By Vince Giuliano
This Part 3 of the Inflammation series of blog entries is concerned with the all-important resolution phase of inflammation, how acute inflammation goes away under ideal conditions instead of hunkering down to lingering and dangerous chronic inflammation. It is concerned with recently identified substances found in fish and flaxseed oils that play important roles in resolving certain kinds of inflammation – what they can do and how they work. Part 1 of the series, already published, is the same as Part 5 of the NAD world. That blog entry is concerned with The pro-inflammatory effects of eNAMPT(extracellular NAMPT, nicotinamide phosphoribosyltransferase). Part 2 relates 1) the “master” pathway network of inflammation (NF-kB) to two other pathway networks clearly implicated in aging and disease processes, 2) Genomic Instability (p53), and 3) Oxidative stress (Nrf2). Part 4 of the inflammation series is concerned with an amazingly effective new approach for averting the biggest killers of older people, atherosclerosis and heart disease – that being inhibition of the PCSK9 protein. Other blog entries in this series will be forthcoming.
A little additional background on the stages of inflammation
Part 2 includes a general introduction to inflammation and to chronic (non-resolving) inflammation – the nemesis of healthy aging and core factor of aging-related diseases. As pointed out there, inflammation is a natural defensive body process that evolved over millions of years, It is a very good thing; we could not live without it. On the other hand, if inflammation sets into place and does not clear up, there can be numerous negative consequences. Chronic inflammation appears to be both a consequence and cause of also almost all aging-related diseases including heart disease, arthritis, cancer, diabetes, osteoporosis, Alzheimer’s and other neurodegenerative diseases, inflammatory bowel disease, asthma, aging, obesity, sinusitis, COPD, atherosclerosis, periodontal disease, blisters, depression, lethargy, fatigue, etc.

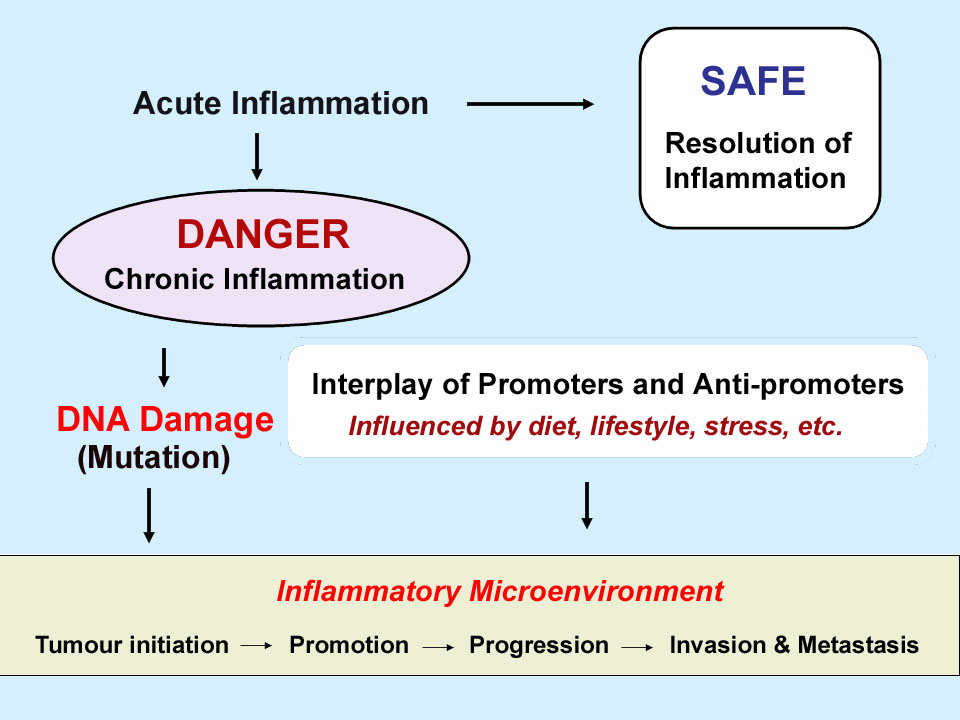
The stages of inflammation
On the most macroscopic level, acute inflammation involves a well-defined and articulated sequence of cellular migrations and physical changes within living tissue. Inflammation is evolutionarily a very ancient defensive response.
- Is normally Initiated by trauma which may be of many kinds including viral and bacterial infection, physical injury to tissues, insect bites, burns, radiation injuries and toxins.
- Manifests the four classical indicators known in Latin since Roman times as rubor (redness), calor (fever), tumor (swelling) and dolor (pain). The symptoms manifested by inflammation include swelling, tender joints, sore throat, rash, runny nose, blisters, bleeding gums, inability to move or exercise, depression, lethargy, fatigue, etc.
- Terminates in Resolution, including repair, regeneration, and remodeling of affected tissues. That is, except when it does not terminate and become chronic inflammation.
Details as to what happens in each of these stages can be very complex and vary widely depending on the nature and duration of the trauma, the tissue or organ involved, the state of the organism, diet, sleep and multiple environmental factors. Traditionally, understanding of inflammation and control of inflammation was focused on the Initiation and Manifestation stages. By the late 1990s, however, focus was shifting to the importance of the resolution stage. And, knowledge was being gained about how mediators might be invoked to assure resolution of inflammation in certain diseases. That is the focus of this blog entry.
A. LIPID MEDIATORS OF INFLAMMATION
The 2009 publication Resolvins and protectins:mediating solutions to inflammation described the overall situation: ”Resolution of inflammation has historically been viewed as a passive process, occurring as a result of the withdrawal of pro-inflammatory signals, including lipid mediators such as leukotrienes and prostaglandins. Thus, most anti-inflammatory drugs have traditionally targeted primarily mediator pathways that are engaged at the onset of inflammation. Only recently has it been established that inflammation resolution is an active process with a distinct set of chemical mediators. Several clinical and epidemiological studies have identified beneficial effects of polyunsaturated fatty acids (PUFAs) for a variety of inflammatory diseases, yet without mechanistic explanations for these beneficial effects. Resolvins and protectins are recently identified molecules that are generated from omega-3 PUFA precursors and can orchestrate the timely resolution of inflammation in model systems. Dysregulation of pro-resolving mediators is associated with diseases of prolonged inflammation, so designing pharmacological mimetics of naturally occurring pro-resolving mediators offers exciting new targets for drug design. This review describes the discovery and synthesis of these novel lipid mediators, their receptors and mechanisms of action, and summarizes the studies to date that have uncovered roles for resolvins and protectins in disease states.”
