By Vince Giuliano
This blog is about Mitohormesis, a different form of hormesis than that discussed in the previous blog entry Radiation Hormesis. Mitohormesis has to do with cell metabolic pathways and oxidative stress, topics I have discussed in many previous blog entries. The proper functioning of mitohormesis has to do with multiple aspects of health and with extended organism longevity in many species, quite probably including our own. And the absence of proper mitohormesis functioning has been shown to be associated with certain disease processes and shortened lifespans in a range of animals including humans. As is the case for radiation hormesis, the concept of mitohormesis has been around for some time but has not been universally accepted. It is only recently being widely acknowledged as an important biological phenomenon. And only relatively recently are its mechanisms of operation being unveiled.
I am grateful for an intensive round of communication with James P Watson, a follower of this blog and stem cell researcher, which helped me to improve this blog entry. Also I feel free to selectively quote some of Watson’s comments sent me by e-mail while this blog entry was being researched. This is not to say that Watson, clearly a very brilliant person, will necessarily agree with all I have to say.
In researching this blog entry I encountered a number of interesting recent research results having to do with topics I have discussed previously including mitochondrial metabolism, the OXPHOS system in general, mitochondrial biogenesis, frataxin, the key roles of SIRT3 in controlling ROS stress in mitochondria, and the health roles of PCG1alpha. And I started to question whether it makes sense to supplement with PQQ. And there are pathways which seem to produce health benefits independently of motohormesis, like inhibition of TOR. Because of the great complexity involved I decided to cover most of those topics in subsequent blog entries – even though they all bear on mitohormesis. So, my intent is to keep this present blog entry focused on mitohormesis itself, what turns it on and off, and the direct health implications of mitohormesis. In particular, I deal with the important practical topic of mitohormesis and exercise, and how some antioxidants nullify the health benefits of regular exercise.
Hormesis in General
The first section of the previous blog entry Radiation Hormesis is a short introduction to the topic of hormesis in general, and I will not repeat that introduction here.
What is Mitohormesis?
A definition and summary introduction to mitohormesis is found in the abstract of the 2010 publication How increased oxidative stress promotes longevity and metabolic health: The concept of mitochondrial hormesis (mitohormesis): “Recent evidence suggests that calorie restriction and specifically reduced glucose metabolism induces mitochondrial metabolism to extend life span in various model organisms, including Saccharomyces cerevisiae, Drosophila melanogaster, Caenorhabditis elegans and possibly mice. In conflict with Harman’s free radical theory of aging (FRTA), these effects may be due to increased formation of reactive oxygen species (ROS) within the mitochondria causing an adaptive response that culminates in subsequently increased stress resistance assumed to ultimately cause a long-term reduction of oxidative stress. This type of retrograde response has been named mitochondrial hormesis or mitohormesis, and may in addition be applicable to the health-promoting effects of physical exercise in humans and, hypothetically, impaired insulin/IGF-1-signaling in model organisms. Consistently, abrogation of this mitochondrial ROS signal by antioxidants impairs the lifespan-extending and health-promoting capabilities of glucose restriction and physical exercise, respectively. In summary, the findings discussed in this review indicate that ROS are essential signaling molecules which are required to promote health and longevity. Hence, the concept of mitohormesis provides a common mechanistic denominator for the physiological effects of physical exercise, reduced calorie uptake, glucose restriction, and possibly beyond.”
The July 2011 review publication Extending life span by increasing oxidative stress also argues for the existence of mitohormesis across very diverse species: “Various nutritional, behavioral, and pharmacological interventions have been previously shown to extend life span in diverse model organisms, including Saccharomyces cerevisiae, Caenorhabditis elegans, Drosophila melanogaster, mice, and rats, as well as possibly monkeys and humans. This review aims to summarize published evidence that several longevity-promoting interventions may converge by causing an activation of mitochondrial oxygen consumption to promote increased formation of reactive oxygen species (ROS). These serve as molecular signals to exert downstream effects to ultimately induce endogenous defense mechanisms culminating in increased stress resistance and longevity, an adaptive response more specifically named mitochondrial hormesis or mitohormesis. Consistently, we here summarize findings that antioxidant supplements that prevent these ROS signals interfere with the health-promoting and life-span-extending capabilities of calorie restriction and physical exercise. Taken together and consistent with ample published evidence, the findings summarized here question Harman’s Free Radical Theory of Aging and rather suggest that ROS act as essential signaling molecules to promote metabolic health and longevity.”
My regular readers will discover in these statements many themes discussed in previous blog entries: Reactive Oxygen Species (ROS) are important signaling molecules(ref), increased ROS expression up to a certain point may be good instead of bad for you(ref)(ref), the stress induced by ROS leads (via the Nrf2 pathway) to activation of multiple endogenous antioxidant and other protective genes(ref)(ref). And, in fact, suppression of ROS by taking antioxidants can be health-damaging and life-shortening(ref). These points were also made in the publications related to radiation hormesis cited in the previous blog entry. They are also articulated in several of the publications that will be cited here as relevant to mitohormesis.
Hormesis, Nrf2 and Antioxidant supplementation – a unifying framework
Before proceeding further with specific findings reported in the literature, I would like to outline a framework for viewing these findings, findings which otherwise sometimes seem inconsistent.
The major points of this framework are:
- I hypothesize that the operation of the the Keap1-Nrf2 pathway is a fundamental mechanism of hormesis. I do that because the operation of the Keap1-Nrf2 pathway is by itself sufficient to explain most if not all the observed phenomena of mitohormesis and hormesis in general.
In reviewing hundreds of research articles relating the Nrf2 pathway, I found hormesis mentioned only rarely. And, in reviewing the literature of hormesis I likewise find that the keap1-Nrf2 pathway is mentioned only rarely. Yet my research suggests that everything that is known to happen associated with hormesis is completely explainable by the operation of keap1-Nrf2 pathway, without reference to other pathways normally associated with hormesis such as the heat-shock response. See the subsection entitled The single most-important biological pathway responsible for radiation hormesis is probably the activation of Nrf2 by radiation-created oxidative stress in the blog entry Radiation Hormesis. “I have written extensively about Nrf2 in this blog. Nrf2 is the master regulator of the body’s response to stresses of most kinds. Activating hundreds of the body’s natural antioxidant and stress defense genes, the Nrf2/Keap1 pathway provides a central mechanism for most if not all forms of hormesis. Specifically, you can see the blog entries The pivotal role of Nrf2. Part 1 – a new view on the control of oxidative damage and generation of hormetic effects, The pivotal role of Nrf2. Part 2 – foods, phyto-substances and other substances that turn on Nrf2 and The pivotal role of Nrf2. Part 3– Is promotion of Nrf2 expression a viable strategy for human human healthspan and lifespan extension?.”
In stressing the role of the keap1-Nrf2 pathway in hormesis, I of course acknowledge that this is a simplification and far from tells the whole story. Many other pathways downstream from keap1-Nrf2 are involved in a hormetic response , namely the ones activated by the AREs (antioxidant response genes) after Nrf2 translocates to the cell’s nucleus. SIRT3and the heat shock proteins like HPS70 play important roles in hormesis(ref)(ref), along with and possibly due to expression triggered by Nrf2 expression(ref)(ref)(ref). L-proline catabolism can also be involved(ref). HDAC inhibition and histone-dependent gene silencing can be involved. And the whole subject is wrapped up in the vastly complex subjects of ROS dynamics and cell metabolism involving AMPK, PGC1-alpha, PPAR-gamma, SOD2, OXPHOS, P38, etc.. And of course there are other largely independent body stress-response pathways like the UPR (unfolded protein response)(ref) and the various kinds of DNA repair machinery. But seeing the keap1-Nrf2 pathway as primary can be a very useful one, because research on Nrf2 has been yielding many insights relevant to understanding the scope and relevancy of hormesis So, the hypothesis is an application of Occam’s Razor, where simplicity is paramount.
2. A key to understanding what is going on in hormesis is understanding how the hormetic dose-response curve works, and this can be formulated in terms of NRF2 expression.
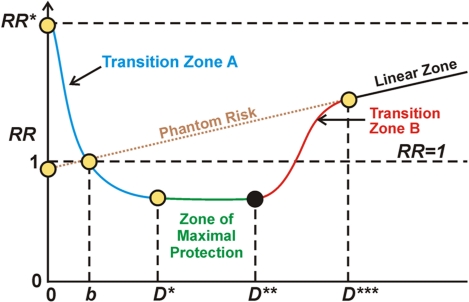
Graphic depiction of the stochastic hormetic relative risk model as given in Scott et al. (2009).
Understanding the typical dose-response curve associated with hormesis is critical for interpreting seemingly contradictory research. In my interpretation, the horizontal axis depicts level of stress, say as driven by ROS load. The vertical axis represents relative risk, level of probable pathological organismic response where normal level is 1. To the left of the first axis crossing in the diagram (point D) the Keap1-Nrf2 pathway is progressively kicking in but not sufficiently so as to overcome the direct negative effects of ROS stress. So, in Transition zone A there is under-expression of the ARE genes and a negative health condition Between stress levels b and D*** there is hormetic protection compared to what would be expected given a linear model of negative response to stress and better-than-expected health, due at least in part to activation of Nrf2 and the ARE genes. The zone of maximum protection is between D* and D**. Starting at D** to D*** the stress load begins to overwhelm the defensive activities of the ARE genes and the protection becomes less and less until at point D*** the hormetic response associated with ARE gene activation becomes negligible. In the case of radiation damage at least, beyond point D*** the damage according to conventional wisdom is in linear proportion to the stressor, the amount of radiation. Phantom risk is theoretical risk for low stress levels that would apply if the linear model were extrapolated for low stress dosages.
