% inhibition of glucose oxidation. I don’t know if these levels are achievable with most oral Glc-N supplements.
On the other hand, 2-Deoxyglucose reduces glucose oxidation by over 90%. However. 2DG has been shown to reduce lifespan in rodents although it increases lifespan in nematoides. Glc-N does not have such a negative effect.
- Glc-N: an AMPK activator – The mechanism by which glycolysis inhibition produces longevity is via AMPK(ref). AMPK is an “Energy sensing enzyme” that is activated when the cells energy levelsare low (I.e. Low ATP, high ADP and AMP). Glc-N therapy results in an ATP deficiency which then activates the regulatory unit of AMPK in the nematode, which is AAK-2. AAK-2 is activated with Glc-N treatment. AMPK is the main “on switch” for mitochondrial biogenesis. It turns on over 1500 nuclear encoded genes via a gene activating Cofactor called PGC-1a in the nucleus of the cell. Also, AMPK is a powerful autophagy activator.
3. Glc-N: mechanism is not significantly dependent on Sirtuins – They used the Glc-N on a Sir-2 deficient strain of nematodes as well. There was a reduced effect that did not change the lifespan of the nematodes in these Sir-2 deficient worms. This suggests that the mechanism of action of Glc-N is independent of nuclear levels of NAD+.
4. The mitochondrial biogenesis was due to AMPK – This is not surprising, since AMPK is the primary activator of PGC-1a. However, the article mentions that mitochondrial ROS generation transiently increased for 7 days after Glc-N treatment, but then normalized when the antioxidant enzymes MnSOD and catalase were unregulated.
This suggests that there is an increase in ROS-activated antioxidant gene expression, which probably is due to an Nrf2/Keap dependent mechanism. We have discussed NRF2/Keap extensively in this blog (ref)(ref)(ref).
5. Antioxidants prevent Glc-N-mediated lifespan extension – this is yet another added piece of evidence to the mounting pile of evidence that exogenous antioxidants can be bad for us. They used N-acetylcystein and butylated hydroxylanisole. I think this is an important “take-home message” that we must get out.
6. Mitohormesis – Transient Increase in ROS required for producing oxidative stress-resistant worms. This I believe is one of the most important lessons to learn from this study. Rarely do you see scientists outside the hormesis community who use the word “mitohormesis“. This is good to see. However, I must qualify this by saying that the “upstream issues” that produce the mitohormesis are sill glycolysis inhibition and AMPK activation. Mitohormesis is “downstream event” in this cascade, not the “upstream event”.
7. MAPK pathways and Nrf2 pathways are both important in mediating the lifespan lengthening effects of Glc-N. This again goes back to what we have discussed many times. Specifically, the p38/PMK-1 version of the MAPK pathway is activated with Glc-N treatment. This is a ROS-dependent activation. Likewise, the Nrf2 pathway activation is obviously ROS-dependent as well.
8. Glc-N intake in humans does not increase blood glucose –This is good news, but still puzzling. I would have expected that the inhibition of glycolysis would have raised blood glucose (via TORC2 inhibition)
9. SKN-1: the transcription factor that turns on 36% of the genes affected by GlcN.The paper mentions that when they analyzed promoter sites ofr GlcN-dependent genes, SKN-1 was the activator for 36% of the genes. We recall that this result is for nematodes and SKN-1 is the nematode counterpart of NRF2 in mammals.
10. N-acetyl-glucosamine has a different mechanism of action from D-glucosamine – The last paragraph in the discussion section of this paper mentions that another group has shown a lifespan extending effect of N-acetyl-glucosamine, but the mechanism of life span extension is by inhibiting the hexosamine pathway, not via AMPK.
11. Glc-N is safe, unlike 2-Deoxyglucose – 2DG has a lot of side effects. So does Metformin and Rapamycin. For this reason, GlcN may be a much better compound for humans for lifespan extension.