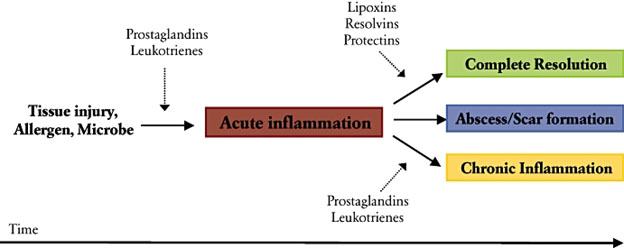
Further detail is found in the 2000 publication Natural resolution of inflammation. “Inflammation is a protective response essential for maintaining human health and for fighting disease. As an active innate immune reaction to challenge, inflammation gives rise to clinical cardinal signs: rubor, calor, dolor, tumor and functio laesa. Termination of acute inflammation was previously recognized as a passive process; a natural decay of pro-inflammatory signals. We now understand that the natural resolution of inflammation involves well-integrated, active, biochemical programs that return tissues to homeostasis. This review focuses on recent advances in the understanding of the role of endogenous lipid mediators that modulate cellular fate and inflammation. Biosynthesis of eicosanoids and other lipids in exudates coincides with changes in the types of inflammatory cells. Resolution of inflammation is initiated by an active class switch in lipid mediators, such as classic prostaglandins and leukotrienes, to the production of proresolution mediators. Endogenous pro-resolving lipid mediators, including arachidonic acid-derived lipoxins, aspirin-triggered lipoxins, ω3-eicosapentaenoic acid-derived resolvins of the E-series, docosahexaenoic acid-derived resolvins of the D-series, protectins and maresins, are biosynthesized during the resolution phase of acute inflammation. Depending on the type of injury and the type of tissue, the initial cells that respond are polymorphonuclear leukocytes, monocytes/macrophages, epithelial cells or endothelial cells. The selective interaction of specific lipid mediators with G protein-coupled receptors expressed on innate immune cells (e.g. G protein-coupled receptor 32, lipoxin A4 receptor/formyl peptide receptor2, chemokine-like receptor 1, leukotriene B4 receptor type 1 and cabannoid receptor 2) induces cessation of leukocyte infiltration; vascular permeability/edema returns to normal with polymorphonuclear neutrophil death (mostly via apoptosis), the nonphlogistic infiltration of monocyte/macrophages and the removal (by macrophages) of apoptotic polymorphnuclear neutrophils, foreign agents (bacteria) and necrotic debris from the site. While an acute inflammatory response that is resolved in a timely manner prevents tissue injury, inadequate resolution and failure to return tissue to homeostasis results in neutrophil-mediated destruction and chronic inflammation. A better understanding of the complex mechanisms of lipid agonist mediators, cell targets and actions allows us to exploit and develop novel therapeutic strategies to treat human inflammatory diseases, including periodontal diseases.”
The 2014 publication Protectins and maresins: New pro-resolving families of mediators in acute inflammation and resolution bioactive metabolome reports further: “Acute inflammatory responses are protective, yet without timely resolution can lead to chronic inflammation and organ fibrosis. A systems approach to investigate self-limited (self-resolving) inflammatory exudates in mice and structural elucidation uncovered novel resolution phase mediators in vivo that stimulate endogenous resolution mechanisms in inflammation. Resolving inflammatory exudates and human leukocytes utilize DHA and other n-3 EFA to produce three structurally distinct families of potent di- and trihydroxy-containing products, with several stereospecific potent mediators in each family. Given their potent and stereoselective picogram actions, specific members of these new families of mediators from the DHA metabolome were named D-series resolvins (Resolvin D1 to Resolvin D6), protectins (including protectin D1-neuroprotectin D1), and maresins (MaR1 and MaR2). In this review, we focus on a) biosynthesis of protectins and maresins as anti-inflammatory-pro-resolving mediators; b) their complete stereochemical assignments and actions in vivo in disease models. Each pathway involves the biosynthesis of epoxide-containing intermediates produced from hydroperoxy-containing precursors from human leukocytes and within exudates. Also, aspirin triggers an endogenous DHA metabolome that biosynthesizes potent products in inflammatory exudates and human leukocytes, namely aspirin-triggered Neuroprotectin D1/Protectin D1 [AT-(NPD1/PD1)]. Identification and structural elucidation of these new families of bioactive mediators in resolution has opened the possibility of diverse patho-physiologic actions in several processes including infection, inflammatory pain, tissue regeneration, neuroprotection-neurodegenerative disorders, wound healing, and others. This article is part of a Special Issue entitled “Oxygenated metabolism of PUFA: analysis and biological relevance”.