According to this model, supplementation with radical-scavenging supplements like vitamins C and E inhibits the ROS which triggers the release of Nrf2 which produces hormesis when the stress is in the hormesis range, i.e between b and D*** on the diagram. The result of such supplementation is a lower health state. That is, supplementation with such antioxidants when the stress is within the hormesis range of stress can turn off the stress that activates the keap1-Nrf2 pathway therefore turning off hormesis and can do more harm than good. It appears, for example, that this is what happens with exercise where taking antioxidants eliminates the hormetic health benefits of the exercise.
On the other hand, taking radical-scavenging supplements can be very useful and even highly desirable when the stress at a level is to the right of point D*** The traditional Harman free radical theory aging and the invoking of traditional antioxidant protection is valid in the linear zone beyond point D*** or even possibly beyond point D**. Some of the earlier research on free radicals and their damaging effects appears to be concerned with high dosages in this beyond-hormesis region. I am thinking of publications like ß-Cell Glucose Toxicity, Lipotoxicity, and Chronic Oxidative Stress in Type 2 Diabetes, Pancreatic islet β-cell and oxidative stress : The importance of glutathione peroxidase (2007), and Feeding acetyl-L-carnitine and lipoic acid to old rats significantly improves metabolic function with decreasing oxidative stress (2002). So these early anti-ROS and pro-antioxidant papers are valid in their own domains and not in fact contradicted by later papers that show positive health effects associated with lower levels of ROS stress. I believe serious nuclear plant radiation overexposure and accidental ingestion of heavy metals are excellent reasons for taking heavy doses of radical-scavenging antioxidants.
Many phytochemical substances present in certain foods and sold as supplements are called “antioxidants,” but actually are not free radical scavengers. They work by activating Nrf2 and triggering a hormesis response which lowers ROS and produces a wide range of positive health effects. I discuss these and other substances which activate Nrf2 in the blog entry The pivotal role of Nrf2. Part 2 – foods, phyto-substances and other substances that turn on Nrf2. I believe most of the phytosubstances mentioned there are useful to trigger hormesis or reinforce it within the hormesis stress range (up to point D*** in the diagram), and in no-way inhibit hormesis. Of course, they can’t overcome massive doses of ROS which correspond to the region to the right of D***on the hormesis response curve.
Now, moving on to some results reported in the literature:
In some lower organisms, inducing enhanced ROS signaling can lead to mitohormesis and significantly extended lifespans.
The April 2012 publication Impaired insulin/IGF1 signaling extends life span by promoting mitochondrial L-proline catabolism to induce a transient ROS signal reports: “Impaired insulin and IGF-1 signaling (iIIS) in C. elegans daf-2 mutants extends life span more than 2-fold. Constitutively, iIIS increases mitochondrial activity and reduces reactive oxygen species (ROS) levels. By contrast, acute impairment of daf-2 in adult C. elegans reduces glucose uptake and transiently increases ROS. Consistent with the concept of mitohormesis, this ROS signal causes an adaptive response by inducing ROS defense enzymes (SOD, catalase), culminating in ultimately reduced ROS levels despite increased mitochondrial activity. Inhibition of this ROS signal by antioxidants reduces iIIS-mediated longevity by up to 60%. Induction of the ROS signal requires AAK-2 (AMPK), while PMK-1 (p38) and SKN-1 (NRF-2) are needed for the retrograde response. IIIS upregulates mitochondrial L-proline catabolism, and impairment of the latter impairs the life span-extending capacity of iIIS while L-proline supplementation extends C. elegans life span. Taken together, iIIS promotes L-proline metabolism to generate a ROS signal for the adaptive induction of endogenous stress defense to extend life span.”
Many substances can promote mitohormesis at low doses, even poisons which are highly toxic at higher doses.
An example is the highly toxic pesticide rotenone. The publicationEffects of Low Concentrations of Rotenone Upon Mitohormesis in SH-SY5Y Cells reports: “The mitochondrial toxin rotenone exerts cytotoxicity viaoverproduction of reactive oxygen species (ROS) and depolarization of the mitochondrial membrane. We investigated the effects of rotenone (12.5, 25, 50, 100 nmol/L) on mitochondrial biogenesis and the potential roles of ROS production in SH-SY5Y cells. Mitochondrial biogenesis was assessed by counting the number of mitochondria, determining protein expression of peroxisome proliferator-activated receptor γ coactivator α (PGC1-α) and its regulator, SIRT1, and oxygen consumption. ROS production and levels of reduced glutathione (GSH) and oxidized glutathione(GSSG) were also determined. Compared with controls, rotenone (12.5 nmol/L) significantly increased the quantity of mitochondria and amount of oxygen consumption, whereas rotenone at >12.5 nmol/L decreased the quantity of mitochondria and amount of oxygen consumption. GSH contents and GSH/GSSG were also significantly enhanced by rotenone at 12.5 nmol/L and decreased by rotenone at >12.5 nmol/L. Except for ROS production and SIRT1 protein expression, all concentration-response relationships showed a typical inverted-U shape. ROS production was continually increased in cells treated with rotenone. These data indicate that low concentrations of rotenone can induce mitohormesis, which may be attributed to ROS production.”
James P Watson comments about this finding: “This study will not be replicated in humans! It does show that even mitochondrial poison pesticides can have a beneficial mitohormetic effect at low doses. The “mitohormetic ROS dose” is 12.5-25 nmol/L whereas the mitochondrial toxic dose is > 25 nmol/L. PGC-1alpha and SIRT1 increased with “mitohormetic doses” of rotenone, whereas they both decreased with higher doses. Again, more proof of mitohormetic dose-response curve.”
One of several substances that promotes mitohormesis in nematodes is the phytochemical glaucarubinone. The 2011 publication The phytochemical glaucarubinone promotes mitochondrial metabolism, reduces body fat, and extends lifespan of Caenorhabditis elegans reports: “Naturally occurring compounds that promote energy expenditure and delay aging in model organisms may be of significant interest, since these substances potentially provide pharmaceutical approaches to tackle obesity and promote healthy lifespan in humans. We aimed to test whether pharmaceutical concentrations of glaucarubinone, a cytotoxic and antimalarial quassinoid known from different species of the plant family Simaroubaceae, are capable of affecting metabolism and/or extending lifespan in a nematodal model organism for aging processes, the roundworm Caenorhabditis elegans. — Adult C. elegans roundworms, maintained on agar plates, were fed with E. coli strain OP50 bacteria, and glaucarubinone was applied to the agar to test (i) whether it alters respiration rates and mitochondrial activity, (ii) whether it affects body fat content, and (iii) whether it may promote longevity by quantifying survival in the presence and absence of the compound. We have found that glaucarubinone induces oxygen consumption and reduces body fat content of C. elegans. Moreover and consistent with the concept of mitohormesis, glaucarubinone extends C. elegans lifespan when applied at a concentration of 1 or 10 nanomolar. Taken together, glaucarubinone is capable of reducing body fat and promoting longevity in C. elegans, tentatively suggesting that this compound may promote metabolic health and lifespan in mammals and possibly humans.”
Yet another substance that appears to induce a hormetic-like effect in muscle tissues is Stanozolol. The 2011 publication Stanozolol treatment decreases the mitochondrial ROS generation and oxidative stress induced by acute exercise in rat skeletal musclereports: “Anabolic androgenic steroids are used in the sport context to enhance muscle mass and strength and to increase muscle fatigue resistance. Since muscle fatigue has been related to oxidative stress caused by an exercise-linked reactive oxygen species (ROS) production, we investigated the potential effects of a treatment with the anabolic androgenic steroid stanozolol against oxidative damage induced on rat skeletal muscle mitochondria by an acute bout of exhaustive exercise. Mitochondrial ROS generation with complex I- and complex II-linked substrates was increased in exercised control rats, whereas it remained unchanged in the steroid-treated animals. Stanozolol treatment markedly reduced the extent of exercise-induced oxidative damage to mitochondrial proteins, as indicated by the lower levels of the specific markers of protein oxidation, glycoxidation, and lipoxidation, and the preservation of the activity of the superoxide-sensitive enzyme aconitase. This effect was not due to an enhancement of antioxidant enzyme activities. Acute exercise provoked changes in mitochondrial membrane fatty acid composition characterized by an increased content in docosahexaenoic acid. In contrast, the postexercise mitochondrial fatty acid composition was not altered in stanozolol-treated rats. Our results suggest that stanozolol protects against acute exercise-induced oxidative stress by reducing mitochondrial ROS production, in association with a preservation of mitochondrial membrane properties.”
The September 2011 publication Lonidamine extends lifespan of adult Caenorhabditis elegans by increasing the formation of mitochondrial reactive oxygen species reports: “Compounds that delay aging in model organisms may be of significant interest to antiaging medicine, since these substances potentially provide pharmaceutical approaches to promote healthy lifespan in humans. The aim of the study was to test whether pharmaceutical concentrations of the glycolytic inhibitor lonidamine are capable of extending lifespan in a nematodal model organism for aging processes, the roundworm Caenorhabditis elegans. Several hundreds of adult C. elegans roundworms were maintained on agar plates and fed E. coli strain OP50 bacteria. Lonidamine was applied to test whether it may promote longevity by quantifying survival in the presence and absence of the compound. In addition, several biochemical and metabolic assays were performed with nematodes exposed to lonidamine. — Lonidamine significantly extends both median and maximum lifespan of C. elegans when applied at a concentration of 5 micromolar by 8% each. Moreover, the compound increases paraquat stress resistance, and promotes mitochondrial respiration, culminating in increased formation of reactive oxygen species (ROS). Extension of lifespan requires activation of pmk-1, an orthologue of p38 MAP kinase, and is abolished by co-application of an antioxidant, indicating that increased ROS formation is required for the extension of lifespan by lonidamine. Consistent with the concept of mitohormesis, lonidamine is capable of promoting longevity in a pmk-1 sensitive manner by increasing formation of ROS.