Glucosamine may also be the basis of anti-cancer therapies
One mechanism may be down-regulation of expression of the heat shock survival protein HIF-1α in cancer cells. The 2014 publication Short-term treatment with glucosamine hydrochloride specifically downregulates hypoxia-inducible factor-1α at the protein level in YD-8 human tongue cancer cells reports: “Hypoxia-inducible factor-1 (HIF-1) is a tumor angiogenic transcription factor composed of an α and β subunit. We investigated the effect of glucosamine hydrochloride (GS-HCl) on the expression of HIF-1α and HIF-1β in serumtreated YD-8 human tongue cancer cells. While long-term (24 h) treatment with GS-HCl strongly repressed the expression of HIF-1α and HIF-1β at both the protein and mRNA levels, short-term (4 h) GS-HCl treatment inhibited HIF-1α at the protein level. Short-term GS-HCl treatment also decreased phosphorylation of p70S6K and S6, translation-related proteins. However, the results of subsequent pharmacological inhibition and protein stability analyses indicated that HIF-1α protein downregulation induced by short-term GS-HCl treatment was not through modulation of the mTOR/p70S6K/S6 signaling pathways, the 26S proteasomal and lysosomal activities and HIF-1α protein stability. Importantly, our further analyses identified that HIF-1α protein downregulation induced by short-term GS-HCl treatment was blunted by exogenous administration of the citric acid cycle metabolites citrate and 2-oxoglutarate, but not the glycolytic end byproducts pyruvate and lactate. These findings demonstrate firstly that short-term GS treatment selectively downregulates HIF-1α at the protein level in YD-8 cells via interference of production of the citric acid cycle metabolites. It is proposed that short-term GS-HCl exposure may be applied for the treatment of oral tumors with high expression of HIF-1α.”
Anti-cancer effects of glucosmine have been long-noted. The 1969 publication d-Glucosamine and Other Sugar Analogs on the Viability and Transplantability of Ascites Tumor Cells reported in summary: “d-Glucosamine has been shown to have a powerful cytotoxic effect on various ascites tumor lines, resulting in a decrease in viability and transplantability of the neoplastic cells. The toxic effect of glucosamine was not significantly altered by addition of either glucose or pyruvate. The effects of d-glucosamine and other sugar analogs were evident histologically even before the cells became stainable by trypan blue.”
The 2007 publication d-Glucosamine inhibits proliferation of human cancer cells through inhibition of p70S6Kreports: “Although d-glucosamine has been reported as an inhibitor of tumor growth both in vivo and in vitro, the mechanism for the anticancer effect of d-glucosamine is still unclear. Since there are several reports suggesting d-glucosamine inhibits protein synthesis, we examined whether d-glucosamine affects p70S6K activity, an important signaling molecule involved in protein translation. In the present study, we found d-glucosamine inhibited the activity of p70S6K and the proliferation of DU145 prostate cancer cells and MDA-MB-231 breast cancer cells. d-Glucosamine decreased phosphorylation of p70S6K, and its downstream substrates RPS6, and eIF-4B, but not mTOR and 4EBP1 in DU145 cells, suggesting that d-glucosamine induced inhibition of p70S6K is not through the inhibition of mTOR. In addition, d-glucosamine enhanced the growth inhibitory effects of rapamycin, a specific inhibitor of mTOR. These findings suggest that d-glucosamine can inhibit growth of cancer cells through dephosphorylation of p70S6K.”
Other publications relating glucosamine to cancers include:
Glucosamine induces cell death via autophagy in some cancer cell lines.
The research literature also indicates that glucosamine induces autophagy, a process of cellular house cleaning extremely important for health that we have discussed previously(ref)(ref). This is not surprising since AMPK is a powerful autophagy activator.
One is the 2010 article Glucosamine induces autophagic cell death through the stimulation of ER stress in human glioma cancer cells“Autophagy can promote cell survival or death, but the molecular basis of its dual role in cancer is not well understood. Here, we report that glucosamine induces autophagic cell death through the stimulation of endoplasmic reticulum (ER) stress in U87MG human glioma cancer cells. Treatment with glucosamine reduced cell viability and increased the expression of LC3 II and GFP-LC3 fluorescence puncta, which are indicative of autophagic cell death. The glucosamine-mediated suppression of cell viability was reversed by treatment with an autophagy inhibitor, 3-MA, and interfering RNA against Atg5. Glucosamine-induced ER stress was manifested by the induction of BiP, IRE1α, and phospho-eIF2α expression. Chemical chaperon 4-PBA reduced ER stress and thereby inhibited glucosamine-induced autophagic cell death. Taken together, our data suggest that glucosamine induces autophagic cell death by inducing ER stress in U87MG glioma cancer cells and provide new insight into the potential anticancer properties of glucosamine.”