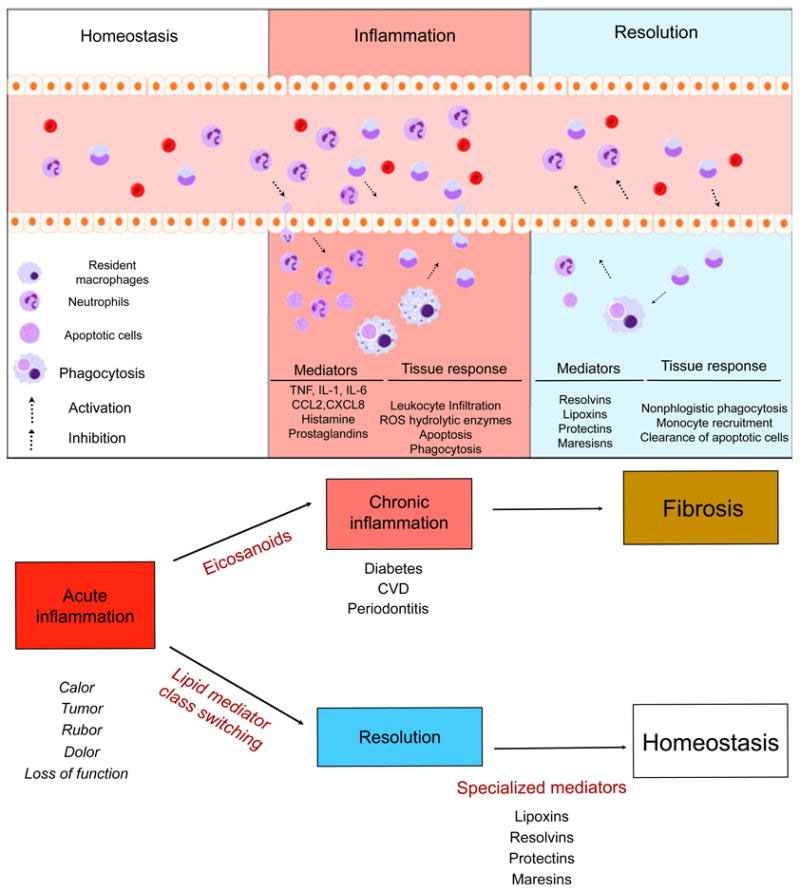
Image and legend source “Natural inflammation resolution response. Acute inflammation is a self-limiting response. In conditions of homeostasis, resident cells maintain normal conditions and remove apoptotic cells and debris. Upon challenge to tissue, the inflammatory response begins. Cell communication mediators activate chemoattraction, vascular permeability and infiltration of leukocytes to the periphery. Microorganisms and dead cells are taken up by phagocytes. The outcome of acute inflammation (chronicity, fibrosis or resolution) is influenced by the type of factors and the degree to which they are involved in the response. Resolution is the re-establishment of normal (homeostasis) and is an actively regulated, well-coordinated process. In resolution, inhibition of leukocyte infiltration, nonphlogistic phagocytosis and vascular and tissue function return to normal.”
The line of reasoning outlined in the above documents has been pursued by many researchers and in specific circumstances since. For example, the 2016 publication DHA- and EPA-derived resolvins, protectins, and maresins in airway inflammation. Reports: “Essential fatty acids can serve as important regulators of inflammation. A new window into mechanisms for the resolution of inflammation was opened with the identification and structural elucidation of mediators derived from these fatty acids with pro-resolving capacity. Inflammation is necessary to ensure the continued health of the organism after an insult or injury; however, unrestrained inflammation can lead to injury “from within” and chronic changes that may prove both morbid and fatal. The resolution phase of inflammation, once thought to be a passive event, is now known to be a highly regulated, active, and complex program that terminates the inflammatory response once the threat has been contained. Specialized pro-resolving mediators (SPMs) are biosynthesized from omega-3 essential fatty acids to resolvins, protectins, and maresins and from omega-6 fatty acids to lipoxins. Through cell-specific actions mediated through select receptors, these SPMs are potent regulators of neutrophil infiltration, cytokine and chemokine production, and clearance of apoptotic neutrophils by macrophages, promoting a return to tissue homeostasis. (Italic for emphasis is mine VG) This process appears to be defective in several common human lung diseases, such as asthma and COPD, which are characterized by chronic unrestrained inflammation and significant associated morbidity. Here, we highlight translational research in animal models of disease and with human subjects that sheds light on this rapidly evolving area of science and review the molecular and cellular components of the resolution of lung inflammation.”
Chronic inflammation may facilitate or further many disease process, for example in cancer:

THE CAST OF CHARACTERS
Much of the story to be told here revolves around major group of bioactive compounds which are natural pro-inflammatory or anti-inflammatory mediators. Many are derivatives from Polyunsaturated Fatty Acids (PUFAs). So here are some of the important characters.
PUFAs – Polyunsaturated Fatty Acids
PUFAs are fatty acids containing more than one double bond in their backbone chemical structure There are a large number of them important in biology, including many implicated in inflammation and its resolution.. Related to inflammation and its resolution, members of the Omega-6 and Omega-3 families are particularly important. This chart is from a Wikipedia article.
Omega-3 Omega-3 fatty acids, polyunsaturated
| Common name | Lipid name | Chemical name |
| Hexadecatrienoic acid (HTA) | 16:3 (n-3) | all-cis 7,10,13-hexadecatrienoic acid |
| Alpha-linolenic acid (ALA) | 18:3 (n-3) | all-cis-9,12,15-octadecatrienoic acid |
| Stearidonic acid (SDA) | 18:4 (n-3) | all-cis-6,9,12,15,-octadecatetraenoic acid |
| Eicosatrienoic acid (ETE) | 20:3 (n-3) | all-cis-11,14,17-eicosatrienoic acid |
| Eicosatetraenoic acid (ETA) | 20:4 (n-3) | all-cis-8,11,14,17-eicosatetraenoic acid |
| Eicosapentaenoic acid (EPA, Timnodonic acid) | 20:5 (n-3) | all-cis-5,8,11,14,17-eicosapentaenoic acid |
| Heneicosapentaenoic acid (HPA) | 21:5 (n-3) | all-cis-6,9,12,15,18-heneicosapentaenoic acid |
| Docosapentaenoic acid (DPA, Clupanodonic acid) | 22:5 (n-3) | all-cis-7,10,13,16,19-docosapentaenoic acid |
| Docosahexaenoic acid (DHA, Cervonic acid) | 22:6 (n-3) | all-cis-4,7,10,13,16,19-docosahexaenoic acid |
| Tetracosapentaenoic acid | 24:5 (n-3) | all-cis-9,12,15,18,21-tetracosapentaenoic acid |
| Tetracosahexaenoic acid (Nisinic acid) | 24:6 (n-3) | all-cis-6,9,12,15,18,21-tetracosahexaenoic acid |
Omega-6 Omega-6 fatty acids, polyunsaturated
| Common name | Lipid name | Chemical name |
| Linoleic acid | 18:2 (n-6) | all-cis-9,12-octadecadienoic acid |
| Gamma-linolenic acid (GLA) | 18:3 (n-6) | all-cis-6,9,12-octadecatrienoic acid |
| Eicosadienoic acid | 20:2 (n-6) | all-cis-11,14-eicosadienoic acid |
| Dihomo-gamma-linolenic acid (DGLA) | 20:3 (n-6) | all-cis-8,11,14-eicosatrienoic acid |
| Arachidonic acid (AA) | 20:4 (n-6) | all-cis-5,8,11,14-eicosatetraenoic acid |
| Docosadienoic acid | 22:2 (n-6) | all-cis-13,16-docosadienoic acid |
| Adrenic acid | 22:4 (n-6) | all-cis-7,10,13,16-docosatetraenoic acid |
| Docosapentaenoic acid (Osbond acid) | 22:5 (n-6) | all-cis-4,7,10,13,16-docosapentaenoic acid |
| Tetracosatetraenoic acid | 24:4 (n-6) | all-cis-9,12,15,18-tetracosatetraenoic acid |
| Tetracosapentaenoic acid | 24:5 (n-6) | all-cis-6,9,12,15,18-tetracosapentaenoic acid |
In particular we hear a lot about the Omega-6 fatty acid AA and the Omega-3 fatty acids EPA and DHA. Here is a simplified diagrammatic depiction of relationships among Omega-3 and Omega-6 fatty acids.