IMitohormesis appears to be a pathway response preserved by evolution over many species, ranging from yeast cells to nematode worms to humans.
An editorial article in the journal publishing the previous-cited report entitled Alternative mitochondrial fuel extends life span had to say: “In this issue of Cell Metabolism, Ristow and colleagues (Zarse et al., 2012) elucidate a conserved mechanism through which reduced insulin-IGF1 signaling activates an AMP-kinase-driven metabolic shift toward oxidative proline metabolism. This, in turn, produces an adaptive mitochondrial ROS signal that extends worm life span. These findings further bolster the concept of mitohormesis as a critical component of conserved aging and longevity pathways.”
The November 2011 publication Mitochondria, reactive oxygen species, and chronological aging: a message from yeast reports: “As a major intracellular source of reactive oxygen species (ROS), mitochondria are involved in aging and lifespan regulation. Using the yeast chronological aging model, researchers have identified conserved signaling pathways that affect lifespan by modulating mitochondrial functions. Caloric restriction and a genetic mimetic with reduced target of rapamycin signaling globally upregulate the mitochondrial proteome and respiratory functions. — Recent discoveries support the notion that an altered mitochondrial proteome induces mitohormesis. Mitohormesis involves a variety of ROS during several growth stages and extends lifespan in yeast and other organisms. Here we recap recent advances in understanding of ROS as signals that decelerate chronological aging in yeast. We also discuss parallels between yeast and worm hypoxic signaling. In sum, this mini-review covers mitochondrial regulation by nutrient-sensing pathways and the complex underlying interactions of ROS, metabolic pathways, and chronological aging.”
Exploitation of mitohormesis could become an important strategy for prevention and control of heart diseases.
The February 2012 publication Mitochondria as a source and target of lipid peroxidation products in healthy and diseased heart reports: “The heart is a highly oxidativ1e organ in which cardiomyocyte turnover is virtually absent, making it particularly vulnerable to accumulation of lipid peroxidation products (LPP) formed as a result of oxidative damage. 2. Reactive oxygen and nitrogen species are the most common electrophiles formed during lipid peroxidation and lead to the formation of both stable and unstable LPP. Of the LPP formed, highly reactive aldehydes are a well-recognized causative factor in ageing and age-associated diseases, including cardiovascular disease and diabetes. 3. Recent studies have identified that the mitochondria are both a primary source and target of LPP, with specific emphasis on aldehydes in cardiomyocytes and how these affect the electron transport system and Ca(2+) balance. 4. Numerous studies have found that there are functional consequences in the heart following exposure to specific aldehydes (acrolein, trans-2-hexanal, 4-hydroxynonenal and acetaldehyde). Because these LPP are known to form in heart failure, cardiac ischaemia-reperfusion injury and diabetes, they may have an underappreciated role in the pathophysiology of these disease processes. 5. Lipid peroxidation products are involved in the transcriptional regulation of endogenous anti-oxidant systems. Recent evidence demonstrates that transient increases in LPP may be beneficial in cardioprotection by contributing to mitohormesis (i.e. induction of anti-oxidant systems) in cardiomyocytes. Thus, exploitation of the cardioprotective actions of the LPP may represent a novel therapeutic strategy for future treatment of heart disease.”
The 2010 publication Activation of mitochondrial energy metabolism protects against cardiac failure spoke to the same basic points: “Cardiac failure is the most prevalent cause of death at higher age, and is commonly associated with impaired energy homeostasis in the heart. Mitochondrial metabolism appears critical to sustain cardiac function to counteract aging. In this study, we generated mice transgenically over-expressing the mitochondrial protein frataxin, which promotes mitochondrial energy conversion by controlling iron-sulfur-cluster biogenesis and hereby mitochondrial electron flux. Hearts of transgenic mice displayed increased mitochondrial energy metabolism and induced stress defense mechanisms, while overall oxidative stress was decreased. Following standardized exposure to doxorubicin to induce experimental cardiomyopathy, cardiac function and survival was significantly improved in the transgenic mice. The insulin/IGF-1 signaling cascade is an important pathway that regulates survival following cytotoxic stress through the downstream targets protein kinase B, Akt, and glycogen synthase kinase 3. Activation of this cascade is markedly inhibited in the hearts of wild-type mice following induction of cardiomyopathy. By contrast, transgenic overexpression of frataxin rescues impaired insulin/IGF-1 signaling and provides a mechanism to explain enhanced cardiac stress resistance in transgenic mice. Taken together, these findings suggest that increased mitochondrial metabolism elicits an adaptive response due to mildly increased oxidative stress as a consequence of increased oxidative energy conversion, previously named mitohormesis. This in turn activates protective mechanisms which counteract cardiotoxic stress and promote survival in states of experimental cardiomyopathy. Thus, induction of mitochondrial metabolism may be considered part of a generally protective mechanism to prevent cardiomyopathy and cardiac failure.”
A comment on this publication sent me by James P Watson is: “ This is a transgenic mouse model of chemotherapy-induced CHF. The chemotherapy drug is a common FDA approved drug that causes cardiomyopathy when patients are treated with too much of this drug. The mechanism that causes the doxorubicin-induced cardiomyopathy is down-regulation at 3 points of the intra-cytoplasmic Insulin/IGF-1 pathway (Protein Kinase B, Akt, and glycogen synthetase kinase 3). In this experiment, they created a transgenic mouse that over-expressed the mitochondrial protein frataxin, which increases expression of a critical rate-limiting complex in the OxPhos pathway (iron-sulphur-cluster, ISC) and in Kreb’s cycle (aconitase, which is also dependent on ISC). If a human does not produce enough frataxin, this is called Fredrich’s ataxia and you will die in your 30s from heart failure (along with diabetes, muscle wasting, and ataxia). In the frataxin-over expressed mice, there was an induction of mitochondrial metabolism and ROS defense. When given doxorubicin, 85% of the frataxin mice survived instead of 55% in the wild type mice.”
Statins induce mitohormesis, mitochondrial biogenesis and associated positive health effects in cardiac tissue but do not induce mitohormesis and instead induce excessive ROS stress and negative health effects in skeletal muscles.
This interesting observation is reported in the June 2012 publication Opposite effects of statins on mitochondria of cardiac and skeletal muscles: a ‘mitohormesis’ mechanism involving reactive oxygen species and PGC-1. “AIMS: Statins protect against cardiovascular-related mortality but induce skeletal muscle toxicity. To investigate mechanisms of statins, we tested the hypothesis that statins optimized cardiac mitochondrial function but impaired vulnerable skeletal muscle by inducing different level of reactive oxygen species (ROS). METHODS AND RESULTS: In atrium of patients treated with statins, ROS production was decreased and oxidative capacities were enhanced together with an extensive augmentation of mRNAs expression of peroxisome proliferator-activated receptor gamma co-activator (PGC-1) family. However, in deltoid biopsies from patients with statin-induced muscular myopathy, oxidative capacities were decreased together with ROS increase and a collapse of PGC-1 mRNA expression. Several animal and cell culture experiments were conducted and showed by using ROS scavengers that ROS production was the triggering factor responsible of atorvastatin-induced activation of mitochondrial biogenesis pathway and improvement of antioxidant capacities in heart. Conversely, in skeletal muscle, the large augmentation of ROS production following treatment induced mitochondrial impairments, and reduced mitochondrial biogenesis mechanisms. Quercetin, an antioxidant molecule, was able to counteract skeletal muscle deleterious effects of atorvastatin in rat. CONCLUSION: Our findings identify statins as a new activating factor of cardiac mitochondrial biogenesis and antioxidant capacities, and suggest the importance of ROS/PGC-1 signalling pathway as a key element in regulation of mitochondrial function in cardiac as well as skeletal muscles.”
The report highlights the importance of dosage in determining whether ROS stress is in the range where the positive effects of hormesis are realized (where the hormetic response curve is above the line), or the stress is too great (where the hormetic response curve is below the line), and the net health result is negative. The dose response curve must be scaled different for cardiac and muscle tissue. Apparently, a dose of statins with a response above the line for cardiac tissue produces a result below the line for muscle tissue.
A second point of the above publication is the importance of the ROS.PGC-1 signaling pathway in regulation of mitochondrial biogenesis and functions.
For background on PGC-1, see the blog entries PGC-1alpha and exercise, AMPK and longevity and PQQ – activator of PGC-1alpha, SIRT3 and mitochondrial biogenesis. “PGC-1-alpha is a gene co-activator, necessary to turn on the PPARG gene and essential in the metabolic process. PGC-1-alpha (Peroxisome proliferator-activated receptor gamma coactivator 1-alpha) “is a protein that in humans is encoded by the PPARGC1Agene.[1] The protein encoded by this gene is a transcriptional coactivator that regulates the genes involved in energy metabolism. This protein interacts with the nuclear receptorPPAR-gamma, which permits the interaction of this protein with multiple transcription factors. This protein can interact with, and regulate the activities of, cAMP response element binding protein (CREB) and nuclear respiratory factors (NRFs). It provides a direct link between external physiological stimuli and the regulation of mitochondrial biogenesis, and is a major factor that regulates muscle fiber type determination. This protein may be also involved in controlling blood pressure, regulating cellular cholesterol homoeostasis, and the development of obesity(ref).[2]”
Regarding this article, James P Watson had some very interesting comments which he communicated to me privately, as follow.