Another relevant article just appeared a few days ago (June 2014) Enhanced expression of proapoptotic and autophagic proteins involved in the cell death of glioblastoma induced by synthetic glycans, Further this article suggests that glucosamine could possibly serve as a therapy for the deadly and incurable brain cancer glioblastoma, “OBJECT – Glioblastoma is the most aggressive malignant brain tumor, and overall patient survival has not been prolonged even by conventional therapies. Previously, the authors found that chemically synthesized glycans could be anticancer agents against growth of a series of cancer cells. In this study, the authors examined the effects of glycans on the growth of glioblastoma cells both in vitro and in vivo. METHODS – The authors investigated not only the occurrence of changes in the cell signaling molecules and expression levels of various proteins related to cell death, but also a mouse model involving the injection of glioblastoma cells following the administration of synthetic glycans. RESULTS – Synthetic glycans inhibited the growth of glioblastoma cells, induced the apoptosis of the cells with cleaved poly (adenosine diphosphate-ribose) polymerase (PARP) expression and DNA fragmentation, and also caused autophagy, as shown by the detection of autophagosome proteins and monodansylcadaverine staining. Furthermore, tumor growth in the in vivo mouse model was significantly inhibited. A dramatic induction of programmed cell death was found in glioblastoma cells after treatment with synthetic glycans. CONCLUSIONS – These results suggest that synthetic glycans could be a promising novel anticancer agent for performing chemotherapy against glioblastoma.”
Epidemiological studies
The bottom line is that glucosamine supplementation lowers human mortality
Glucosamine and Chondroitin Sulfate have already been shown in a large epidemiological study to lower overall mortality and reduce cancer risks – a 5-year study of 77,719 elderly residents of Washington State. We found several publications based on different analyses of data based on this population.
The 2010 publication Total mortality risk in relation to use of less-common dietary supplements reported: “Background: Dietary supplement use is common in older US adults; however, data on health risks and benefits are lacking for a number of supplements. Objective: We evaluated whether 10-y average intakes of 13 vitamin and mineral supplements and glucosamine, chondroitin, saw palmetto, Ginko biloba, garlic, fish-oil, and fiber supplements were associated with total mortality. Design: We conducted a prospective cohort study of Washington State residents aged 50–76 y during 2000–2002. Participants (n= 77,719) were followed for mortality for an average of 5 y. Results:A total of 3577 deaths occurred during 387,801 person-years of follow-up. None of the vitamin or mineral 10-y average intakes were associated with total mortality. Among the nonvitamin-nonmineral supplements, only glucosamine and chondroitin were associated with total mortality. The hazard ratio (HR) when persons with a high intake of supplements (≥4 d/wk for ≥3 y) were compared with nonusers was 0.83 (95% CI: 0.72, 0.97;Pfor trend = 0.009) for glucosamine and 0.83 (95% CI: 0.69, 1.00;Pfor trend = 0.011) for chondroitin. There was also a suggestion of a decreased risk of total mortality associated with a high intake of fish-oil supplements (HR: 0.83; 95% CI: 0.70, 1.00), but the test for trend was not statistically significant. Conclusions:For most of the supplements we examined, there was no association with total mortality. Use of glucosamine and use of chondroitin were each associated with decreased mortality.”