EFAs – Essential Fatty Acids – “An unsaturated fatty acid that is essential to human health, but cannot be manufactured in the body. Abbreviated EFA. There are three types of EFAs: arachnoidic acid, linoleic acid, and linolenic acid. When linoleic acid is obtained in the diet, it can be converted to both arachnoidic and linolenic acid. Linoleic acid is commonly found in cold-pressed oils, especially oils extracted from cold-water fish and certain seeds. Supplementation with EFAs appears to be useful as a treatment for certain neurological disorders.(ref).”
Eicosanoids – “any of a class of compounds (as the prostaglandins) derived from polyunsaturated fatty acids (as arachidonic acid) and involved in cellular activity(ref)”
Prostaglandins – “The prostaglandins(PG) are a group of physiologically active lipid compounds having diverse hormone-like effects in animals. Every prostaglandin contains 20 carbon atoms, including a 5-carbon ring. They are a subclass of eicosanoids and of the prostanoid class of fatty acid derivatives. The structural differences between prostaglandins account for their different biological activities. A given prostaglandin may have different and even opposite effects in different tissues in some cases. The ability of the same prostaglandin to stimulate a reaction in one tissue and inhibit the same reaction in another tissue is determined by the type of receptor to which the prostaglandin binds. They act as autocrine or paracrine factors with their target cells present in the immediate vicinity of the site of their secretion. Prostaglandins differ from endocrine hormones in that they are not produced at a specific site but in many places throughout the human body(ref)).” They are pro-inflammatory lipid mediators.
Lipoxins – “Lipoxins are endogenous anti-inflammatory, pro-resolving molecules that play a vital role in reducing excessive tissue injury and chronic inflammation. — Lipoxins regulate components of both the innate and adaptive immune systems including neutrophils, macrophages, T-, and B-cells. Lipoxins also modulate levels of various transcription factors such as nuclear factor κB, activator protein-1, nerve growth factor-regulated factor 1A binding protein 1, and peroxisome proliferator activated receptor γ and control the expression of many inflammatory genes(ref).” They are anti-inflammatory lipid mediators.
Lleukotrienes – “Leukotrienes are a family of eicosanoid inflammatory mediators produced in leukocytes by the oxidation of arachidonic acid (AA) and the essential fatty acid eicosapentaenoic acid (EPA) by the enzyme arachidonate 5-lipoxygenase(ref).” They are pro-inflammatory lipid mediators.
Maresins – Maresins are novel macrophage mediators with potent antiinflammatory and proresolving actions. They are biosynthesized by macrophages from the essential fatty acid docosahexaenoic acid (DHA). Again, they are anti-inflammatory lipid mediators.(ref)
Resolvins and Protectins – “Resolvins and protectins are recently identified molecules that are generated from ω-3 PUFA precursors and can orchestrate the timely resolution of inflammation in model systems (ref). They are anti-inflammatory lipid mediators.
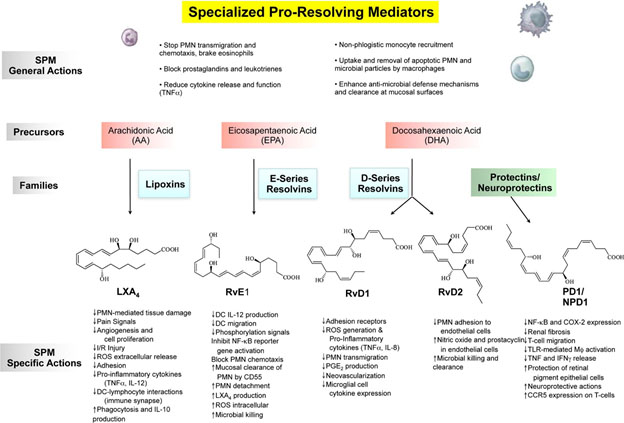
Image source: 2010 Novel Lipid Mediators and Resolution Mechanisms in Acute Inflammation
C. SOME RELEVANT RESEARCH FINDINGS AND DIRECTIONS
1..Lipid mediators that work to resolve inflammation can operate by affecting ion channels in neurons
The just-published November 2016 publication Modulation of the Activities of Neuronal Ion Channels by Fatty Acid-Derived Pro-Resolvents reports: “Progress of inflammation depends on the balance between two biological mechanisms: pro-inflammatory and pro-resolving processes. Many extracellular and intracellular molecular components including cytokines, growth factors, steroids, neurotransmitters, and lipidergic mediators and their receptors contribute to the two processes, generated from cellular participants during inflammation. Fatty acid-derived mediators are crucial in directing the inflammatory phase and orchestrating heterogeneous reactions of participants such as inflamed cells, innate immune cells, vascular components, innervating neurons, etc. As well as activating specific types of receptor molecules, lipidergic mediators can actively control the functions of various ion channels via direct binding and/or signal transduction, thereby altering cellular functions. Lipid mediators can be divided into two classes based on which of the two processes they promote: pro-inflammatory, which includes prostaglandins and leukotrienes, and pro-resolving, which includes lipoxins, resolvins, and maresins. The research on the modulations of neuronal ion channels regarding the actions of the pro-inflammatory class has begun relatively earlier while the focus is currently expanding to cover the ion channel interaction with pro-resolvents. As a result, knowledge of inhibitory mechanisms by the pro-resolvents, historically seldom found for other known endogenous modulators or pro-inflammatory mediators, is accumulating particularly upon sensory neuronal cation channels. Diverse mechanistic explanations at molecular levels are being proposed and refined. Here we overviewed the interactions of lipidergic pro-resolvents with neuronal ion channels and outcomes from the interactions, focusing on transient receptor potential (TRP) ion channels. We also discuss unanswered hypotheses and perspectives regarding their interactions.”