“This is a very interesting article that basically say that statins are “good for your heart” and “bad for your skeletal muscles”. They also clearly established that this effect was mediated by PGC-1alpha and PGC-1beta in the nucleus and also affected mitochondrial gene expression. IN BOTH NUCLEAR GENES AND MITOCHONDRIAL GENES, THIS EFFECT WAS ABOLISHED BY ANTIOXIDATNS! This is very convincing evidence of an ROS-mediated mitohormetic mechanism in the heart for statins and an opposite mitodestructive mechanism in skeletal muscles with statin therapy.
If true, this is an example of “organ pleiotropy” – i.e. what is good for one organ is bad for another. In medicine, we call this “drug side effects”. In pharmacology, they call this “off target effects”. The study included both an animal study and a human study of both cardiac and skeletal muscle (well done) and also included a statin + quercetin arm to the study. Here are the results summarized:
- 1. Animals: This was actually a mitochondrial biogenesis study
Nuclear co-activators and gene expression;
statin alone
=> reduced PGC-1alpha and PGC-1beta in skeletal muscle => reduced Cox1 and NRF1 gene expression
=> increased PGC-1beta in heart, no change in PGC-1alpha in the heart => increased Cox1 and NRF1 gene expression
statin + anti-oxidant (quercetin)
=> abolished all effects in both cardiac and skeletal muscle
mitochondrial gene expression
statin alone
=> reduced Cytochrome b and pyruvate kinase gene expression in skeletal muscle mitochondria
=> increased Cytochrome b and pyruvate kinase gene expression in cardiac muscle mitochondria
statin + quercetin (antioxidant)
=> abolished all effects in both cardiac and skeletal mitochondria
ROS production
statin alone
=> increased ROS in skeletal muscle
=> reduced ROS in cardiac muscle
statin + quercetin
=> abolished effect in skeletal muscle, no change in cardiac muscle
2, Humans:
This study did not include anti-oxidants. It was just a skeletal muscle vs cardiac muscle biopsy study, done during open heart surgery.”
statins
=> reduced PGC-1alpha, PGC-1beta, SOD1, and SOD2 in skeletal muscle
=> increased PGC-1alpha, PGC-1beta, SOD1, and SOD2 in cardiac muscle
My comment (James P Watson, continuing) on this article and diagram:
This is the first article that I have seen that shows changes in both nuclear gene expression (of extrinsic mitochondrial genes) and intrinsic mitochondrial gene expression with statins and that this occurs via an ROS-dependent manner. This study explains why patients on statins live longer than controls and that this may be a mitohormesis effect, and NOT a cholesterol lowering effect of statins. It also is the first to show “organ pleiotropic” effects of a drug and explains the known side effect of muscle pain due to statin therapy. Taking anti-oxidants to prevent this side effect would abolish the beneficial effect of statins on the heart, so anti-oxidant supplementation would NOT be a good idea for patients on statins.
I really like this diagram, because it has one up arrow for ROS production in the heart and four up arrows for ROS production in skeletal muscle. This clearly shows that statins have a “hermetic ROS dose range” for the heart and a “toxic ROS dose range” for skeletal muscle. This is “proof” that ROS dosing is the clear reason for the difference between hermetic doses and “Denham Harmon doses” of ROS.”
Another publication relating exercise, ROS and the impact of statins is the 2011 publication Atorvastatin treatment reduces exercise capacities in rats: involvement of mitochondrial impairments and oxidative stress. “Physical exercise exacerbates the cytotoxic effects of statins in skeletal muscle. Mitochondrial impairments may play an important role in the development of muscular symptoms following statin treatment. Our objective was to characterize mitochondrial function and reactive oxygen species (ROS) production in skeletal muscle after exhaustive exercise in atorvastatin-treated rats. — Our results show that exhaustive exercise exacerbated metabolic perturbations and ROS production in skeletal muscle, which may reduce the exercise capacity and promote the muscular symptoms in sedentary atorvastatin-treated animals.”
By blocking mitohormesis, regular supplementation with antioxidants may block the beneficial effects of regular exercise.
This is a sobering revelation for those who both regularly exercise and take antioxidant supplements, believing that these interventions work synergistically. A 2009 publication Antioxidants prevent health-promoting effects of physical exercise in humans speaks as to how by blocking mitohormesis, supplementation with Vitamins C and E nullifies the health-producing effects of physical exercise. “Exercise promotes longevity and ameliorates type 2 diabetes mellitus and insulin resistance. However, exercise also increases mitochondrial formation of presumably harmful reactive oxygen species (ROS). Antioxidants are widely used as supplements but whether they affect the health-promoting effects of exercise is unknown. We evaluated the effects of a combination of vitamin C (1000 mg/day) and vitamin E (400 IU/day) on insulin sensitivity as measured by glucose infusion rates (GIR) during a hyperinsulinemic, euglycemic clamp in previously untrained (n = 19) and pretrained (n = 20) healthy young men. Before and after a 4 week intervention of physical exercise, GIR was determined, and muscle biopsies for gene expression analyses as well as plasma samples were obtained to compare changes over baseline and potential influences of vitamins on exercise effects. Exercise increased parameters of insulin sensitivity (GIR and plasma adiponectin) only in the absence of antioxidants in both previously untrained (P < 0.001) and pretrained (P < 0.001) individuals. This was paralleled by increased expression of ROS-sensitive transcriptional regulators of insulin sensitivity and ROS defense capacity, peroxisome-proliferator-activated receptor gamma (PPARgamma), and PPARgamma coactivators PGC1alpha and PGC1beta only in the absence of antioxidants (P < 0.001 for all). Molecular mediators of endogenous ROS defense (superoxide dismutases 1 and 2; glutathione peroxidase) were also induced by exercise, and this effect too was blocked by antioxidant supplementation. Consistent with the concept of mitohormesis, exercise-induced oxidative stress ameliorates insulin resistance and causes an adaptive response promoting endogenous antioxidant defense capacity. Supplementation with antioxidants may preclude these health-promoting effects of exercise in humans.”
Image source “Mitohormesis links physical exercise and subsequent formation of reactive oxygen species to insulin sensitivity and antioxidant defense. Physical exercise exerts ameliorating effects on insulin resistance by increasing mitochondrial formation of reactive oxygen species in skeletal muscle to induce expression of PGC1α, PGC1β, and PPARγ as inducers of insulin sensitivity, as well as superoxide dismutases 1 and 2 and glutathione peroxidase 1, key enzymes of ROS defense. Notably, by blocking exercise-dependent formation of reactive oxygen species due to ingestion of antioxidant supplements, health promoting effects of physical exercise are abolished, and physical exercise fails to promote insulin sensitivity and antioxidant defense in the presence of vitamin C and vitamin E.” I note that this finding seems to be relevant within the hormetic range of the ROS dose-response curve.
The patterns of coupling the taking of antioxidants along with regular physical exercise in the interest of general health is, for many people, deeply entrenched, encouraged by much advertising, and only recently being seriously questioned. The August 2012 publication Does vitamin C and e supplementation impair the favorable adaptations of regular exercise? does not directly discuss mitohormesis but reports: “The detrimental outcomes associated with unregulated and excessive production of free radicals remains a physiological concern that has implications to health, medicine and performance. Available evidence suggests that physiological adaptations to exercise training can enhance the body’s ability to quench free radicals and circumstantial evidence exists to suggest that key vitamins and nutrients may provide additional support to mitigate the untoward effects associated with increased free radical production. However, controversy has risen regarding the potential outcomes associated with vitamins C and E, two popular antioxidant nutrients. Recent evidence has been put forth suggesting that exogenous administration of these antioxidants may be harmful to performance making interpretations regarding the efficacy of antioxidants challenging. The available studies that employed both animal and human models provided conflicting outcomes regarding the efficacy of vitamin C and E supplementation, at least partly due to methodological differences in assessing oxidative stress and training adaptations. Based on the contradictory evidence regarding the effects of higher intakes of vitamin C and/or E on exercise performance and redox homeostasis, a permanent intake of non-physiological dosages of vitamin C and/or E cannot be recommended to healthy, exercising individuals.” – “The discrepancy among studies described above regarding the influence of vitamin C and/or E supplementation on adaptations in redox homeostasis also applies to the effects of antioxidant supplementation on alterations in redox homeostasis in response to acute exercise. In fact, most of the relevant studies have reported that vitamin C and/or E supplementation does not affect redox status [14, 18, 36–40], less studies have reported that attenuates oxidative stress [41–45] and there are even reports indicating a pro-oxidant effect [46, 47]. We believe that this “agreement on disagreement” between responses and adaptations to exercise indicates the inherent complexity of redox biochemistry and signifies the difficulty in providing unidirectional predictions after supplementation with antioxidant agents.” I speculate that these disagreements among studies can be explained by reference to the ROS dose-response curve for hormesis discussed above. .
Long-term antioxidant supplementation, specifically with Vitamin C and alpha-lipoic acid, reduces mitochondrial biogenesis in skeletal muscle tissue by interfering with ROS signaling that triggers mitohormesis.