The reported hazard ratio for mortality reduction associated with GlcN consumption is reduction: 0.83. This means that the probability of death for those consuming glucosamine per unit time is 83% of that of controls. In my mind this is impressive
The second publication related to the same epidemiological study is from 2012: Use of glucosamine and chondroitin in relation to mortality: “Glucosamine and chondroitin are products commonly used by older adults in the US and Europe. There is limited evidence that they have anti-inflammatory properties, which could provide risk reduction of several diseases. However, data on their long-term health effects is lacking. To evaluate whether use of glucosamine and chondroitin are associated with cause-specific and total mortality. Participants (n = 77,510) were members of a cohort study of Washington State (US) residents aged 50–76 years who entered the cohort in 2000–2002 by completing a baseline questionnaire that included questions on glucosamine and chondroitin use. Participants were followed for mortality through 2008 (n = 5,362 deaths). Hazard ratios (HR) for death adjusted for multiple covariates were estimated using Cox models. Current (baseline) glucosamine and chondroitin use were associated with a decreased risk of total mortality compared to never use. The adjusted HR associated with current use of glucosamine (with or without chondroitin) was 0.82 (95 % CI 0.75–0.90) and 0.86 (95 % CI 0.78–0.96) for chondroitin (included in two-thirds of glucosamine supplements). Current use of glucosamine was associated with a significant decreased risk of death from cancer (HR 0.87 95% CI 0.76–0.98) and with a large risk reduction for death from respiratory diseases (HR 0.59 95% CI 0.41–0.83). Use of glucosamine with or without chondroitin was associated with reduced total mortality and with reductions of several broad causes of death. Although bias cannot be ruled out, these results suggest that glucosamine may provide some mortality benefit.”
This time the hazard ratio for mortality reduction is .82: 18% fewer deaths per unit of time for those consuming the supplement.
Both of these papers were done on the same group of 77,719 people. We note that this is as great an effect as combining vegan diet and fish consumption! We find this quite surprising. Glucosamine appears to have a comparable or greater effect on mortality reduction and lifespan extension than Meformin, Rapamycin, 2DG, Veganism, and Resveratrol in nematodes and rodents.
Going further based on the same population, a 2013 publication looked at the epidemiological consequences of glucosamine consumption on risk of incurring correctal cancer. Use of glucosamine and chondroitin supplements and risk of colorectal cancer. “PURPOSE: Glucosamine and chondroitin are non-vitamin, non-mineral supplements which have anti-inflammatory properties. These supplements are typically used for joint pain and osteoarthritis and are commonly taken as either glucosamine alone or glucosamine plus chondroitin. An exploratory analysis conducted within the VITamins And Lifestyle (VITAL) study observed any use of glucosamine and chondroitin to be associated with reduced risk of colorectal cancer (CRC) after 5 years of follow-up. METHODS: With two additional years of follow-up, we have studied these associations in greater depth, including associations by frequency/duration of use and by formulation, and have evaluated whether observed associations are modified by factors associated with inflammation. Participants include 75,137 western Washington residents aged 50-76 who completed the mailed VITAL questionnaire between 2000 and 2002. Use of glucosamine and chondroitin was ascertained by questions about supplement use during the 10-year period prior to baseline, and participants were followed for CRC through 2008 (n = 557). Cox regression was used to estimate hazard ratios (HRs) and 95 % confidence intervals (CIs). RESULTS: Persons reporting use of glucosamine + chondroitin on 4+ days/week for 3+ years had a non-statistically significant 45 % lower CRC risk than non-users (HR: 0.55; 95 % CI 0.30-1.01; p-trend: 0.16). This association varied by body mass index (p-interaction: 0.006), with inverse association observed among the overweight/obese (p-trend: 0.02), but not among the underweight/normal weight. Use of glucosamine alone was not significantly associated with CRC risk. CONCLUSIONS: There is great need to identify safe and effective cancer preventive strategies, suggesting that glucosamine and chondroitin may merit further attention as a potential chemopreventive agent.”
Again, we are impressed by the 45% risk reduction for CRC.
Epidemiological evidence exists that glucosamine and chondroitin supplementation reduces inflammatory biomarkers.