The practical implications of the Omega-3 derived lipid inflammation-resolving mediators, protectins, marasins and resolvins, is potentially very great. So, especially in the last 4 years, researchers have been examining implication for various inflammation-related disease processes. Some of the things being learned are exemplified by the following items 2. -5.
2. The inflammation mediators work by regulating miRNAs via pro-resolving G-protein coupled receptors
The 2012 publication Pro-Resolving Lipid Mediators (SPMs) and Their Actions in Regulating miRNA in Novel Resolution Circuits in Inflammation explains “Unresolved inflammation is associated with several widely occurring diseases such as arthritis, periodontal diseases, cancer, and atherosclerosis. Endogenous mechanisms that curtail excessive inflammation and prompt its timely resolution are of considerable interest. In recent years, previously unrecognized chemical mediators derived from polyunsaturated fatty acids were identified that control the acute inflammatory response by activating local resolution programs. Among these are the so-called specialized pro-resolving lipid mediators (SPMs) that include lipoxins (LX), resolvins (Rv), protectins (PD), and maresins (MaR), because they are enzymatically biosynthesized during resolution of self-limited inflammation. They each possess distinct chemical structures and regulate cellular pathways by their ability to activate pro-resolving G-protein coupled receptors (GPCRs) in a stereospecific manner. For instance, RvD1 controls several miRNAs of interest in self-limited acute inflammation that counter-regulate the mediators and proteins that are involved in inflammation. Here, we overview some of the biosynthesis and mechanisms of SPM actions with focus on the recently reported miR involved in their pro-resolving responses that underscore their beneficial actions in the regulation of acute inflammation and its timely resolution. The elucidation of these mechanisms operating in vivo to keep acute inflammation within physiologic boundaries as well as stimulate resolution have opened resolution pharmacology and many new opportunities to target inflammation-related human pathologies via activating resolution mechanisms.”
3. Markers of inflammation in activated microglial cells can be reversed by DHA
Inflammatory microglial activation is implicated in several neurological disesases and conditions such as Alzheimer’s disease and spinal cord injury. See the earlier blog entries Key roles of glia and microglia in age-related neurodegenerative diseases and New views of Alzheimer’s disease and new approaches to treating it. DHA (docosahexaenoic acid) is shown by the above-cited research to be precursors of maresins, protectins and resolvins, mediators that resolve inflammation, How this plays out in terms of activated microglia is described in the 2016 publication Remodeling of lipid bodies by docosahexaenoic acid in activated microglial cells. “Organelle remodeling processes are evolutionarily conserved and involved in cell functions during development, aging, and cell death. Some endogenous and exogenous molecules can modulate these processes. Docosahexaenoic acid (DHA), an omega-3 polyunsaturated fatty acid, has mainly been considered as a modulator of plasma membrane fluidity in brain development and aging, while DHA’s role in organelle remodeling in specific neural cell types at the ultrastructural level remains largely unexplored. DHA is notably incorporated into dynamic organelles named lipid bodies (LBs). We hypothesized that DHA could attenuate the inflammatory response in lipopolysaccharide (LPS)-activated microglia by remodeling LBs and altering their functional interplay with mitochondria and other associated organelles. RESULTS: We used electron microscopy to analyze at high spatial resolution organelle changes in N9 microglial cells exposed to the proinflammogen LPS, with or without DHA supplementation. Our results revealed that DHA reverses several effects of LPS in organelles. In particular, a large number of very small and grouped LBs was exclusively found in microglial cells exposed to DHA. In contrast, LBs in LPS-stimulated cells in the absence of DHA were sparse and large. LBs formed in the presence of DHA were generally electron-dense, suggesting DHA incorporation into these organelles. The accumulation of LBs in microglial cells from mouse and human was confirmed in situ. In addition, DHA induced numerous contacts between LBs and mitochondria and reversed the frequent disruption of mitochondrial integrity observed upon LPS stimulation. Dilation of the endoplasmic reticulum lumen was also infrequent following DHA treatment, suggesting that DHA reduces oxidative stress and protein misfolding. Lipidomic analysis in N9 microglial cells treated with DHA revealed an increase in phosphatidylserine, indicating the role of this phospholipid in normalization and maintenance of physiological membrane functions. This finding was supported by a marked reduction of microglial filopodia and endosome number and significant reduction of LPS-induced phagocytosis.”sponse in LPS-stimulated microglial cells by remodeling LBs and altering their interplay with mitochondria and other associated organelles. Our findings point towards a mechanism by which omega-3 DHA participates in organelle reorganization and contributes to the maintenance of neural cell homeostasis.”