Antioxidants produce a result similar to that produced by statins as described above, inhibiting mitohormesis, The June 2012 publication Antioxidant supplementation reduces skeletal muscle mitochondrial biogenesisreports “PURPOSE: Exercise increases the production of reactive oxygen species (ROS) in skeletal muscle, and athletes often consume antioxidant supplements in the belief they will attenuate ROS-related muscle damage and fatigue during exercise. However, exercise-induced ROS may regulate beneficial skeletal muscle adaptations, such as increased mitochondrial biogenesis. We therefore investigated the effects of long-term antioxidant supplementation with vitamin E and α-lipoic acid on changes in markers of mitochondrial biogenesis in the skeletal muscle of exercise-trained and sedentary rats. METHODS: Male Wistar rats were divided into four groups: 1) sedentary control diet, 2) sedentary antioxidant diet, 3) exercise control diet, and 4) exercise antioxidant diet. Animals ran on a treadmill 4 d · wk at ∼ 70%VO2max for up to 90 min · d for 14 wk. RESULTS: Consistent with the augmentation of skeletal muscle mitochondrial biogenesis and antioxidant defenses, after training there were significant increases in peroxisome proliferator-activated receptor γ coactivator 1α (PGC-1α) messenger RNA (mRNA) and protein, cytochrome C oxidase subunit IV (COX IV) and cytochrome C protein abundance, citrate synthase activity, Nfe2l2, and SOD2 protein (P < 0.05). Antioxidant supplementation reduced PGC-1α mRNA, PGC-1α and COX IV protein, and citrate synthase enzyme activity (P < 0.05) in both sedentary and exercise-trained rats.”
The March 2012 review study Antioxidants and Skeletal Muscle Performance: “Common Knowledge” vs. Experimental Evidence also shows absence of research support for the popular idea that antioxidant supplementation is a good thing to do associated with fitness exercising. “Antioxidants are assumed to provide numerous benefits, including better health, a reduced rate of aging, and improved exercise performance. Specifically, antioxidants are commonly “prescribed” by the media, supplement industry, and “fitness experts” for individuals prior to training and performance, with assumed benefits of improved fatigue resistance and recovery. This has provoked expansion of the supplement industry which responded by creation of a plethora of products aimed at facilitating the needs of the active individual. However, what does the experimental evidence say about the efficacy of antioxidants on skeletal muscle function? Are antioxidants actually as beneficial as the general populous believes? Or, could they in fact lead to deleterious effects on skeletal muscle function and performance? This Mini Review addresses these questions with an unbiased look at what we know about antioxidant effects on skeletal muscle, and what we still need to know before conclusions can be made.” — “Conclusion: Experimental evidence does not support the “common knowledge” that antioxidant treatment greatly improves exercise performance and recovery. On the contrary, studies with antioxidant supplementations generally show no effect on muscle function during and after exercise. The exception is NAC treatment, which has been found to improve performance during submaximal exercise. The limited effects of ROS/RNS and antioxidants during exercise are unexpected in that increases in ROS/RNS are likely to occur and these are potentially harmful. It appears that muscle fibers are in some way protected against deleterious effects of oxidants during exercise and fibers are generally much more sensitive to exposure to oxidants in the rested state than during fatigue.” While mitohormesis is not mentioned, clearly that explains what is happening.
The March 2012 publication Rationale for antioxidant supplementation in sarcopenia sounds a similar note of caution regarding antioxidant supplementation: “Sarcopenia is an age-related clinical condition characterized by the progressive loss of motor units and wasting of muscle fibers resulting in decreased muscle function. The molecular mechanisms leading to sarcopenia are not completely identified, but the increased oxidative damage occurring in muscle cells during the course of aging represents one of the most accepted underlying pathways. In fact, skeletal muscle is a highly oxygenated tissue and the generation of reactive oxygen species is particularly enhanced in both contracting and at rest conditions. It has been suggested that oral antioxidant supplementation may contribute at reducing indices of oxidative stress both in animal and human models by reinforcing the natural endogenous defenses. Aim of the present paper is to discuss present evidence related to possible benefits of oral antioxidants in the prevention and treatment of sarcopenia. – Conclusion: In summary, there is some evidence that oral antioxidant supplementation may reduce muscle damage, but experimental results are largely preliminary and far to be clinically relevant, at least, as suggestive of positive benefits. In fact, a large body of evidence may indicate extreme cautiousness in taking antioxidant supplementation as preventive measures against aging process and age-related conditions. Further studies are needed to support the widespread practice of oral antioxidant supplementation and to determine appropriate recommendations in elderly. — ” Although this article contains significant discussion on ROS during exercise and the body’s endogenous antioxidant defense system, no mention is made of the key mechanisms of mitohormesis and the actions of Nrf2. Instead, seemingly puzzled, the authors suggest a need for more research to relate sarcopenia to antioxidant use.
In acknowledgement of the existence of mitohormesis, metabolic researchers are beginning to think in terms not only of oxidative stress but also in terms of antioxidant stress, especially associated with the consumption of antioxidants..
The May 2012 review publication The neglected significance of “antioxidative stress”summarizes the situation “Oxidative stress arises when there is a marked imbalance between the production and removal of reactive oxygen species (ROS) in favor of the prooxidant balance, leading to potential oxidative damage. ROSs were considered traditionally to be only a toxic byproduct of aerobic metabolism. However, recently, it has become apparent that ROS might control many different physiological processes such as induction of stress response, pathogen defense, and systemic signaling. Thus, the imbalance of the increased antioxidant potential, the so-called antioxidative stress, should be as dangerous as well. Here, we synthesize increasing evidence on “antioxidative stress-induced” beneficial versus harmful roles on health, disease, and aging processes. Oxidative stress is not necessarily an un-wanted situation, since its consequences may be beneficial for many physiological reactions in cells. On the other hand, there are potentially harmful effects of “antioxidative stress,” especially in the cases of overconsumption of synthetic antioxidants. Antioxidants can neutralize ROS and decrease oxidative stress; however, this is not always beneficial in regard to disease formation or progression (of, e.g., cancer) or for delaying aging.”
Another publication making the same point is the February 2012 e-publication review Antioxidant-induced stress: “Antioxidants are among the most popular health-protecting products, sold worldwide without prescription. Indeed, there are many reports showing the benefits of antioxidants but only a few questioning the possible harmful effects of these “drugs”. The normal balance between antioxidants and free radicals in the body is offset when either of these forces prevails. The available evidence on the harmful effects of antioxidants is analyzed in this review. In summary, a hypothesis is presented that “antioxidant-induced stress” results when antioxidants overwhelm the body’s free radicals.” Clearly, this applies only within the range of normal endogenous ROS loads.
Negative health effects due to inhibition of ROS and consequent inhibition of mitohormesis applies to classical antioxidants, substances like vitamins C and E that act as free radical scavengers. On the other hand many phytosubstances commonly referred to also as “antioxidants” are not in fact free radical scavengers, do not inhibit mitohormesis and exercise positive health effects. Instead such phytosubstances act via activating the body’s endogenous antioxidant defense and stress response system.
Regarding this point, see the blog entry The pivotal role of Nrf2. Part 2 – foods, phyto-substances and other substances that turn on Nrf2.
Speaking also directly to this point is the March 2012 publication Oligomerized lychee fruit extract (OLFE) and a mixture of vitamin C and vitamin E for endurance capacity in a double blind randomized controlled trial. “Antioxidant supplementations are commonly used as an ergogenic aid for physical exercise despite its limited evidence. The study aimed to investigate the effects of a polyphenol mixture and vitamins on exercise endurance capacity. Seventy regularly exercising male participants were randomly assigned to receive oligomerized lychee fruit extract, a mixture of vitamin C (800 mg) and E (320 IU), or a placebo for 30 consecutive days. The study results showed that oligomerized lychee fruit extract significantly elevated the submaximal running time (p = 0.01). The adjusted mean change was 3.87 min (95% CI: 1.29, 6.46) for oligomerized lychee fruit extract, 1.33 (-1.23, 3.89) for the vitamins, and 1.60 (-1.36, 4.56) for the placebo (p = 0.33 in between groups). Oligomerized lychee fruit extract significantly increased the anaerobic threshold by 7.4% (1.8, 13.0). On the other hand, vitamins significantly attenuated VO(2)max by -3.11 ml/kg/m (-5.35, -0.87). Their effects on plasma free radical amount, however, were similar. Our results suggest that a polyphenol-containing supplement and typical antioxidants may have different mechanisms of action and that the endurance-promoting effect of oligomerized lychee fruit extract may not directly come from the scavenging of free radicals but may be attributed to other non-antioxidant properties of polyphenols, which requires further investigation.”
Consistant with the above discussions regarding mitohormesis, researchers are continuing to discover additional important biological roles played by oxidative stress, even extending to sexual signaling.
The August 2012 e-publication Oxidative stress and condition-dependent sexual signals: more than just seeing red relates: “The links between fitness, health, sexual signals and mate choice are complex and subject to ongoing study. In 1999, von Schantz et al. made the valuable suggestion that oxidative stress may be an important missing piece of this complex puzzle. Their suggestion has been enthusiastically tested, with over 300 studies citing their paper, but most effort has concerned carotenoid-based (and to a lesser extent melanin-based) visual signals, predominantly in birds and fishes. Today, we know a great deal more about oxidative stress and related physiology, in both a pathological and regulatory sense, than we did in 1999. We revisit von Schantz et al.’s predictions and, more importantly, highlight novel mechanisms that could link oxidative stress with a range of energetically demanding signals, greatly increasing the scope from visual signalling systems that are usually discussed and nearly always tested. In particular, we argue that differences between individuals in their ability to regulate physiology related to oxidative stress may be an important factor influencing the production of sexual signals and the costs that are incurred from investment.”