The recent April 2014 publication Associations Between Glucosamine and Chondroitin Supplement Use and Biomarkers of Systemic Inflammation looks at the same population from a different viewpoint and reports: “Objectives: Glucosamine and chondroitin supplements have been shown to have anti-inflammatory properties in both in vitro studies and animal models; however, little is known about these relationships in humans. The VITamins and Lifestyle (VITAL) biomarker study evaluated the associations between use of these supplements and a panel of circulating inflammatory biomarkers. Design: Study participants included 217 men and women age 50-75 years living in the Seattle metropolitan area. Use of glucosamine and chondroitin supplements was ascertained by home interview/supplement inventory. Inflammation was assessed by using blood and urine collected at the time of home interview. Measures of systemic inflammation included plasma high-sensitivity C-reactive protein (hsCRP), interleukin (IL)-1β, IL-6, IL-8, tumor necrosis factor (TNF)-α, soluble TNF receptors I and II, and urinary prostaglandin E2-metabolite (PGE-M). Multivariate-adjusted linear regression was used to evaluate the associations between supplement use and biomarkers of inflammation. Results: High users (14 or more pills/week) of chondroitin had 36% lower hsCRP (ratio, 0.64; 95% confidence interval [CI], 0.39-1.04; p for trend=.03) and 27% lower PGE-M (ratio, 0.73; 95% CI, 0.5-0.98; p for trend=.07) than nonusers. Compared with nonusers, high users of glucosamine had 28% lower hsCRP (ratio, 0.72; 95% CI, 0.47-1.08; p for trend=.09) and 24% lower PGE-M (ratio, 0.76; 95% CI, 0.59-0.97; p for trend=0.10). Use of glucosamineand chondroitin supplements was not associated with the other markers of inflammation. Conclusions: These results support prior research suggesting that use of glucosamine and chondroitin is associated with reduced hsCRP and PGE2, but further work is needed to more definitively evaluate the anti-inflammatory potential of these supplements.”
Glucosamine supplementation may be protective against lung cancer.
Data from the same Washington State cohort also provided the basis for the 2011 publication Use of glucosamineand chondroitin and lung cancer risk in the VITamins And Lifestyle (VITAL) cohort. “OBJECTIVE: Inflammation plays an important role in lung carcinogenesis. Epidemiologic studies have reported inverse associations of non-steroidal anti-inflammatory drug (NSAID) use and lungcancerrisk. Previously, we found that ever use ofglucosamineand chondroitin, which have anti-inflammatory properties, were inversely associated with lungcancerrisk. After an additional year of follow-up, we further examined the association including frequency/duration of use, interaction with factors associated with inflammation, and lungcancerhistology. METHODS: Participants were members of the VITamins And Lifestyle cohort. Adults, aged 50-76 years, who were residents of western Washington State, completed a baseline questionnaire in 2000-2002 (n = 76,904). Participants were queried on their use of glucosamine and chondroitin, over the 10 years prior to baseline, and categorized as nonuser, low use < 4 days/week or < 3 years, or high use ≥ 4 days/week and ≥ 3 years. Lung ases (n = 808) were ascertained through linkage to the Surveillance, Epidemiology, and End Resultscancer registry. RESULTS: High 10-year use ofglucosamine[hazard ratio (HR), 0.77; 95% CI: 0.56-1.07; p trend = 0.04] but not chondroitin was associated with a reduction in lungcancer risk. The association with glucosamine was limited to adenocarcinoma (HR, 0.49; 95% CI: 0.27-0.90; p trend <0.01) and was not modified by NSAID use or smoking status. CONCLUSIONS: Our results for glucosamineuse are similar to the prior human studies of NSAID use and lungcancer, both in magnitude and the limitation of the association to adenocarcinoma. Unlike NSAIDs, glucosaminehas no known adverse effects. Although confirmatory studies are needed, glucosamine is an attractive candidate for lung cancer chemoprevention.
It is interesting to note that in all of these studies related to the beneficial effects of glucosamine on mortality and cancers, the VITAL cohort consists of older people between 50 and 76.
The effectiveness of glucosamine for treating osteoarthritis remains a matter of controversy.