4. Aspirin also triggers a DHA resolvin
For example, see the 2007 article Resolvin D1 and Its Aspirin-triggered 17REpimer. “We recently uncovered two new families of potent docosahexaenoic acid-derived mediators, termed D series resolvins (Rv; resolution phase interaction products) and protectins. Here, we assign the stereochemistry of the conjugated double bonds and chirality of alcohols present in resolvin D1 (RvD1) and its aspirin-triggered 17R epimer (AT-RvD1) with compounds prepared by total organic synthesis. In addition, docosahexaenoic acid was converted by a single lipoxygenase in a “one-pot” reaction to RvD1 in vitro. The synthetic compounds matched the physical and biological properties of those enzymatically generated. RvD1 proved to be 7S,8R,17S-trihydroxy-4Z,9E,11E,13Z,15E,19Z-docosahexaenoic acid, AT-RvD1 matched 7S,8R,17R-trihydroxy-4Z,9E,11E,13Z,15E,19Z-docosahexaenoic acid, and they both stopped transendothelial migration of human neutrophils (EC50 ∼30 nM). In murine peritonitis in vivo, RvD1 and AT-RvD1 proved equipotent (at nanogram dosages), limiting polymorphonuclear leukocyte infiltration in a dose-dependent fashion.”
D. TRANSATIONAL APPLICATIONS TO SPECIFIC INFLAMMATORY DISEASE CONDITIONS
There is a significant amount of recent and ongoing research aimed at translating knowledge about protectins, resolvins and maresins into practical clinical interventions for controlling specific inflammatory disease processes. I cite a few examples of hundreds or thousands of publications illustrating this point.
1. The inflammation-resolving mediators in DHA and EPA might prove to be very useful for preventing cognitive impairment and treating dementias.
The 2015 publication Specialized Pro-Resolving Mediators from Omega-3 Fatty Acids Improve Amyloid-β Phagocytosis and Regulate Inflammation in Patients with Minor Cognitive Impairment relates: “In this review we discuss the immunopathology of Alzheimer’s disease (AD) and recent advances in the prevention of minor cognitive impairment (MCI) by nutritional supplementation with omega-3 fatty acids. Defective phagocytosis of amyloid-β (Aβ) and abnormal inflammatory activation of peripheral blood mononuclear cells (PBMCs) are the two key immune pathologies of MCI and AD patients. The phagocytosis of Aβ by PBMCs of MCI and AD patients is universally defective and the inflammatory gene transcription is heterogeneously deregulated in comparison to normal subjects. Recent studies have discovered a cornucopia of beneficial anti-inflammatory and pro-resolving effects of the specialized proresolving mediators (SPMs) resolvins, protectins, maresins, and their metabolic precursors. Resolvin D1 and other mediators switch macrophages from an inflammatory to a tissue protective/pro-resolving phenotype and increase phagocytosis of Aβ. In a recent study of AD and MCI patients, nutritional supplementation by omega-3 fatty acids individually increased resolvin D1, improved Aβ phagocytosis, and regulated inflammatory genes toward a physiological state, but only in MCI patients. Our studies are beginning to dissect positive factors (adherence to Mediterranean diet with omega-3 and exercise) and negative factors (high fat diet, infections, cancer, and surgeries) in each patient. The in vitro and in vivo effects of omega-3 fatty acids and SPMs suggest that defective phagocytosis and chronic inflammation are related to defective production and/or defective signaling by SPMs in immune cells.”
2. Going back to the early 2000s, there has been interest in the role of DHA for the control of Alzheimer’s disease.
Even though back then protectins,resolvins and maresins were not yet heard of.
A 2006 publication was Docosahexaenoic acid protects from amyloid and dendritic pathology in an Alzheimer’s disease mouse model.
A 2016 publication Role of docosahexaenoic acid in the modulation of glial cells in Alzheimer’s disease reports: “Docosahexaenoic acid (DHA) is an omega-3 (ω-3) long-chain polyunsaturated fatty acid (LCPUFA) relevant for brain function. It has largely been explored as a potential candidate to treat Alzheimer’s disease (AD). Clinical evidence favors a role for DHA in the improvement of cognition in very early stages of the AD. In response to stress or damage, DHA generates oxygenated derivatives called docosanoids that can activate the peroxisome proliferator-activated receptor γ (PPARγ). In conjunction with activated retinoid X receptors (RXR), PPARγ modulates inflammation, cell survival, and lipid metabolism. As an early event in AD, inflammation is associated with an excess of amyloid β peptide (Aβ) that contributes to neural insult. Glial cells are recognized to be actively involved during AD, and their dysfunction is associated with the early appearance of this pathology. These cells give support to neurons, remove amyloid β peptides from the brain, and modulate inflammation. Since DHA can modulate glial cell activity, the present work reviews the evidence about this modulation as well as the effect of docosanoids on neuroinflammation and in some AD models. The evidence supports PPARγ as a preferred target for gene modulation. The effective use of DHA and/or its derivatives in a subgroup of people at risk of developing AD is discussed.”
3. The inflammation resolution-phase mediators in Omega-3 oils are also being seriously looked at as candidates for ending inflammation in rheumatoid arthritis
The 2016 publication Implications for eicosapentaenoic acid- and docosahexaenoic acid-derived resolvins as therapeutics for arthritis relates: “Omega-3 polyunsaturated fatty acids are essential for health and are known to possess anti-inflammatory properties, improving cardiovascular health as well as benefiting inflammatory diseases. Indeed, dietary supplementation with omega-3 polyunsaturated fatty acids has proved efficacious in reducing joint pain, morning stiffness and nonsteroidal anti-inflammatory drugs usage in rheumatoid arthritis patients. However, the mechanisms by which omega-3 polyunsaturated fatty acids exert their beneficial effects have not been fully explored. Seminal discoveries by Serhan and colleagues have unveiled a novel class of bioactive lipid mediators that are enzymatically biosynthesized in vivo from omega-3 eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), termed resolvins, protectins and maresins. These bioactive pro-resolving lipid mediators provide further rationale for the beneficial effects of fish-oil enriched diets. These endogenous lipid mediators are spatiotemporally biosynthesized to actively regulate resolution by acting on specific G protein-coupled receptors (GPCRs) to initiate anti-inflammatory and pro-resolving signals that terminate inflammation. In this review, we will discuss the mechanism of actions of these molecules, including their analgesic and bone-sparing properties making them ideal therapeutic agonists for the treatment of inflammatory diseases such as rheumatoid arthritis.”