Wrapping it up
Mitohormesis, like radiation hormesis exists and is important. Here I have been able to discuss some key aspects of mitohormesis, but there is probably much more to come. My experience in writing this blog entry was the usual one of more questions being raised than answered. The leading edges of science are never neat and nicely wrapped up. James P Watson has argued with me privately, for example, that most all scientifically proven lifespan extension phenomena are due to a mitohormetic principle. And I hope that some combination of he and I will be able to lay that argument out in a blog posting to come.
Me, working up a little mitohormesis in the gym. I also benefit from blogohormesis: Working to get my mind around the subject of this blog entry initially produced a similar experience of nearly-impossible weight and stress. This was slowly replaced by a feeling of wellbeing, one that will be good hopefully until I get deeply into the next blog entry.
Vince





I did not know where else to ask—Art Devany one of the fore-fathers of the Paleo diet (and longevity advocate/scientist) posted that there is anew form of glutathione that can be absorbed in a better manner. I was hoping you could comment on this. The product is called Guardian : http://artdevanyonline.com/guardian.html.
Thank you so very much!!!
Suzette
Vince, are you available as a paid lecturer to speak to groups interested in the science of longevity? I am trying to get our University more involved in basic aging research, and I think that outside help would further this cause.
Stephen Miller
806-778-4213 (Cell)
stephen.miller@responsive-services.com
Stephen
Yes I am. You can e-mail me at vegiuliano@comcast.net or call me 508-358-5629, 508-277-0751 cell.
Vince
Hi
This is a very interesting series of articles. It seems to bring us back to seeking set points of the biological system (and all the different subsystems). Using the analogy with an equilibrium state, it would seem to eliminate almost all strategies that would produce a “linear” improvement. I’m wondering how you evaluate your “firewall” in light of this new understanding. There we have a little of this and some more of that until we reach what appears to be a very large dosing — which for many subsystems might push us past D***.
How do we rationally plan the doses that should be taken of the various supplements?
What measurements might we take of the body and its systems that might guide us?
Thank you for your work and presenting this problem in such a clear light.
Jim Rose
Jim Rose
I agree with your point regarding non-linearity. As to re-evaluating my supplement “firewall,” that is something I have started to think about and need to get into more deeply. As a personal matter I have already halved my daily doses of vitamins C and E and alpha-lipoic acid, and eliminated NAC. I will possibly make additional changes, such as further drastically reducing or eliminating radical-scavenging antioxidants . I have also discontinued with PQQ supplementation which I am conserned may lead to overexpression of PGC1-alpha. The key question, of course, is related to dosages at which effects thake place, a matter not well studied at all. I expect I will modify the published regimen soon
I wish I knew how to answer your last two questions which are excellent ones. I am afraid real answers will take a series of carefully designed studies. Meanwhile on an anecdotal basis with sample size being 1, for myself I am looking to macroscopic indicators such as sense of general health, weight gain/loss, stamina and mental alertness.
Vince
Bravo! Vince
You again have “bucked the conventional wisdom of the free radical theory of aging” and clearly showed that free radicals are “signaling molecules” between the mitochondria and the cytoplasm and are “good” at hormetic doses, not “bad at all doses” as the free radical theory of aging predicts. If Denham Harmon had just done his experiments
at a range of “free radical dosing”, he would have discovered mitohormesis in 1954 and the free radical theory of aging would have NEVER been developed! Hormesis is simply a “U-shaped” or “J-shaped” dodose response curve and free radicals are simply the “drug” that is being dosed. I am going to play “the Devil” and criticize your blog for the sake of stirring up interest, even though I agree with most all of your points.
1. Exercise Dosing for the D*-D** “sweet spot” on your graph.
You have “NO REAL NUMBERS” on the Stochastic Hormetic Relative Risk Graph (Scott, et al, 2009)!
We need numbers for points “b”, D*, D**, and D*** on the graph. We need “real numbers” for the mitohormetic effects of exercise, rotenone, CR, lonidamine. For example, what is the “mitohormetic ROS dosing” for swimming
laps in the pool? (Is 100o meters in 40 minutes = D* and 5,000 meters in 4 hours D**? ). The only “hormetic ROS
dose that I know right now is for rotenone (the pesticide) which is 12.5 nmol for D* and 25 nmol for D**! Despite my strong belief in mitohormesis, however, I don’t think I am going to start ingesting pesticides ! (I would participate in an IRB approved clinical trial for radiation hormesis, however). Please give us “real numbers” for
mitohormesis and not “graphic illustrations with no X-axis data points!
2. Anti-oxidant supplements (Vitamin C, E, etc.) are bad with exercise!
If mitohormesis is “the truth” and anti-oxidant supplements “extinguish” the beneficial effects of mitohormesis
(as demonstrated in several of the articles you quoted), then you must take the radical step of telling us mere mortals that ANTIOXIDANT SUPPLEMENTS are bad for us! Now I personally believe this is a “partial truth”
and will only confuse people who do not really study this subject. We know that our bodies need vitamins. Vitamins are co-factors that are essential for normal cellular molecular enzyme activity and that our own cells do not make these molecules anymore (there are pseudogenes within our DNA for most all of the vitamins, but the mammalian copies of these pseudogenes have been nonfunctional for millions of years. Ex: Vit C became a pseudogene about 12.5-60 million years ago). Also, not all vitamins are antioxidants. For instance, Vitamin A,cB12 and folate. These are not “mitohormetic extinguishers” and will not reduce NRF2 gene up regulation or reduce PGC-1alpha induction of mitochondrial biogenesis. On the other hand, Vitamin C, E, and many of the supplements that are sold in shops and on-line are powerful anti-oxidants that will EXTINGUISH mitohormesis! This is something that the supplement industry has soundly ignored and we are “fighting an up hill battle” to convince the
public of this. Also, we do NOT want to suggest to the public that they stop eating their fruits and vegetables!
There are many important compounds in fruits and vegetables that UPREGULATE NRF2 genes, such as flavones, isoflavones, flavonoids, and the adaptogen herbs that Vince has spoken about with such clarity in his previous blogs on Rhodiola, etc. These polyphenols do have an “anti-oxidant effect” and can extinguish mitohormesis in theory, but that is NOT the main beneficial mechanism of action. There main beneficial mechanism of action is probably via the polyphenols inducing Keap1/NRF2 genes and affecting epigenetics via DNA methylation and histone modifications (I will cover this in a separate commentary).
3. Indirect ROS dose metering (i.e. determining D* to D** dose range for each exercise)
This leads me to the 3rd important point which involves #1 and #2: Determining the “hormetic ROS dose range”
for specific exercises, such as running, biking, swimming, weight lifting, CR, etc. How can we determine “real time
ROS production” which is necessary to determining “hormetic ROS dose”? I have several suggestions, some that are for humor value only and others are serious.
a. We could make mice run for various “ROS doses”, sacrifice the mouse, and do various ROS measurements in
muscle, heart, and brain tissue. The exercise would help us but the mice would not like it.
b. We could make people run for various “ROS dose” distances, sacrifice the people, and do various ROS
measurements of muscle, heart, and brain tissue. The exercise would help the subjects in the experiment and
the MICE WOULD BE HAPPY.
c. Or we could find a ROS measuring tool that did NOT require sacrificing either the mice or the humans. This
would probably be more politically correct! 🙂 One method is measuring aldehydes in exhaled air. (Ex:
Revelar system). Although there is a company selling such a device, I don’t know how accurate it is and the
company is “clueless” about mitohormesis, hawking the device with the old mantra that “all ROS are bad”.
Another possibility would be measuring urinary metabolites of free radical damage. My problem with this
approach is that I DO NOT WANT TO allow “suprahormetic ROS doses” to damage my lipids, proteins, and
DNA! Most all of the urinary tests measure metabolites of damaged molecules. This is NOT
mitohormesis.
In summary, we need more data to fill numbers on the X-axis of the first graph that Vince used in this blog.
I want practical advice that I can use. How far should I run this morning? Should I run till my muscles are sore
with lactic acid? Should I lift weights this afternoon till I reach that “burn pain” that the body builders
talk about with the phrase “no pain, no gain”? How do I get my 8-10 servings of fruits and vegetables and not
counter-act the beneficial effects of exercise via mitohormesis? This is why I want to start a society of experimental
biology for us mere mortals that is akin to the Experimental Aviation Association that the amateur pilots have.
We can create the “data points” for all forms of hormesis, including exercise, radiation, etc. We can
also do “consumer microarrays” to make sure we are getting enough NRF2 gene up regulation of SOD-like scavengers and the Phase II enzymes. We can develop “consumer microarrays” and measure our mRNA for
PGC-1alpha to see if we are adequately up regulating mitochondrial biogenesis. It is time for “evolved thinkers” like Vince Guiliano and those that read this blog to do some practical science besides arm chair science.
This is what I want to do. We are approaching the point that one microarray shared with 100 people could
easily measure 300 mRNAs per consumer at a cost of $100 per person. Let’s do it!
me to take action
upregulation
James REPLY TO JAMES WATSON’S COMMENTS PART 1
Thanks for your complements and very astute comments. Since this is a plain-text field I will comment selectively to your comments IN CAPS.