While the evidence for glucosamine being life-extending and cancer preventing, earlier studies relating to its effectiveness in treating osteoarthritis have been challenged. The bottom-line on the effectiveness of this approach – the main reason why millions have been taking the supplement – appears to be not really in. The 2014 publication Glucosamine for Osteoarthritis: Biological Effects, Clinical Efficacy, and Safety on Glucose Metabolism reports: “Osteoarthritis is a chronic degenerative disorder that currently represents one of the main causes of disability within the elderly population and an important presenting complaint overall. The pathophysiologic basis of osteoarthritis entails a complex group of interactions among biochemical and mechanical factors that have been better characterized in light of a recent spike in research on the subject. This has led to an ongoing search for ideal therapeutic management schemes for these patients, where glucosamine is one of the most frequently used alternatives worldwide due to their chondroprotective properties and their long-term effects. Its use in the treatment of osteoarthritis is well established; yet despite being considered effective by many research groups, controversy surrounds their true effectiveness. This situation stems from several methodological aspects which hinder appropriate data analysis and comparison in this context, particularly regarding objectives and target variables. Similar difficulties surround the assessment of the potential ability of glucosamine formulations to alter glucose metabolism. Nevertheless, evidence supporting diabetogenesis by glucosamine remains scarce in humans, and to date, this association should be considered only a theoretical possibility.”So, we may have a situation where millions of people who are taking glucosamine is a supplement are probablysignificantly benefiting from it, but they may be taking it for the wrong reason
There may be a downside to taking too much glucosamine
A 2010 publication suggests that taking high doses of glucosamine or prolonged use it glucosamine reduces levels of SIRT1, leads to apoptosis of pancreatic cells and could increase the risk of developing diabetes. Hexosamines stimulate apoptosis by altering T1 action and levels in rodent pancreatic β-cells: “The activity and levels of SIRT1, which promotes cell survival in several models, are linked to glucose concentrations and cellular energy metabolism. The present study aimed to determine whether impaired Sirt1 activity is involved in the induction of apoptosis by the nutrient-sensing hexosamine biosynthesis pathway (HBP). Pancreatic Nit-1, Rin-m5F, and Min6 β-cells were acutely treated at different doses and times with glucosamine, which enters and stimulates the HBP. Sirt1 levels were genetically modulated by retroviral infection. Expression levels, cellular localization, and activity of apoptosis-related markers were determined by qPCR, immunoblotting, and co-immunoprecipitation. Glucosamine treatment dose- and time dependently induced cell apoptosis in all cell lines studied. HBP stimulation time dependently modified SIRT1 protein levels, notably in the cytoplasm. This was concomitant with increased E2F1 binding to the c-myc promoter. In both NIT-1 and min6 β-cells, genetic knockdown of Sirt1 expression resulted in higher susceptibility to HBP-stimulated apoptosis, whereas overexpression of Sirt1 had the opposite impact. These findings indicate that reduction of SIRT1 levels by hexosamines contributes to β-cell apoptosis. Methods to increase SIRT1 levels or activity could thus prevent the decrease in β-cell mass, notably that observed in type 2 diabetes.”
Relative to this issue, the 2014 publication 17A-Estradiol protects against glucosamine-induced pancreatic A-cell dysfunction concludes “: GlcN impairs insulin secretion of [beta]-cells by inhibiting Ca2+ influx and enhancing [beta]-cell apoptosis with increases in ER stress-related proteins, whereas E2 counters these adverse effects of GlcN.”
Glucosamine supplementation and other research approaches to mitochondrial health
There has been a wealth of research going on recently related to mitochondrial health and interventions that could enhance it. To put the above results related to glucosamine in context, I comment very briefly here on some of the “traditional” and “newer” health supplements and interventions intended to impact on the mitochondria.
Glucosamine – activates AMPK, and NRF2, enhances amino acid transport, inhibits glycolysis, mitigates against Warburg, activates autophagy – as above.
Co-enzyme Q10 (ubiquinone)- A powerful mitochondrial endogenous antioxidant. Extremely popular as a dietary supplement, often prescribed, consumed by millions, a $700 million worldwide business. Thought to reduce excess mitochondrial ROS. But currently there is much skepticism as to effectiveness of exogenous administration because of very limited bioavailability (ref)(ref)
Melatonin – This is a circadian-activated hormone that exercises multiple regulatory functions over metabolism. Found with high concentration in mitochondria, it is known primarily for its antioxidant properties. Thought to increase OXPHOS efficiency, reduce mitochondrial membrane potential and mildly promote glycolysis, it transiently raises not only the expression of neuronal NOS mRNA, nNOS protein synthesis and the nitric oxide oxidation products, nitrite and nitrate(ref)(ref).
Acetyl-l-carnitine and alpha-lipoic acid. The Juvenon combination of bioavailable antioxidants is purported to cross the blood-brain barrier and penetrate the mitochondrial membranes. Presumably, they function as antioxidants to reduce excessive ROS produced by old and inefficient mitochondria, and reduce excess mitochondrial membrane polarization(ref).
PQQ – This is a redox cofactor that activates PGC1alpha and the mitochondrial sirtuin SIRT3. Above, we mentioned how PGC1alpha is essential for mitochondrial biogenesis(ref).