4. Inflammatory Bowel Disease (IBD)
The same or similar story is being described in the research literature last year or this year for essentially all of the inflammation-related diseases. The 2016 publication Inflammatory bowel disease: can omega-3 fatty acids really help? relates: “Adjuvants to the traditional therapy of inflammatory bowel disease (IBD) have been studied to enhance the efficacy of the treatment and improve patients’ quality of life. Omega-3 polyunsaturated fatty acids (ω3FA) have been associated with attenuation of the inflammatory responses in IBD, possibly acting as substrates for anti-inflammatory eicosanoid production, similar to prostaglandins and leukotrienes. ω3FA also act as substrates for the synthesis of resolvins, maresins and protectins, indispensable in resolving inflammation processes. These acids may influence the development or course of IBD by: reducing oxidative stress, production of tumor necrosis factor-α and proinflammatory cytokines; working as chemopreventive agents; and decreasing the expression of adhesion molecules. There are numerous controversies in the literature on the effects of ω3FA in the prevention or treatment of IBD, but their effects in reducing inflammation is incontestable. Therefore, more studies are warranted to elucidate the pathophysiological mechanisms and establish the recommended daily intake to prevent or induce remission in IBD patients.”
This 2016 publication relates to inflammatory bowel disease, EPA- and DHA-derived resolvins’ actions in inflammatory bowel disease. “Inflammatory bowel diseases are chronic diseases divided into two major forms, ulcerative colitis and Crohn’s disease, which are both associated with a chronic inflammatory condition of the gastrointestinal tract. Recent studies have shown that the resolution of inflammatory conditions is a biosynthetically active process where new pro-resolution lipid mediators derived from omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), such as E- and D-series resolvins, protectins, and macrophage mediator in resolving inflammation (maresins), have potent anti-inflammatory activity and serve as specialised mediators that play an important role in the resolution of inflammation. Recent studies have also shown the role of resolvins in referred hyperalgesia associated with different inflammatory processes, such as the visceral pain caused by inflammatory bowel disease. There are many reports describing the principal effects of EPA- and DHA-derived mediators in experimental models of inflammatory bowel diseases. This review focuses on the recent studies on the important role played by pro-resolution lipid mediators in controlling the inflammatory process associated with inflammatory bowel diseases.”
5. How about lung inflammation due to cigarette smoking? Yes, that might be helped too, at least by one resolvin.
The 2013 publication A novel anti-inflammatory and pro-resolving role for resolvin D1 in acute cigarette smoke-induced lung inflammation reported “INTRODUCTION: Cigarette smoke is a profound pro-inflammatory stimulus that contributes to acute lung injuries and to chronic lung disease including COPD (emphysema and chronic bronchitis). Until recently, it was assumed that resolution of inflammation was a passive process that occurred once the inflammatory stimulus was removed. It is now recognized that resolution of inflammation is a bioactive process, mediated by specialized lipid mediators, and that normal homeostasis is maintained by a balance between pro-inflammatory and pro-resolving pathways. These novel small lipid mediators, including the resolvins, protectins and maresins, are bioactive products mainly derived from dietary omega-3 and omega-6 polyunsaturated fatty acids (PUFA). We hypothesize that resolvin D1 (RvD1) has potent anti-inflammatory and pro-resolving effects in a model of cigarette smoke-induced lung inflammation. METHODS: Primary human lung fibroblasts, small airway epithelial cells and blood monocytes were treated with IL-1β or cigarette smoke extract in combination with RvD1 in vitro, production of pro-inflammatory mediators was measured. Mice were exposed to dilute mainstream cigarette smoke and treated with RvD1 either concurrently with smoke or after smoking cessation. The effects on lung inflammation and lung macrophage populations were assessed. RESULTS: RvD1 suppressed production of pro-inflammatory mediators by primary human cells in a dose-dependent manner. Treatment of mice with RvD1 concurrently with cigarette smoke exposure significantly reduced neutrophilic lung inflammation and production of pro-inflammatory cytokines, while upregulating the anti-inflammatory cytokine IL-10. RvD1 promoted differentiation of alternatively activated (M2) macrophages and neutrophil efferocytosis. RvD1 also accelerated the resolution of lung inflammation when given after the final smoke exposure. CONCLUSIONS: RvD1 has potent anti-inflammatory and pro-resolving effects in cells and mice exposed to cigarette smoke. Resolvins have strong potential as a novel therapeutic approach to resolve lung injury caused by smoke and pulmonary toxicants.”
Further research is reported in the 2016 paper Resolvin D1 prevents smoking-induced emphysema and promotes lung tissue regeneration. “RvD1 attenuated smoking-induced emphysema in vivo by reducing inflammation and promoting tissue regeneration. This result suggests that RvD1 may be useful in the prevention and treatment of emphysema.
6. Asthma and Allergic Diseases
The 2015 publication Role of omega-3 fatty acids and their metabolites in asthma and allergic diseases relates: “Large numbers of epidemiological and observational studies investigating the effect of fish intake or omega-3 fatty acid supplementation during pregnancy, lactation, infancy, childhood, and adulthood on asthmatic and allergic outcomes have been conducted. They mostly indicate protective effects and suggest a causal relationship between decreased intake of fish oil in modernized diets and an increasing number of individuals with asthma or other allergic diseases. Specialized pro-resolving mediators (SPM: protectins, resolvins, and maresins) are generated from omega-3 fatty acids such as EPA and DHA via several enzymatic reactions. These mediators counter-regulate airway eosinophilic inflammation and promote the resolution of inflammation in vivo. Several reports have indicated that the biosynthesis of SPM is impaired, especially in severe asthma, which suggests that chronic inflammation in the lung might result from a resolution defect. This article focuses on the beneficial aspects of omega-3 fatty acids and offers recent insights into their bioactive metabolites including resolvins and protectins.”