“Bravo! Vince
If Denham Harmon had just done his experiments at a range of “free radical dosing”, he would have discovered mitohormesis in 1954 and the free radical theory of aging would have NEVER been developed!
QUITE POSSIBLY SO. AMAZING THE QUIRKS THAT SHAPE THE DEVELOPMENT OF SCIENCE
“Hormesis is simply a “U-shaped” or “J-shaped” dodose response curve and free radicals are simply the “drug” that is being dosed. I am going to play “the Devil” and criticize your blog for the sake of stirring up interest, even though I agree with most all of your points.”
OK, GOOD
“1. Exercise Dosing for the D*-D** “sweet spot” on your graph. You have “NO REAL NUMBERS” on the Stochastic Hormetic Relative Risk Graph (Scott, et al, 2009)! We need numbers for points “b”, D*, D**, and D*** on the graph. We need “real numbers” for the mitohormetic effects of exercise, rotenone, CR, lonidamine. For example, what is the “mitohormetic ROS dosing” for swimming laps in the pool? (Is 100o meters in 40 minutes = D* and 5,000 meters in 4 hours D**? ). The only “hormetic ROS dose that I know right now is for rotenone (the pesticide) which is 12.5 nmol for D* and 25 nmol for D**! Despite my strong belief in mitohormesis, however, I don’t think I am going to start ingesting pesticides ! (I would participate in an IRB approved clinical trial for radiation hormesis, however). Please give us “real numbers” for mitohormesis and not “graphic illustrations with no X-axis data points!
I WISH I HAD SUCH NUMBERS OR KNEW WHERE TO GET THEM. AND I DON’T NKOW HOW SIMILAR THE GRAPH IS FROM ONE INDIVIDUAL TO ANOTHER OR HOW IT MAY VARY BY NUMEROUS BODY CONDITIONS. I THINK THIS IS AN AREA WHERE MUCH FUTURE RESEARCH IS JUSTIFIED.
i WILL CONTINUE RESPONSINGIN ADDITIONAL COMMENTS
Vince
RESPONSE TO JAMES WATSON’S COMMENTS PART 2
“2. Anti-oxidant supplements (Vitamin C, E, etc.) are bad with exercise! If mitohormesis is “the truth” and anti-oxidant supplements “extinguish” the beneficial effects of mitohormesis (as demonstrated in several of the articles you quoted), then you must take the radical step of telling us mere mortals that ANTIOXIDANT SUPPLEMENTS are bad for us! Now I personally believe this is a “partial truth” and will only confuse people who do not really study this subject. “
I QUITE AGREE WITH THIS JAMES. THAT IS WHY I HAVE BEEN VERY SLOW TO MODIFY MY PERSONAL SUPPLEMENT REGIMEN OR THAT SUGGESTED IN MY TREATISE.
‘We know that our bodies need vitamins. Vitamins are co-factors that are essential for normal cellular molecular enzyme activity and that our own cells do not make these molecules anymore (there are pseudogenes within our DNA for most all of the vitamins, but the mammalian copies of these pseudogenes have been nonfunctional for millions of years. Ex: Vit C became a pseudogene about 12.5-60 million years ago). Also, not all vitamins are antioxidants. For instance, Vitamin A,cB12 and folate. These are not “mitohormetic extinguishers” and will not reduce NRF2 gene up regulation or reduce PGC-1alpha induction of mitochondrial biogenesis.”
‘
YES, YES – VERY IMPORTANT
“On the other hand, Vitamin C, E, and many of the supplements that are sold in shops and on-line are powerful anti-oxidants that will EXTINGUISH mitohormesis!”
YES, YES, AND THEREIN LIES THE PARADOX OF HOW TO KNOW WHAT BEST PRACTICALLY SUPPORTS PERSONAL HEALTH.
“This is something that the supplement industry has soundly ignored and we are “fighting an up hill battle” to convince the public of this. Also, we do NOT want to suggest to the public that they stop eating their fruits and vegetables! There are many important compounds in fruits and vegetables that UPREGULATE NRF2 genes, such as flavones, isoflavones, flavonoids, and the adaptogen herbs that Vince has spoken about with such clarity in his previous blogs on Rhodiola, etc. These polyphenols do have an “anti-oxidant effect” and can extinguish mitohormesis in theory, but that is NOT the main beneficial mechanism of action. There main beneficial mechanism of action is probably via the polyphenols inducing Keap1/NRF2 genes and affecting epigenetics via DNA methylation and histone modifications (I will cover this in a separate commentary). ”
THANK YOU FOR PICKING UP AND AMPLIFYING THIS POINT JAMES. I BELIEVE IT IS VERY CENTRAL. SINCE THE SUPPLEMENT INDUSTRY ALSO MARKETS NUMEROUS PHYTOSUBSTANCES THAT ACTIVATE NRF2, THEY MIGHT BE WILLING TO PICK UP ON THIS POINT.
RESPONSE TO JAMES WATSON’S COMMENTS PART 3 OF 3
“3. Indirect ROS dose metering (i.e. determining D* to D** dose range for each exercise) his leads me to the 3rd important point which involves #1 and #2: Determining the “hormetic ROS dose range” or specific exercises, such as running, biking, swimming, weight lifting, CR, etc. How can we determine “real time ROS production” which is necessary to determining “hormetic ROS dose”? I have several suggestions, some that are for humor value only and others are serious. a. We could make mice run for various “ROS doses”, sacrifice the mouse, and do various ROS measurements in muscle, heart, and brain tissue. The exercise would help us but the mice would not like it.”
TRUE, BUT SCIENTISTS USUALLY IGNORE MOUSE FEELINGS ANYWAY. MICE ARE NOT A SIGNIFICANT POLITICAL CONSTITUENCY AND DO NOT USUALLY SIT ON RESEARCH GRANT COMMITTES.
“b. We could make people run for various “ROS dose” distances, sacrifice the people, and do various ROS measurements of muscle, heart, and brain tissue. The exercise would help the subjects in the experiment and the MICE WOULD BE HAPPY.”
TRUE, BUT IN OUR CONTEMPORARY SOCIETY THIS WOULD RAISE EYEBROWS AND “BLEEDING HEART” GROUPS WOULD NO-DOUBT OBJECT. COULD GET IN THE WAY OF NIH FUNDING.
“c. Or we could find a ROS measuring tool that did NOT require sacrificing either the mice or the humans. This would probably be more politically correct! One method is measuring aldehydes in exhaled air. (Ex: Revelar system). Although there is a company selling such a device, I don’t know how accurate it is and the company is “clueless” about mitohormesis, hawking the device with the old mantra that “all ROS are bad”.
HMNN. PERHAPS THE REVELAR FOLKS COULD BE ENROLLED, OR OTHER MEDICAL TECHNOLOGY COMPANIES FOR THAT MATTER.
“Another possibility would be measuring urinary metabolites of free radical damage. My problem with this approach is that I DO NOT WANT TO allow “suprahormetic ROS doses” to damage my lipids, proteins, and DNA! Most all of the urinary tests measure metabolites of damaged molecules. This is NOT Mitohormesis.”
YES – NEW TESTS ARE NEEDED. WHAT ABOUT MEASUREMENTS OF EXPRESSION LEVELS OF OTHER METABOLIC INDICATORS SUCH AS PGC1-ALPHA, ALBEIT INDIRECT MEASUREMENTS?
“4 In summary, we need more data to fill numbers on the X-axis of the first graph that Vince used in this blog. I want practical advice that I can use. How far should I run this morning? Should I run till my muscles are sore ith lactic acid? Should I lift weights this afternoon till I reach that “burn pain” that the body builders talk about with the phrase “no pain, no gain”? How do I get my 8-10 servings of fruits and vegetables and not counter-act the beneficial effects of exercise via mitohormesis?”
I FACE THE SAME QUESTIONS AS DO ALL HEALTH-CONSCIOUS PEOPLE. LIFE ITSELF IS A VERY PRAGMATIC PROCESS AND IT SEEMS WE ALL MUDDLE THROUGH BEST WE CAN GIVEN THE KNOWLEDGE WE HAVE TILL WE DIE. BRINGING MORE KNOWLEDGE TO BEAR WILL LIKELY HELP US LIVE BETTER AND LONGER.
“This is why I want to start a society of experimental biology for us mere mortals that is akin to the Experimental Aviation Association that the amateur pilots have. We can create the “data points” for all forms of hormesis, including exercise, radiation, etc. We can also do “consumer microarrays” to make sure we are getting enough NRF2 gene up regulation of SOD-like scavengers and the Phase II enzymes. We can develop “consumer microarrays” and measure our mRNA for PGC-1alpha to see if we are adequately up regulating mitochondrial biogenesis.”
STRIKES ME AS A GREAT IDEA. MY SENSE IS THAT A SUFFICIENT UNDERLYING KNOWLEDGE AND TECHNOLOGY BASE TO MAKE THIS POSSIBLE IS ONLY NOW EMERGING – A BASE THAT GETS US FURTHER AWAY FROM FLAT-WORLD, ALCHEMY OR NARROW COMMERCIAL OR QUACK APPROACHES TO PRACTICAL HEALTH.
“It is time for “evolved thinkers” like Vince Guiliano and those that read this blog to do some practical science besides arm chair science.”