Exercise – activates AMPK, PGC1alpha, mitochondrial biogenesis, and autophagy(ref)(ref).
Calorie restriction – Calorie restriction in this various forms, including intermittent fasting, upgrades expression of AMPK with its downstream benefits on mitochondria such as those described above including mitochondrial biogenesis(ref)(ref). Both exercise and diet regulate expression of sirtuins, particularly SIRT1 and SIRT3. SIRT3 is a mitochondrial protein important for deacelating acetyllysine-modified proteins in mitochondria.
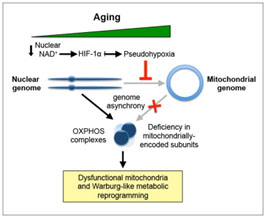
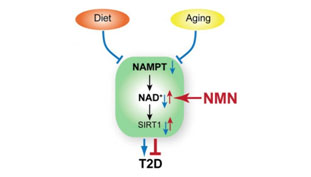
NMN (Nicotinamide Mononucleotide) – Assuring adequate levels of nuclear NAD+ is essential for the prevention of a state of pseudo-hypoxia in the nucleus with the result being an adequate generation of mitochondrial proteins essential for averting cellular relapse into Warburg–type metabolism and a number of associated age-related problems such as hearing loss(ref). DNA repair machinery (PARPs) also draws down on NAD+ and it is therefore in short supply with aging. NAD+ is a cofactor for both Sirtuins(and PARPs). There are 7 Sirtuins and over 30 different PARPs. NAD+ is an “on switch” for part of the mitochondrial encoded genes required for Complex II and Complex IV in the mitochondrial electron transfer chain. One thought is that supplementation with NMN could enhance production of NAD+ and be very useful both for DNA repair and mitochondrial health, But this is an extremely expensive substance at the present time(ref)(ref). Our blog was one of the first to suggest NMN supplementation in the 2012 entry by Victor Circadian Regulation,NMN, Preventing Diabetes, and Longevity There is a great deal of current research in this area.
Image source Image source NMN promotes nuclear NAD+ which upregulates expression of SIRT1 averting the Warburg effect and Type 2 diabetes.
NR (nicotinamide riboside) – This is a readily available supplement that is also sought possibly to be an effective promoter of nuclear NAD+. Actually. NR is the intermediary in the NMN inducement of NAD+ (ref)(ref)
Resveratrol – promotes expression of SIRT1 needed for mitochondrial health and the mitochondrial proteins SIRT3-4-5. Further, it is an activator of AMPK and autophagy(ref)(ref).
Centrophenoxine (meclophenoxate) – this is an older drug still used in Europe to treat forms of dementia. About half the research publications on it were published prior to 1980(ref) and my impression is that few researchers have seriously looked at it from a contemporary understanding of mitochondria and molecular biology. So, while it is known to have major impacts on cell metabolism, exactly how it works with respect to mitochondria is unclear to me. Among its impacts (disputed) is clearing of lipofuscin from cells(ref)(ref)(ref). This might have a positive impact on mitophagy.
C60 fullerenes in olive oil – functions as antioxidant to reduce excessive ROS produced by old and inefficient mitochondria, reduces excess mitochondrial membrane polarization(ref)(ref).
MitoQ – Co-enzyme Q-10 molecularly modified by adding a charged anion designed to facilitate it penetrating the mitochondrial membrane. Functions as antioxidant to reduce excessive ROS produced by old and inefficient mitochondria, reduces excess membrane polarization(ref)(ref).
SkQ -This list is not exhaustive; other supplements have been researched such as SkQ, like MitoQ, a charged antioxidant molecule designed so it can effectively traverse the mitochondrial membrane(ref)
We seriously question whether certain of these approaches are compatible. Specifically the demonstrated results with glucosamine suggest that ROS plays a very important signaling function in achieving health and longevity benefits via mitohormesis. Therefore, the exogenously administered antioxidants could interfere with such signaling and produce negative impacts for normal healthy people. On the other hand, strong mitochondrial antioxidants could be useful for people whose mitochondria are old and leaky and are already heavily spinning out ROS.