7. Cancers
Several recent papers supporting more or less the same story – 1. How dietary Omega6 eicosanoids can contribute to cancer progression and 2. how the resolution-phase immune modulators are likely to be useful in prevention of and therapies for cancers. Including:
2014 Involvement of eicosanoids in the pathogenesis of pancreatic cancer: the roles of cyclooxygenase-2 and 5-lipoxygenase. “The interplay between inflammation and cancer progression is a growing area of research. A combination of clinical, epidemiological, and basic science investigations indicate that there is a relationship between inflammatory changes in the pancreas and neoplastic progression. Diets high in ω-6 polyunsaturated fatty acids provide increased substrate for arachidonic acid metabolism by cyclooxygenase-2 (COX-2) and 5-lipoxygenase (5-LOX) to form eicosanoids. These eicosanoids directly contribute to pancreatic cancer cell proliferation. Both COX-2 and 5-LOX are upregulated in multiple cancer types, including pancreatic cancer. In vitro studies using pancreatic cancer cell lines have demonstrated upregulation of COX-2 and 5-LOX at both the mRNA and protein levels. When COX-2 and 5-LOX are blocked via a variety of mechanisms, cancer cell proliferation is abrogated both in vitro and in vivo.”
2015 Eicosanoid pathway in colorectal cancer: Recent updates.
2011 Regulation of inflammation in cancer by eicosanoids.
8. Cardiovascular disease risk reduction
Again, there are many recent publications on this, including:
2013 Do omega-3 polyunsaturated Fatty acids prevent cardiovascular disease? A review of the randomized clinical trials. “Fish oil is rich in the omega-3 polyunsaturated fatty acids (PUFAs) eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). Numerous epidemiological studies and several large randomized clinical trials have shown that modest doses of omega-3 PUFAs significantly reduce the risk of unstable angina, myocardial infarction, and sudden cardiac death as well as death in coronary artery disease and heart failure patients. Based on the scientific evidence, the American Heart Association (AHA) has recommended all individuals eat fish at least twice a week to prevent cardiovascular disease. For individuals with coronary artery disease, the recommended dose of omega-3 PUFAs is 1 g of EPA and DHA daily. To lower triglyceride levels, much higher doses are needed. However, more recent randomized clinical trials have questioned the cardiovascular benefits of fish oil. These studies have contributed to the uncertainty health care providers face when recommending omega-3 PUFA supplementation according to clinical guidelines. The purpose of this review is to examine the randomized clinical trials and scientific evidence between omega-3 PUFAs and cardiovascular outcomes to better understand the current role of omega-3 PUFAs in improving cardiovascular health.”
2008 Fatty acid facts, Part III: Cardiovascular disease, or, a fish diet is not fishy.
2012 (n-3) fatty acids and cardiovascular health: are effects of EPA and DHA shared or complementary?
2011 Role of ω3 long-chain polyunsaturated fatty acids in reducing cardio-metabolic risk factors.
9. Metabolic syndrome and obesity liver impacts
The 2015 publication Pro-resolving mediators produced from EPA and DHA: Overview of the pathways involved and their mechanisms in metabolic syndrome and related liver diseases reports: “A novel genus of pro-resolving lipid mediators endogenously generated from omega-3 polyunsaturated fatty acids has been identified in exudates obtained during the resolution phase of acute inflammation. The term specialized pro-resolving mediators (SPM) has been coined for these lipid mediators, comprising four novel chemical mediator families designated resolvins of the E series (if derived from eicosapentaenoic acid) and resolvins of the D series, protectins and maresins (if generated from docosahexaenoic acid). These SPM act not only as “stop-signals” of inflammatory response, but also as facilitators of the ability of macrophages to clear apoptotic cells (efferocytosis) and migrate to peripheral lymph nodes (efflux), thus, expediting their removal from sites of inflammation. In this review, we provide an overview of the current efforts to elucidate the structure-function, biosynthesis and actions of these omega-3-derived SPM in the context of inflammatory diseases. We specifically highlight the role of these SPM as endogenous counter-regulators of the persistent inflammatory status present in adipose tissue of obese individuals and describe the potential therapeutic impact of these bioactive lipid autacoids on the prevention of hepatic co-morbidities associated with obesity and the metabolic syndrome.”
E. COMING IN THIS BLOG
We expect this blog series on inflammation will go on with more articles. As we see it now: Part 4 will be concerned with the frequent decoupling of transcription and translation in protein synthesis, the code for what goes on at the Internal Ribosome Entry Sites” (or IRES). And it will be concerned with implications of decoupling for both generation of inflammation and its consequences, particularly insulin resistance. Jim Watson has already drafted this. Part 5 will be concerned with inflammasomes, those strange self-assembling structures which lead to persistent inflammation. And Part 6 will be concerned about several traditional herbal remedies for inflammation and how their effectiveness and bioavability can be multiplied by judicious combination and by current nano-delivery techniques as ancient Chinese and Ayurvedic medicine joins up with high-tech in the emerging present.


Pingback: ON AGING - AGINGSCIENCES™ - Anti-Aging Firewalls™AGINGSCIENCES™ – Anti-Aging Firewalls™
Pingback: INFLAMMATION PART 6: THE SCIENCE BEHIND THE 4 HERB SYNERGY DIETARY SUPPLEMENT - AGINGSCIENCES™ - Anti-Aging Firewalls™AGINGSCIENCES™ – Anti-Aging Firewalls™
Pingback: FUNNY THINGS ARE HAPPENING TO ME ON THE WAY TO 100 - AGINGSCIENCES™ - Anti-Aging Firewalls™AGINGSCIENCES™ – Anti-Aging Firewalls™
Pingback: Inflammation Part 4 - PCSK9 inhibition - Also Part 1 of interventions that reduce all cause mortality (ACM) - AGINGSCIENCES™ - Anti-Aging Firewalls™AGINGSCIENCES™ – Anti-Aging Firewalls™
Pingback: Inflammation Part 7: Neurohormesis, neuroinflammatory diseases, and their treatment by mushroom substances (Section 1) - AGINGSCIENCES™ - Anti-Aging Firewalls™AGINGSCIENCES™ – Anti-Aging Firewalls™
Pingback: MECHANISMS OF AGING AND INTERVENTIONS FOR LONGEVITY PART 1 THE FIRST 100 YEARS - AGINGSCIENCES™ - Anti-Aging Firewalls™AGINGSCIENCES™ – Anti-Aging Firewalls™