TRUE, ALTHOUGH I DO LOVE MY SWIVEL ARM CHAIR AND NOT EVERYBODY THINKS I AM THAT EVOLVED. MORE THAN ANYTHING, I WOULD LIKE TO SEE ACCELERATION IN WHAT IS HAPPENING. ALREADY IN LESS THAN 30 YEARS WE HAVE SEEN A MAJOR SHIFT IN ATTITUDES TOWARDS HEALTH: DECLINE IN SMOKING, FOCUS ON EXERCISE, CONCERNS WITH JUNK FOODS AND TOXINS LIKE TRANS-AND SATURATED FATS, CLEANER WATER AND AIR, MORE AND MORE PEOPLE TAKING FISH OIL AND VITAMIN D SUPPLEMENTS, PERSONAL MICRO-ELECTRONIC HEALTH MONITORING DEVICES, ETC. NEW FOCUS ON OBESITY. AND WE ARE LIVING MUCH LONGER.
NOW PERHAPS IS THE TIME TO TAKE THE KNOWLEDGE OF HORMESIS AND NRF2 OUT TO THE PUBLIC AND SEE THE CREATION OF A WHOLE BUNCH OF PERSONAL BIOMARKER TESTS THAT ORDINARY PEOPLE CAN USE. I WOULD SIGN UP FOR THAT.
“This is what I want to do. We are approaching the point that one microarray shared with 100 people could easily measure 300 mRNAs per consumer at a cost of $100 per person.”
TRUE.
“Let’s do it!”
OK, LET’S.. TO DO IT WILL TAKE SOME SIGNIFICANT SOCIAL ORGANIZATION AND ACTION. I AM NOT SURE NOW WHERE TO BEST START.
OTHER READERS OF THIS BLOG – WANT TO WEIGH IN ON THIS DIALOG?
Hi
The underlying premise is that mitochondrial health drives the health of the organism.
How well do we know if this is true?
How do we measure mitochondrial health? Numbers in a cell? size? shape? efficiency? could we stress test our mitochondria?
We need a solid biological marker of aging — one that I have seen proposed recently is the time derivative of telomere length. On the face of it, this would seem to take months or maybe years to get a measurement. Are there other (proposable)markers that could be measured within seconds or minutes? Then the microarray chip could look for correlations between interventions and aging and map out our J-shaped curves.
What are the biological markers of aging?
The brain is a special subsystem. It seems to be particularly dependent on the diurnal variations in cortisol and BDNF. A little temporary extra stress and cortisol (e.g. doing something hard for 30 minutes) seems to hormectically induce BDNF and be neuroprotective. However, chronic stress and the resulting excess cortisol seems to kill the neurons in the hippocampus and depress or overwhem the production of BDNF. Some of the relevant hormones can be measured in the saliva — commercially available via the bio-identical hormone industry. So lets add saliva and maybe sweat to the list of measureable body products along with breath, urine and blood.
Bruce Ames is making a “final” research career our of many of these questions of aging. Could he be drawn into this discussion?
Jim Rose
Jim Rose:
Thanks for the provoking thoughts. Some comments on your points:
The underlying premise is that mitochondrial health drives the health of the organism. How well do we know if this is true?
• Jim, I don’t see it that way. Rather I would say that mitochondrial health is an important determining factor in determining overall health, but only one of many. Just like the health of the engine is an important determining factor in determining the health of a car, but only one of many. In both cases lots can go wrong. See http://en.wikipedia.org/wiki/Mitochondrial_disease for an impressive list of how mitochondrial problems can screw up the entire organism.
How do we measure mitochondrial health? Numbers in a cell? size? shape? efficiency? could we stress test our mitochondria?
• Good question. Overall metabolic health is a good macro measure, but I am not sure how to do this best on a micro level.
We need a solid biological marker of aging — one that I have seen proposed recently is the time derivative of telomere length. On the face of it, this would seem to take months or maybe years to get a measurement.
• Problem is, telomere lengths get shorter generally with aging but not necessarily. As I recall, there is research that says in a six month period, one third of a population had their telomeres actually get longer. Are they aging and un-aging?
Are there other (proposable) markers that could be measured within seconds or minutes? Then the microarray chip could look for correlations between interventions and aging and map out our J-shaped curves.
• A very good question. And I get where you are going with this. Possibly selected DNA methylation markers might work. Or protein measurements related to gene activation. Over 100 genes are strongly upregulated with aging and over 100 genes strongly downregulated with aging. Perhaps enough of these could be combined on a single chip to do as you suggest, Hmnn!!! Another kind of research that is needed that probably nobody is doing,
• Actually combining a variety of measures might give better measures of aging than any single one. I remember a carnival guy who claimed accuracy at guessing chronological ages +/- two years with 98% accuracy.
What are the biological markers of aging?
• Good question. Everybody knows macroscopic ones such as baldness, grey hair, wrinkled skin, eventually frailty, etc.
The brain is a special subsystem. It seems to be particularly dependent on the diurnal variations in cortisol and BDNF. A little temporary extra stress and cortisol (e.g. doing something hard for 30 minutes) seems to hormectically induce BDNF and be neuroprotective. However, chronic stress and the resulting excess cortisol seems to kill the neurons in the hippocampus and depress or overwhem the production of BDNF. Some of the relevant hormones can be measured in the saliva — commercially available via the bio-identical hormone industry. So lets add saliva and maybe sweat to the list of measureable body products along with breath, urine and blood.
• Yes
Bruce Ames is making a “final” research career out of many of these questions of aging. Could he be drawn into this discussion?
• Quite possibly. Bruce, are you out there? If so, please chime in. Levels of critical micronutrients?
Vince
Perhaps the following article would be relevant to the extraction of D* etc.
Evidence for hormesis in mutagenicity dose–response relationships by Calabrese et al in MUTATION RESEARCH-GENETIC TOXICOLOGY AND ENVIRONMENTAL MUTAGENESIS Volume: 747 Issue: 1 Pages: 157-157 DOI: 10.1016/j.mrgentox.2012.04.008 Published: AUG 30 2012
Jim Rose
Pingback: Editorial -Bridging the Great Divide | AGING SCIENCES – Anti-Aging Firewalls
Pingback: The Hormetic Wild Animal “Zoo” and Their “Zookeepers” | AGING SCIENCES – Anti-Aging Firewalls
Hi
I am interested in the shape of the hormectic curves — “well being” versus dose — admittedly rather vague.
Do you know of any cases where the whole curve is rather well known?
In particular, I am confused and interested in the idea that there is a linear region. It seems reasonable that when one only has limited data at moderate doses for a noxious agent — one might force the data through zero with a line — for lack of anything better.
However, if we assume that there is a (hormectic) minimum in the curve then the conservative curve-fitting extrapolation is to assume that the curve is quadratic about the minimum. Thus, if the curve is actually linear (say for radiation exposure)– there has to be some scientific reason for linearity.
Does a linear region generally exist? If so, why?
Are there any indications that the curves are actually exponential in the noxious region?
Any hints, clues, references would be appreciated.
Thanks, Jim Rose
Jim
Thanks for your thought-provoking questions. I struggle with them a little here but basically have no good answers. I want to point out that progress in science is often that way – the asking of good questions being more powerful than the coming up with answers. Many basic questions in physics, for example, have survived multiple generations of answers.
“I am interested in the shape of the hormectic curves — “well being” versus dose — admittedly rather vague. ”
It is.
Do you know of any cases where the whole curve is rather well known?”
I don’t, though they may exist. The curve is a conceptual entity which appears to apply very generally. Yet, specific numerical values on the curve could be, and I am afraid likely to be, different among organs in an individual, and different from individual to individual. Further they could be dependent on multiple factors affecting individuals and organs in a time-dependent factor, such as levels of key hormones or inflammatory condition.. We just don’t know.
“In particular, I am confused and interested in the idea that there is a linear region. It seems reasonable that when one only has limited data at moderate doses for a noxious agent — one might force the data through zero with a line — for lack of anything better. — However, if we assume that there is a (hormectic) minimum in the curve then the conservative curve-fitting extrapolation is to assume that the curve is quadratic about the minimum. Thus, if the curve is actually linear (say for radiation exposure)– there has to be some scientific reason for linearity.”
Apparently for radiation damage, the original linear no-threshold model appears to have some 1940s and 1950s emperical research behind it, as measured for radiation levels well above the hormesis range. To me this is contraintuitive since it would seem that a damaged organism would have progressively less capability to protect itself from increasing doses.
I agree that the logical arguments for linearity are extremely weak, basically being that curve fitting is most easily done with a ruler. What we have is extremely complex system dynamics behavior probably poorly represented by a line or even a polynomial function for that matter
“Does a linear region generally exist? If so, why? — Are there any indications that the curves are actually exponential in the noxious region?” —
I suspect they are but can’t cite emperical research to this effect.
“Any hints, clues, references would be appreciated.”
I think the stress-related feedback systems are so multiple and interacting that not even systems dynamics modeling will give us an answer. We don’t know what the formulas and coefficients in such a model should be. So I think the best approach is going to be a. developing good easily measurable biomarkers of stress, Nrf2 expression and stress-related damage, and then b, proceeding emperically with small animal and eventually human models, relating stress levels to Nrf2 expression and biomarkers of damage. We may or may not eventually get that way to standardized quantifiable hormesis curves.
Vince
Pingback: Plant polyphenols –six epigenetic knockout punches against cancers | AGING SCIENCES – Anti-Aging Firewalls
Pingback: Mitochondria in health and aging, and possibilities for life prolongation – Part 1:basics | AGING SCIENCES – Anti-Aging Firewalls
Pingback: Mitochondria Part 2: Mitochondrial Responses to Stress: Mitochondrial Signaling: Survival and Death Pathways | AGING SCIENCES – Anti-Aging Firewalls