The purpose of mentioning these alternative approaches to mitochondrial health is not to compare their efficacy or characterize what they really do. It is to establish a single point: their mechanisms of operation appear to be very different. Therefore we wonder about which approaches are effective under what conditions, which approaches are compatible, and whether adopting multiple approaches to mitochondrial health might produce benefits that are additive or even possibly multiplicative for healthy people. We intend to explore the rich possibilities inherent in this question in subsequent blog entries.


To add to the complexity of the resveratrol picture as an anti-oxidant: It’s now implicated in increasing klotho levels in the kidney.
Resveratrol increases anti-aging Klotho gene expression via the activating transcription factor 3/c-Jun complex-mediated signaling pathway (2014) http://www.ncbi.nlm.nih.gov/pubmed/24911970
The Kidney Is the Principal Organ Mediating Klotho Effects (2014) http://www.ncbi.nlm.nih.gov/pubmed/24854271
I know anti-agingfirewalls has touched on hydrogen sulfide before, but it appears it may also influence Klotho: Hydrogen sulfide, the next potent preventive and therapeutic agent in aging and age-associated diseases (2013) http://www.ncbi.nlm.nih.gov/pubmed/23297346
jz99:
Excellent points. I have a draft update of my old blog entry on Klotho in the works, and will be sure to include this resveratrol effect and these citations.
As to hydrogen sulfide also, alas, Jim Watson and I have had many discussions and e-mail discussions about its hormetic actions as well as those of CO and NO, but only a little of this has found its way into blogs so far. We call them “The Great God Gasses” and they too deserve more attention in the blog than they have gotten so far. Jim and I have a long draft entry on them that somehow got sidetracked months ago.
Consisting with our notion that neither biology nor aging has a center, it is interesting that other health/longevity interventions upgrade expression of klotho, once thought to be a stand-alone pathway.
Vince
As usual, very good post.
I’ve seen that most of the formulations do glucosamine are in the glucosamine sulfate potassium chloride form, and many of them contain MSM, which might have other effects in its own.
Best
Recorte:
Good observations. Because the literature does not generally speak to specific impacts of the chemical compounds I did not cover this. I tried to stick to d- glucosamine in the blog, though some of the results are for supplements which also contain MSM or chondratin. I don’t think the main results are confounded.
Vince
This is not the first time I have read about glucosamine and it’s poitive effect on longevity/cancer prevention. There are a number of articles that have appeared over the past 5 years pertaining to this.
Here are a couple that people may well be interested in reading.
http://www.ncbi.nlm.nih.gov/pubmed/19744341
http://www.ncbi.nlm.nih.gov/pubmed/19423520
As stated in the blog, the effectiveness of glucosamine and co. on joint health is highly questionable – but it seems clear the mentioned supplements do have positive effects on one’s health in totally different areas than what they are marketed for.
Thanks for another great article.
“this is yet another added piece of evidence to the mounting pile of evidence that exogenous antioxidants can be bad for us . . . I think this is an important “take-home message” that we must get out.”
What is that message that must get out? Cut out all supplemental antioxidants? Or consume them, but only randomly? I’ve cut most supplements out altogether. I still take a few (curcumin, SGS), but only every other day, or every third day. No science to back this up, but just trying to avoid sending a continuous signal with doses that are higher than found in food.
Thanks for a great article Vince!
Based on the above considerations, what would your current recommendations as to the form and dosages of glucosamine (sulfate or other form?) and other agents mentioned in the blog. How much of and on what schedule are you taking these, and which are you currently avoiding or limiting? There is expressed above both a question of whether oral doses can achieve effects, but yet another section that cautions against too high of a dose. I am also not sure that most glucosamine sulfates on the market specify as the D-form. I assume the opposite form is L- and if so, what effects would this or a mixture have?
Pingback: Glucosamine For Dogs Scientific Evidence Of Life After Death | glucosamine for dogs
Pingback: Glucosamine & Chondroitin 500/400mg Revolution | glucosamine for dogs
I’m a bit confused; article states:
“5. Antioxidants prevent Glc-N-mediated lifespan extension – this is yet another added piece of evidence to the mounting pile of evidence that exogenous antioxidants can be bad for us. They used N-acetylcystein and butylated hydroxylanisole. I think this is an important “take-home message” that we must get out.”
But Vincent you include in your firewalls NAC … so is this not ‘mediating’ the lifespan extension you would hope to gain from the glucosamine?
Thank you,
cam